
Organic Chemistry, 12e Study Guide/Student Solutions Manual
12th Edition
ISBN: 9781119077329
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter F, Problem 4PP
Interpretation Introduction
Interpretation:
The compound British Anti – Lewisite (BAL) is to be synthesised from allyl alcohol.
Concept introduction:
The compound British Anti – Lewisite (BAL) is used as inorganic mercury, which is effective in protecting the kidney from 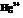 . In BAL, both the thiol groups
. In BAL, both the thiol groups 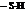 lose hydrogen and binds with the
lose hydrogen and binds with the 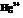 .
.
Addition of bromine across the double bond is done via electrophilic addition reaction. This can be shown as follows:
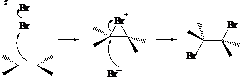
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Need help with chemistry question
Keeping in mind the leveling effect, can the following species be used as a reactant in ethanamine (CH,CH,NH,)?
(1) ©OH
(c) O,
´CH3
(d) ONH2
(e) O
ʼNH4
(a) c
(b)
HCI
Solve both part and detail solution
Chapter F Solutions
Organic Chemistry, 12e Study Guide/Student Solutions Manual
Knowledge Booster
Similar questions
- Show all working explaining detailly each step. Answer should be type written using a computer Keyboard!arrow_forwardplease answer them correctlyarrow_forwardWhy does structure have 6 pi bonds? I understand Huckel's rule which determines if a structure is aromatic or not. Please explain which structures are aromatic according to the options givenarrow_forward
- 6.2.Q (A) it is the major product of the following Stork reaction? (C) (B) Sue St Stork Reaction (D) ?arrow_forwardQuestions molian 1015MSC Chemistry of Biological Systems II 1. A compound of molecular formula CsH₁00 forms a yellow precipitate with 2,4-dinitrophenylhydrazine reagent and a yellow precipitate with reagents for the iodoform test. a) Draw the structural formulae on a piece of paper, take a picture of them and upload the image below. b) Name the two (2) compounds that fit these tests. bns zabyrisblaarrow_forwardNonearrow_forward
- 7.1 Provide the line structures of the products formed in the following reactions. One block may represent more than one product. (a) HBr Diethyl ether (b) H,0, H*arrow_forwardGive detailed Solution with explanation needed (Give answer all sub parts if you not then don't give answer) I will give you a good reviewarrow_forwardQuestion 8 of 75 Provide the correct IUPAC name for S»Cls. (1) (11) (1II) (IV) tri- tetra- di- hepta- octa- mono- heха- penta- chloride sulfite chloride sulfur sulfide chlorite chlorate hydrate acid Deletearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY