
Chemistry: An Introduction to General, Organic, and Biological Chemistry, Books a la Carte Edition (13th Edition)
13th Edition
ISBN: 9780134554631
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.3, Problem 9.24PP
State whether each of the following refers to a saturated or an unsaturated solution:
- A spoonful of salt added to boiling water dissolves.
- A layer of sugar forms on the bottom of a glass of tea and as ice is added.
- A kidney stone of calcium phosphate from in the Kidneys when urine become concentrated.
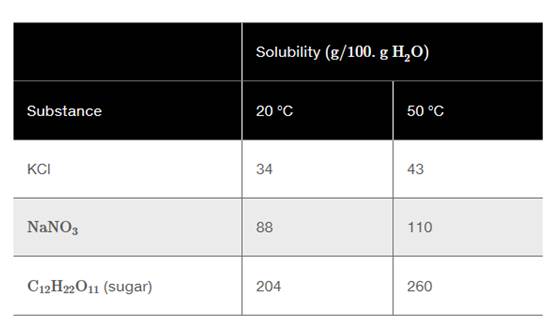
Use the following table for problems 9.25 to 9.28
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Salt water solution has salt as the solute and water as the solvent. If the salt water is diluted, what will happen to the mole of salt in the solution? Which one is it?
1. The moles of salt decrease.
2. The mole of salt remain the same.
3. The moles of salt increase.
4. There is not enough information given.
Complete the table by calculating the amounts of colored after and distilled water required to make the 4mL of each of the following solution concentrations.
% of colored water solution:
25%
50%
75%
100%
Amount of colored water:
Amount of distilled water:
As the temperature of a solution increases, the solubility of salts____and the solubility of gases____
a. doesn’t change, doesn’t change
b. increases, decreases
c. increases, increases
d. decreases, decreases
e. decreases, increases
Chapter 9 Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry, Books a la Carte Edition (13th Edition)
Ch. 9.1 - Prob. 9.1PPCh. 9.1 - Prob. 9.2PPCh. 9.1 - Describe the formation of an aqueous KI solution,...Ch. 9.1 - Describe the formation of an aqueous LiBr...Ch. 9.1 - Prob. 9.5PPCh. 9.1 - Water is a polar solvent and hexane (C6H12)is a...Ch. 9.2 - Prob. 9.7PPCh. 9.2 - NaOHis a strong electrolyte, and CH3OH is a...Ch. 9.2 - Write a balanced equation for the dissociation of...Ch. 9.2 - Write the balanced equation for the dissociation...
Ch. 9.2 - Indicate whether aqueous solutions of each of the...Ch. 9.2 - Prob. 9.12PPCh. 9.2 - Prob. 9.13PPCh. 9.2 - Prob. 9.14PPCh. 9.2 - Calculate the number of equivalents in each of the...Ch. 9.2 - Calculate the number of equivalents in each of the...Ch. 9.2 - An intravenous saline solution contains 154 mEq/L...Ch. 9.2 - An intravenous solution to replace potassium loss...Ch. 9.2 - Prob. 9.19PPCh. 9.2 - A Ringer's solutioncontains the following...Ch. 9.2 - When Michael's blood was tested, the chloride...Ch. 9.2 - After dialysis, the level of magnesium in...Ch. 9.3 - State whether each of the following refers to a...Ch. 9.3 - State whether each of the following refers to a...Ch. 9.3 - Determine whether each of the following solutions...Ch. 9.3 - Determine whether each of the following solutions...Ch. 9.3 - A solution containing 80. g of KClin 200 g of H2O...Ch. 9.3 - A solution containing 80. g of NaNO3 in 75 g of...Ch. 9.3 - Explain the following observations More sugar...Ch. 9.3 - Explain the following observations: And open can...Ch. 9.3 - Predict whether each of the following organic...Ch. 9.3 - Predict whether each of the following organic...Ch. 9.4 - Calculate the mass percent (m/m) for the solute in...Ch. 9.4 - Calculate the mass percent (m/m) for the solute in...Ch. 9.4 - A mouthwash contains 22.5% (v/v) alcohol.If the...Ch. 9.4 - A bottle of champagne is 11% (v/v) alcohol. If...Ch. 9.4 - What is the difference between a 5.0% (m/m)...Ch. 9.4 - What is the difference between a 10.0% (v/v)...Ch. 9.4 - Calculate the mass/volume(m/v) percent for the...Ch. 9.4 - Calculate the mass/volume (m/v) percent for the...Ch. 9.4 - Calculate the grams or milliliters of solute...Ch. 9.4 - Calculate the grams or ml of solute needed to...Ch. 9.4 - Prob. 9.43PPCh. 9.4 - For each of the following solutions, calculate...Ch. 9.4 - Prob. 9.45PPCh. 9.4 - Prob. 9.46PPCh. 9.4 - Calculate the gram of solely needed to prepare...Ch. 9.4 - Calculate the gram of solute needed to prepare...Ch. 9.4 - For each of the following solutions, calculate...Ch. 9.4 - Prob. 9.50PPCh. 9.4 - A patient received 100 mL of a 20.0% (m/v)...Ch. 9.4 - A patient received 250 mL of a 4.0% (m/v) amino...Ch. 9.4 - A patient needs 100. g of glucose in the next 12...Ch. 9.4 - A patient received 2.0 g of NaCl in 8 h. How many...Ch. 9.5 - Prob. 9.55PPCh. 9.5 - Prob. 9.56PPCh. 9.5 - Determine the final volume, in milliliters, of...Ch. 9.5 - Prob. 9.58PPCh. 9.5 - Prob. 9.59PPCh. 9.5 - Prob. 9.60PPCh. 9.5 - You need 500. mL of a 5.0% (m/v) glucose solution....Ch. 9.5 - A doctor ordered 100. mL of 2.0% (m/v)...Ch. 9.6 - Prob. 9.63PPCh. 9.6 - Identify each of the following as characteristics...Ch. 9.6 - A 10% (m/v) starch solution is separated form a 1%...Ch. 9.6 - A 0.1% (m/v) albumin solution is separated form a...Ch. 9.6 - Indicate the compartment (A or B) that will...Ch. 9.6 - Indicate the compartment (A or B) that will...Ch. 9.6 - Will a red blood cell undergo creation, hemolysis,...Ch. 9.6 - Will a red blood cell undergo creation, hemolysis,...Ch. 9.6 - Each of the following mixtures is placed in a...Ch. 9.6 - Prob. 9.72PPCh. 9.6 - 9.73 After her latest dialysis treatment, Michael...Ch. 9.6 - Prob. 9.74PPCh. 9.6 - 9.75 A CaCl2solution is given to increase blood...Ch. 9.6 - 9.76 10 intravenous solution of mannitol is used...Ch. 9 - Prob. 9.77UTCCh. 9 - Prob. 9.78UTCCh. 9 - Prob. 9.79UTCCh. 9 - Prob. 9.80UTCCh. 9 - Prob. 9.81UTCCh. 9 - Whydo lettuces leaves in a salad with after a...Ch. 9 - Prob. 9.83UTCCh. 9 - Prob. 9.84UTCCh. 9 - Prob. 9.85APPCh. 9 - Prob. 9.86APPCh. 9 - Prob. 9.87APPCh. 9 - Prob. 9.88APPCh. 9 - Prob. 9.89APPCh. 9 - Prob. 9.90APPCh. 9 - Prob. 9.91APPCh. 9 - Prob. 9.92APPCh. 9 - Prob. 9.93APPCh. 9 - Prob. 9.94APPCh. 9 - Prob. 9.95APPCh. 9 - Prob. 9.96APPCh. 9 - Prob. 9.97APPCh. 9 - Prob. 9.98APPCh. 9 - Prob. 9.99APPCh. 9 - Prob. 9.100APPCh. 9 - Prob. 9.101APPCh. 9 - Prob. 9.102APPCh. 9 - Prob. 9.103APPCh. 9 - Prob. 9.104APPCh. 9 - Prob. 9.105APPCh. 9 - Prob. 9.106APPCh. 9 - Calculate the final concentration of the solution...Ch. 9 - Calculate the final concentration of the solution...Ch. 9 - Prob. 9.109APPCh. 9 - Prob. 9.110APPCh. 9 - Prob. 9.111APPCh. 9 - Prob. 9.112APPCh. 9 - Prob. 9.113CPCh. 9 - 9.114. In a laboratory experiment, a 15.0-sample...Ch. 9 - Prob. 9.115CPCh. 9 - Prob. 9.116CPCh. 9 - Prob. 9.117CPCh. 9 - Prob. 9.118CPCh. 9 - Prob. 13CICh. 9 - Automobile exhaust is a major cause of air...Ch. 9 - Bleach is often added to a wash to remove stains...Ch. 9 - The compound butyric acid gives rancid butter its...Ch. 9 - Methane is a major component of purified natural...Ch. 9 - The active ingredient in Turns is calcium...Ch. 9 - Tamiflu (oseltamivir), C16H28N2O4, is an antiviral...Ch. 9 - Prob. 20CI
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The most important intermolecular force when the solute CH3-O-CH3 dissolves in water isarrow_forwardWhat happens at the moment of opening a can of soda? Group of answer choices The atmospheric pressure decreases. The gas pressure inside the can decreases. The solubility of carbon dioxide inside the beverage remained the same. The solubility of carbon dioxide inside the beverage increases.arrow_forwardFill in the gaps in the text During a laboratory session, dilute a solution10-2M . You should make 10 ml of solution with a concentration of 10-3M. To make this dilution, take ? ml of the stem solution to ?ml Ringer solutionarrow_forward
- Which of the following is a solution; blood plasma,ciggerate smoke,milk,none of thesearrow_forwardA student is to prepare a 250.-mL solution of 1.0 M glucose (C6H12O6, 180.16 g/mol). Question= A student wishes to prepare 200.-mL of 0.50 M glucose solution. What volume (mL) of the stock 1.0 M glucose solution is needed?arrow_forwardWhen a portion of stock solution is transferred to a beaker, which of the following statements are true? [Select all that apply.] Group of answer choices -The concentration of the solute in the beaker is the same as the concentration in the stock solution. -The volume of the stock solution remains the same. -The total moles of solute in the stock solution decreases. -The concentration of the solute in the beaker is greater than the concentration of the stock solution. -The volume of the stock solution decreases. -The total moles of solute in the stock solution increases. -The concentration of the solute in the beaker is less than the concentration of the stock solution. -The total moles of solute in the stock solution remains the same.arrow_forward
- What would be the molarity of the solution? A saturated solution of 100 mL potassium chloride at 60 C.arrow_forwardIf the Molarity of the solution= 19.12 how do I do the problem below? Now use your molarity in the dilution equation to calculate the number of mL you will need to dilute to make 500 mL of a 0.10 M sodium hydroxide solution.arrow_forwardA 1% solution of morphine has a freezing point depression of 0.08 deg Celsius. How many milligrams of sodium chloride would be necessary to make 20 mL of the solution isotonic? Need only handwritten solution only (not typed one).arrow_forward
- The oven cleaner Easy Off™ is a concentrated solution of sodium hydroxide. With reference to Easy Off™, the term solute refers to Select one: A. the water component of the cleaner B. the sodium hydroxide component of the cleaner C. the use of sodium to dissolve grease stains D. a saturated solution of sodium hydroxide Use the following information to answer the next question. A chemistry student pours 1 L of water into a flask and adds a quantity of solid potassium chloride, stirring the solution. She stoppers the solution and leaves it overnight. In the morning, she notices undissolved solid at the bottom of the flask. The correct statement about this saturated solution is Select one: A. the rates of dissolving and crystallizing are equal B. the solution will become less concentrated as solid continues to settle C. the solution will become more concentrated as time passes D. the solution shows evidence of physical…arrow_forwardA solution of saturated in both potassium chlorate and carbon dioxide gas at room temperature. What happens when the solution is warm to 85° c?arrow_forwardPrepare 100 mL of a 10mM tris solution using a 1M tris stock (include the amount of water in your answer)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY