
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter F, Problem 2PP
Interpretation Introduction
Interpretation:
The products of the given reactions are to be given.
Concept introduction:
Thiols and disulphides undergo
This can be depicted as:
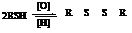
Sulphur ylide acts as a nucleophile and attacks the carbonyl carbon of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Problem 3.3 Draw mechanisms for each of the following substitution reactions.
Remember that in each case, loss of the leaving group is preceded by coordination
of the leaving group to a Lewis acid such as H+.
(a)
(b)
CH3
CH3
HCl
H3C-
OH
H3C-
Cl
CH3
CH3
OH
ZnCl2
Ar
Ar
Ar
Ar
HO
Problem Three
a) Identify the following as nucleophile or electrophile:
iii) C,H,CHO
vi) HOC₂H₂
i) C,H5NH,
iv) (C$Hs)₂CO
ii) (C₂Hs)3N
v) (C₂H5)₂CH+
viii) CH,SH
vii) CN
ix) CH₂CH₂CH₂CH₂ Br
b) i) Draw the curly arrow mechanism for the reaction between v) and vii)
x) (CH3)3CBr
ii) of the two C atoms involved do any change their hybridisation? If so, how?
PROBLEM 3
Suggest mechanisms for these eliminations. Why does the first give a mixture
and the second a single product?
OH
OH
H₂PO4
heat
X³
OH
48% HBr
heat
64% yield, 4:1 ratio
Chapter F Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- OChem Question: Starting with propyne and using any other required reagents, show how you would synthesize (2R, 3R)- and (2S, 3S)-2,3-dibromobutane (as a racemic form)arrow_forwardPractice Supply the missing products, reagents or starting materials as necessary. 1) NaCN 2) НCI а. OH b. 1) NABD4 2) HC, H-О с.arrow_forwardProblem 1: It is desired to prepare polyester with the number average molecular weight, Mn- 5000 g/mol by reacting of 1 mol butane-1,4 diol (HO-CH2-CH2-CH2-CH2-OH) with 1 mol of adipic acid (HOOC-CH2-CH2-CH2-CH2-COOH). (a) Write chemical equation illustrating synthesis of the polyester. (b) Calculate the value of the monomer conversion at which the reaction should be stopped to obtain this polymer, assuming perfect stoichiometric balance. What would be polydispersity index for the polymer obtained? (c) Assuming that 0.5 mol % of the diol is lost to polymerization by a side reaction (dehydratation to a non-reactive substance), what would be the value of Mn if the reaction was carried out to the same extent as in part (b). (d) For the polymerization process value of polymerization rate constant is 1.5*102 L/(mol*s) at 100° C. How long would it take to prepare the polyester with the number average molecular weight, M, 5,000 g/mol at 100 ° C? There is no diol loss for this part of…arrow_forward
- Identify the products of the following reaction. A) H 1) MCPBA 2) H3O+ H3C H3C HO OH H CH3 HO OH B) H&C H H3C CH3 H3C H3C HO OH H CH3 OH D) H3C H H3C CH3 (Select all that apply) U Aarrow_forwardWhat is the major product of the reaction shown below? (1) Li (2) H*/H2O (A) (B) OH (C) (D) OCompound A ) Compound D Compound B Compound Carrow_forwardProblem 4 Complete these acid-base equations. Use the curved arrow method to show the electron movement in the reactions. (a) :NH, (b) CH₂O: H₂O H₂O conjugate acid : NH3 conjugate acid CH₂OH + conjugate base HO: conjugate base H₂Oarrow_forward
- please help with OChem question Predict the oxidative product(s) of 2-methyl-6-octyn-2-ene after undergoing ozonolysis with O3, H2O2arrow_forwardProblem 18 of 50 Submit Curved arrows are used to illustrate the flow of electrons. Follow the arrows and draw the product formed in this reaction. Include all lone-pairs. Ignore any inorganic byproducts. HNO3 H2SO4 مل علم Нarrow_forwardDraw the product of the reaction shown below. Ignore inorganic byproducts. NaH, PhCH2Cl DMSO OH Drawing Q Problem Aton an Draw carrow_forward
- Practice Questiuns Organic Chem 1 Qive the product(s) of following reation. Br C2Br CいeS4 CH2 SH a) Ki la). B.) SH Br D.mixture of BABarrow_forwardProblem 4. Each of the following reactions is displaced to the right. Make a list of all the Bronsted acids that appear in these equations with these acids arranged according to decreasing acid strength. Make a similar list for Bronsted bases. (a) HCO3 + OH H2O + CO32- (b) HC2H3O2 + HS = H2S + C2H3O2" (c) H2S + CO32- = HCO3 + HS- (d) HSO4 + C2H3O2 HC2H3O2 + SO42-arrow_forwardMolecular oxygen (O2), Jones reagent (H2CrO4) and hydrogen peroxide (H2O2) are three common oxidizing agents capable of converting aldehydes to carboxylic acids. Among these three, which one is not a green reagent and why? short and clear answer pleasearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY