
General, Organic, and Biological Chemistry: Structures of Life, Books a la Carte Plus Mastering Chemistry with eText -- Access Card Package (5th Edition)
5th Edition
ISBN: 9780133880304
Author: Karen C. Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9.3, Problem 9.24QAP
Use the following table for problems 9.23 to 9.26:
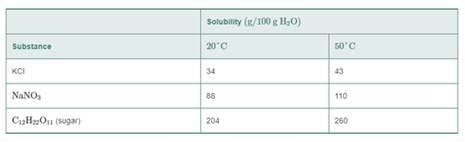
9.24 Determine whether each of the following solutions will be saturated or unsaturated at 50°C:
- adding 25 g of KCl to 50 g of H2O
- adding 150 g of NaNO3 to 75 g of H2O
- adding 80 g of sugar to 25 g of H2O
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How many Calories does a sandwich with 10g of fat, 5g of carbohydrates,and 6g of protein have?
A. 220 B. 123 C. 52 D. 134 E. 540
2. How many grams of water are needed to prepare 100.0 g of a 10.0% by mass cupric sulfate solution?
A. 100g B. 90g C. 10g D. 1000g E. 75g
Household bleach contains 6% sodium hypochlorite. How much household bleach should be combined with 70 L of a weaker 1% hypochlorite solution to form a solution that is 2.5% sodium hypochlorite? Show all work
Solution R is prepared by dissolving 10.00 g of KF in 90.0 g of water. Solution S is prepared by dissolving 4.0 g of KF in 96.0 g of water. Which statement below is true about solutions R and S?
a
Solution S is more dilute than solution R.
b
Solution R is more dilute than solution S.
c
Solution S is more concentrated than solution R.
d
Solution R is less concentrated than solution S.
Chapter 9 Solutions
General, Organic, and Biological Chemistry: Structures of Life, Books a la Carte Plus Mastering Chemistry with eText -- Access Card Package (5th Edition)
Ch. 9.1 - Prob. 9.1QAPCh. 9.1 - Prob. 9.2QAPCh. 9.1 - Prob. 9.3QAPCh. 9.1 - Prob. 9.4QAPCh. 9.1 - Prob. 9.5QAPCh. 9.1 - Water is a polar solvent and hexane is a nonpolar...Ch. 9.2 - Prob. 9.7QAPCh. 9.2 - Prob. 9.8QAPCh. 9.2 - Write a balanced equation for the dissociation of...Ch. 9.2 - Prob. 9.10QAP
Ch. 9.2 - Prob. 9.11QAPCh. 9.2 - Prob. 9.12QAPCh. 9.2 - Prob. 9.13QAPCh. 9.2 - Prob. 9.14QAPCh. 9.2 - Prob. 9.15QAPCh. 9.2 - Prob. 9.16QAPCh. 9.2 - Prob. 9.17QAPCh. 9.2 - Prob. 9.18QAPCh. 9.2 - Prob. 9.19QAPCh. 9.2 - Prob. 9.20QAPCh. 9.2 - Prob. 9.21QAPCh. 9.2 - Prob. 9.22QAPCh. 9.3 - Prob. 9.23QAPCh. 9.3 - Use the following table for problems 9.23 to 9.26:...Ch. 9.3 - Use the following table for problems 9.23 to 9.26:...Ch. 9.3 - Use the following table for problems 9.23 to 9.26:...Ch. 9.3 - Explain the following observations: More sugar...Ch. 9.3 - Explain the following observations: An open can of...Ch. 9.3 - Predict whether each of the following ionic...Ch. 9.3 - Prob. 9.30QAPCh. 9.3 - Determine whether a solid forms when solutions...Ch. 9.3 - Prob. 9.32QAPCh. 9.4 - Calculate the mass percent (m/m) for the solute in...Ch. 9.4 - Calculate the mass percent (m/m) for the solute in...Ch. 9.4 - Calculate the mass/volume percent (m/v) for the...Ch. 9.4 - Calculate the mass/volume percent (m/v) for the...Ch. 9.4 - Prob. 9.37QAPCh. 9.4 - Calculate the grams or milliliters of solute...Ch. 9.4 - A mouthwash contains 22.5% (v/v) alcohol. If the...Ch. 9.4 - A bottle of champagne is 11% (v/v) alcohol. If...Ch. 9.4 - Prob. 9.41QAPCh. 9.4 - Prob. 9.42QAPCh. 9.4 - Prob. 9.43QAPCh. 9.4 - Prob. 9.44QAPCh. 9.4 - Prob. 9.45QAPCh. 9.4 - Prob. 9.46QAPCh. 9.4 - Prob. 9.47QAPCh. 9.4 - Prob. 9.48QAPCh. 9.4 - Prob. 9.49QAPCh. 9.4 - Prob. 9.50QAPCh. 9.4 - Answer the following for the reaction: Mg(s) +...Ch. 9.4 - Prob. 9.52QAPCh. 9.4 - Prob. 9.53QAPCh. 9.4 - Prob. 9.54QAPCh. 9.4 - Prob. 9.55QAPCh. 9.4 - Prob. 9.56QAPCh. 9.4 - Prob. 9.57QAPCh. 9.4 - Prob. 9.58QAPCh. 9.4 - Prob. 9.59QAPCh. 9.4 - Prob. 9.60QAPCh. 9.5 - To make tomato soup, you add one can of water to...Ch. 9.5 - Prob. 9.62QAPCh. 9.5 - Prob. 9.63QAPCh. 9.5 - Prob. 9.64QAPCh. 9.5 - Prob. 9.65QAPCh. 9.5 - Prob. 9.66QAPCh. 9.5 - Prob. 9.67QAPCh. 9.5 - Prob. 9.68QAPCh. 9.5 - Prob. 9.69QAPCh. 9.5 - Prob. 9.70QAPCh. 9.6 - Prob. 9.71QAPCh. 9.6 - Prob. 9.72QAPCh. 9.6 - In each pair, identify the solution that will have...Ch. 9.6 - Prob. 9.74QAPCh. 9.6 - Prob. 9.75QAPCh. 9.6 - Prob. 9.76QAPCh. 9.6 - A 10% (m/v)starch solution is separated from a 1%...Ch. 9.6 - Prob. 9.78QAPCh. 9.6 - Prob. 9.79QAPCh. 9.6 - Prob. 9.80QAPCh. 9.6 - Prob. 9.81QAPCh. 9.6 - Prob. 9.82QAPCh. 9.6 - Prob. 9.83QAPCh. 9.6 - Prob. 9.84QAPCh. 9.6 - Each of the following mixtures is placed in a...Ch. 9.6 - Prob. 9.86QAPCh. 9.6 - Prob. 9.87QAPCh. 9.6 - What is the total positive charge, in...Ch. 9 - Prob. 9.89UTCCh. 9 - Prob. 9.90UTCCh. 9 - Prob. 9.91UTCCh. 9 - Prob. 9.92UTCCh. 9 - Prob. 9.93UTCCh. 9 - Prob. 9.94UTCCh. 9 - Prob. 9.95UTCCh. 9 - Prob. 9.96UTCCh. 9 - Prob. 9.97UTCCh. 9 - Prob. 9.98UTCCh. 9 - Prob. 9.99AQAPCh. 9 - Prob. 9.100AQAPCh. 9 - Prob. 9.101AQAPCh. 9 - Prob. 9.102AQAPCh. 9 - Prob. 9.103AQAPCh. 9 - Prob. 9.104AQAPCh. 9 - Prob. 9.105AQAPCh. 9 - Prob. 9.106AQAPCh. 9 - Prob. 9.107AQAPCh. 9 - Prob. 9.108AQAPCh. 9 - Prob. 9.109AQAPCh. 9 - Prob. 9.110AQAPCh. 9 - Prob. 9.111AQAPCh. 9 - Prob. 9.112AQAPCh. 9 - Prob. 9.113AQAPCh. 9 - What is the molarity of a solution containing 15.6...Ch. 9 - Prob. 9.115AQAPCh. 9 - Prob. 9.116AQAPCh. 9 - Prob. 9.117AQAPCh. 9 - Prob. 9.118AQAPCh. 9 - Prob. 9.119AQAPCh. 9 - Prob. 9.120AQAPCh. 9 - Prob. 9.121AQAPCh. 9 - Calculate the boiling point of each of the...Ch. 9 - Prob. 9.123AQAPCh. 9 - Prob. 9.124AQAPCh. 9 - Prob. 9.125AQAPCh. 9 - Prob. 9.126AQAPCh. 9 - Prob. 9.127AQAPCh. 9 - Prob. 9.128AQAPCh. 9 - Prob. 9.129CQCh. 9 - Prob. 9.130CQCh. 9 - Prob. 9.131CQCh. 9 - Prob. 9.132CQCh. 9 - Prob. 9.133CQCh. 9 - 9.134 A solution is prepared by dissolving 22.0 g...Ch. 9 - Prob. 9.135CQCh. 9 - Prob. 9.136CQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the partial pressure of CO2 (in atm) above a solution if if 8.86 g are dissolved in 371.7 mL of water? KH for CO2 is 0.0310 M/atm. Give your answer to 3 sig figs .arrow_forwardWhen 5mL of vinegar is added to 10 g of baking soda, it produces a bub- bling liquid containing sodium acetate, carbon dioxide and water. What is the most effective way to increase the amount of sodium acetate in less time? a. Add more baking soda b. Add more concentrated vinegar c. Add boiling water d. Add vinegar vaporsarrow_forwardBarium Hydroxide has a solubility of 4.68 g Ba(OH)2 in 100. g H2O at 25°C. Determine if each of the following solutions will be saturated or unsaturated at 25°C: (9.3) a) adding 32 g of Ba(OH)2 to 990 g of H2O b) adding 7.0 g of Ba(OH)2 to 125 g of H2O c) adding 22 g of Ba(OH)2 to 350. g of H2Oarrow_forward
- When 3.00 g of sodium hydroxide (NaOH) was dissolved in 100.00 g of water a value of 11.00oC was obtained for ΔT. 1. Calculate the molarity of the sodium hydroxide solution? _____ 2. Calculate the value (calories) for the heat of solution of 3.00 g of NaOH? ______ 3. Calculate the number of calories that would be produced if one mole of sodium hydroxide was dissolved. (ΔHsolnNaOH)? ______arrow_forwardQ13.58) How much dry solute would you take to prepare each of the following solutions from the dry solute and the solvent? a) 124 mL of 0.115 M NaNO3 b) 128 g of 0.200 m NaNO3 c) 128 g of 1.0 % NaNO3 solution by massarrow_forward11. The heat of solution ( Δ Hsoln) is the algebraic sum of the heat of mixing (Δ Hmix), the heat of solvent (Δ Hsolvent) and the heat of solute (Δ Hsolute). Which of these four enthalpy changes can only be exothermic? W) Δ Hsoln X) Δ Hmix Y) Δ Hsolvent Z) Δ Hsolute Group of answer choices A.) W B.) Z C.) Y & Z D.) Xarrow_forward
- Chemistry Using the vale of 300 mOsmol.L-1 for plasma answer the following questions: A 2.0 L solution contains 18 mmol of sodium chloride (i = 1.8) 20 mmol of potassium chloride (i = 1.8) 10 mmol of calcium chloride (i = 2.5) a) Is the solution isotonic, hypotonic or hypertonic? b) If it is not isotonic, how could the solution be made isotonic with blood using either glucose or water? If using water to make the solution isotonic give the volume to be added in mL If using glucose to make the solution isotonic give the amount to be added in mmol. Please use water to make the solution isotonicarrow_forwardIn order to prepare 50.0 mL 0f 0.100 M C12H22O11 sucrose you will add grams of sucrose to mL of water.arrow_forwardPart G At 60oC, NaNO3 has a solubility of 110 grams NaNO3 per 100 grams of water. How many grams of NaNO3 are needed to prepare a saturated solution using 50. grams of water? 55 grams NaNO3 110 grams NaNO3 40. grams NaNO3 50. grams NaNO3arrow_forward
- What is the molarity of a solution in which 100.0 mL of 1.0 M KCL is diluted to 1.0 L? A. 0.10 M B. 1.0 M C. 10.0 Marrow_forwardBrine is a preservative for vegetables, fish, fruit, and meat. It prevents the growth of bacteria. What is the concentration in ppm of the solution made by dissolving 2.5 g of NaCl in enough water to make mL of brine solution? Theobromine is a bitter alkaloid of cacao plant. It is found in chocolate, leaves of the tea plant, and cola nut. How many grams of theobromine (C7H8N4O2) should be dissolved in 373 g water to make 3.75 m theobromine solution? How many grams of table sugar (C12H22O11) must be added to 200 g H2O to raise the boiling point of water from 100 °C to 102 °C?arrow_forwardHow many grams of oxygen and carbon dioxide are consumed and formed, respectively, during the combustion of 100 grams of this hydrocarbon?ethylene, HC=CH How many grams of molecular oxygen are used when a 5.2 g sample of acetylene CH is burned according to the balanced equation?CH + 2.5 O → 2 CO + HO For the following substance, calculate the freezing point in degrees Celsius for 100 grams in 1000 grams of water.Sucrose CHO, a common sweetener For the following substance, calculate the boiling point in degrees Celsius for 100 grams in 1000 grams of water.NaPO, a saltarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY