
Concept explainers
(a)
Interpretation:
The structure for the given trivial name is to be drawn, and the correct IUPAC name is to be written.
Concept introduction:
The trivial names are commonly used names which are not systematic ones. Many trivial names are accepted by IUPAC. Nitriles are derivatives of
In naming organic compounds, the
Number the carbon chain in a way that the functional group and the substituents attached get the lowest number. The position of the functional group and substituents on the parent chain or ring is indicated by the respective locant number just before the suffix. Prefixes are used to denote the number of identical substituents. The substituents are written in alphabetical order when writing the IUPAC name.
Answer to Problem F.30P
The structure and IUPAC name for the given trivial name is
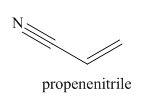
Explanation of Solution
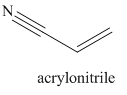
The given trivial name is acrylonitrile.
The given name indicates that the high priority functional group is cynide. The trival name acrylonitrile correlates the derivative of acrylic acid. Structure for acrylic acid is
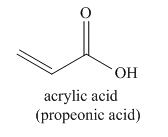
Nitriles are derived from acids where
The main chain in this molecule is of three carbon atoms thus the root name is propan, and the suffix nitrile is added to the root name. The main chain is numbered starting from the carbon atom of the nitrile group. The main chain has a C=C bond; thus the IUPAC name for acrilonitrile is propenenitrile.
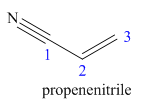
The structure for the given molecule is drawn based on the functional group, and the IUPAC named is written by adding locators to the substituents.
(b)
Interpretation:
The structure for the given trivial name is to be drawn, and the correct IUPAC name is to be written.
Concept introduction:
The trivial names are commonly used names which are not systematic ones. Many trivial names are accepted by IUPAC. Nitriles are derivatives of carboxylic acid. The trivial names of nitriles are deriving from the trivial names of the analogous carboxylic acids.
In naming organic compounds, the functional groups other than the highest priority functional groups are treated as substituents. The root name is established by identifying the longest carbon chain or a ring containing functional group. Remove the e from the normal ane, ene, or yne ending, and add the suffix that corresponds to the highest-priority functional group. Nitriles are the derivatives of carboxylic acid where
Number the carbon chain in a way that the functional group and the substituents attached get the lowest number. The position of the functional group and substituents on the parent chain or ring is indicated by the respective locant number just before the suffix. Prefixes are used to denote the number of identical substituents. The substituents are written in alphabetical order when writing the IUPAC name.
Answer to Problem F.30P
The structure and IUPAC name for the given trivial name is
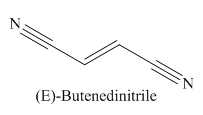
Explanation of Solution
The given trivial name is fumaronitrile.
The given name indicates that the high priority functional group is cynide. The trival name fumaronitrile correlates to the derivative of fumaric acid (dicarboxylic acid). Structure for fumaric acid is
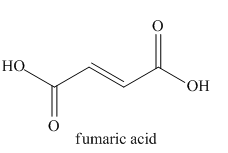
Nitriles are derived from acids where
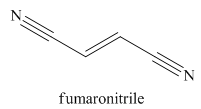
The main chain in this molecule is of four carbon atoms; thus the root name is butan. The structure has two cynide groups at both terminals; hence the suffix dinitrile is added to the root name. The main chain is numbered starting from the carbon atom of the nitrile group.
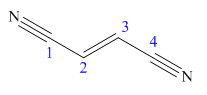
The main chain has a C=C bond; thus the IUPAC name for fumaronitrile is (E) butenedinitrile.
The structure for the given molecule is drawn based on the functional group, and the IUPAC named is written by adding locators to the substituents.
(c)
Interpretation:
The structure for the given trivial name is to be drawn, and the correct IUPAC name is to be written.
Concept introduction:
The trivial names are commonly used names which are not systematic ones. Many trivial names are accepted by IUPAC. Nitriles are derivatives of carboxylic acid. The trivial names of nitriles are deriving from the trivial names of the analogous carboxylic acids.
In naming organic compounds, the functional groups other than the highest priority functional groups are treated as substituents. The root name is established by identifying the longest carbon chain or a ring containing functional group. Remove the e from the normal ane, ene, or yne ending, and add the suffix that corresponds to the highest-priority functional group. Nitriles are the derivatives of carboxylic acid where
Number the carbon chain in a way that the functional group and the substituents attached get the lowest number. The position of the functional group and substituents on the parent chain or ring is indicated by the respective locant number just before the suffix. Prefixes are used to denote the number of identical substituents. The substituents are written in alphabetical order when writing the IUPAC name.
Answer to Problem F.30P
The structure and IUPAC name for the given trivial name is
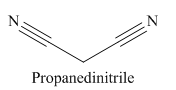
Explanation of Solution
The given trivial name is malonitrile.
The given name indicates that the high priority functional group is cynide. The trival name malonitrile correlates to the derivative of malonic acid (dicarboxylic acid). Structure for malonic acid is
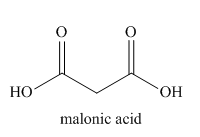
Nitriles are derived from acids where
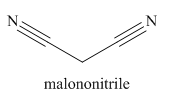
The main chain in this molecule is of three carbon atoms; thus the root name is propane. The structure has two cynide groups; hence the suffix dinitrile is added to the root name. The main chain is numbered starting from the carbon atom of nitrile group.
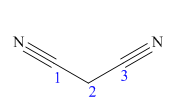
Hence the IUPAC name for malonitrile is propanedinitrile.
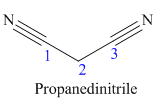
The structure for the given molecule is drawn based on the functional group, and the IUPAC named is written by adding locators to the substituents.
(d)
Interpretation:
The structure for the given trivial name is to be drawn, and the correct IUPAC name is to be written.
Concept introduction:
The trivial names are commonly used names which are not systematic ones. Many trivial names are accepted by IUPAC. Nitriles are derivatives of carboxylic acid. The trivial names of nitriles are deriving from the trivial names of the analogous carboxylic acids.
In naming organic compounds, the functional groups other than the highest priority functional groups are treated as substituents. The root name is established by identifying the longest carbon chain or a ring containing functional group. Remove the e from the normal ane, ene, or yne ending, and add the suffix that corresponds to the highest-priority functional group. Nitriles are the derivatives of carboxylic acid where
Number the carbon chain in a way that the functional group and the substituents attached get the lowest number. The position of the functional group and substituents on the parent chain or ring is indicated by the respective locant number just before the suffix. Prefixes are used to denote the number of identical substituents. The substituents are written in alphabetical order when writing the IUPAC name.
Answer to Problem F.30P
The structure and IUPAC name for the given trivial name is
Explanation of Solution
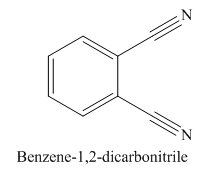
The given trivial name is phthalonitrile.
The given name indicates that the high priority functional group is cynide. The trival name phthalonitrile correlates to the derivative of phthalic acid (dicarboxylic acid). Structure for phthalic acid is
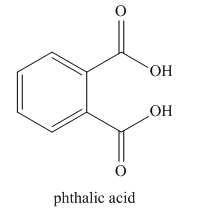
Nitriles are derived from acids where
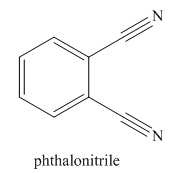
The parent ring in this molecule is benzene ring; thus the root name is benzene. The structure has two cynide groups attached directly to the ring; hence the suffix dicarbonitrile is added to the root name. The ring is numbered such that carbon atoms attached to nitrile groups get least number.
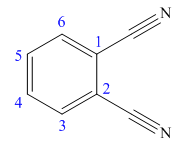
The IUPAC name for phthalonitrile is bemezene-1, 2-dicarbonitrile because two cynide groups are at C1 and C2.
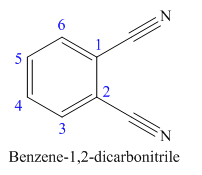
The structure for the given molecule is drawn based on the functional group, and the IUPAC named is written by adding locators to the substituents.
(e)
Interpretation:
The structure for the given trivial name is to be drawn, and the correct IUPAC name is to be written.
Concept introduction:
The trivial names are commonly used names which are not systematic ones. Many trivial names are accepted by IUPAC. Nitriles are derivatives of carboxylic acid. The trivial names of nitriles are deriving from the trivial names of the analogous carboxylic acids.
In naming organic compounds, the functional groups other than the highest priority functional groups are treated as substituents. The root name is established by identifying the longest carbon chain or a ring containing functional group. Remove the e from the normal ane, ene, or yne ending, and add the suffix that corresponds to the highest-priority functional group. Nitriles are the derivatives of carboxylic acid where
Number the carbon chain in a way that the functional group and the substituents attached get the lowest number. The position of the functional group and substituents on the parent chain or ring is indicated by the respective locant number just before the suffix. Prefixes are used to denote the number of identical substituents. The substituents are written in alphabetical order when writing the IUPAC name.
Answer to Problem F.30P
The structure and IUPAC name for the given trivial name is
Explanation of Solution
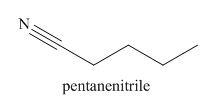
The given trivial name is valeronitrile.
The given name indicates that the high priority functional group is cynide. The trival name valeronitrile correlates to the derivative of valeric acid. Structure for valeric acid is
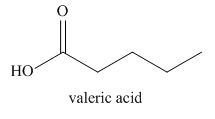
Nitriles are derived from acids where
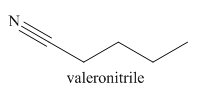
The main chain in this molecule is of five carbon atoms; thus the root name is pentane. The structure has a cynide group; hence the suffix nitrile is added to the root name. The main chain is numbered starting from the carbon atom of nitrile group.
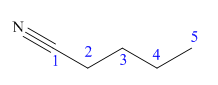
Hence the IUPAC name for valronitrile is pentanenitrile.
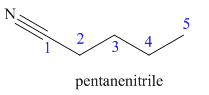
The structure for the given molecule is drawn based on the functional group, and the IUPAC named is written by adding locators to the substituents.
Want to see more full solutions like this?
Chapter F Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Draw the structural formula of trichloroethene, a metal degreaser that was once used to decaffeinate coffee. Why isn't the IUPAC name for this substance 1, 1, 2-trichloroethene?arrow_forwardIn each of the organic compound classes given below, write a molecule with a total of 16 carbons (by express formula), with at least one aromatic group and one alkyl group in its structure. Name each structure you write according to the IUPAC system.g) Write down and name a dithiol molecule.h) Write down and name a lactone molecule.i) Write and name a 3,4-unsaturated aldehyde molecule.arrow_forwardwrite a molecule with a total of 16 carbons with at least one aromatic group and one alkyl group in its structure to each of the given classes of organic compounds(with an open formula). Name each structure you write according to the IUPAC system. a) write and name a lactone molecule.b) write and name a 3,4-unsaturated aldehyde molecule.c) write and name a molecule of corboxylated acid anhydride.d) write and name a semi-ketal molecule.e) write and name an n, n-diarylamide molecule.arrow_forward
- Draw the structure of a.) 3-ethyl-5-methyloctane and b.) 5-tert-butyl-3-ethyl-6-isopropylnonane.arrow_forwardGive correct detailed Solution of all sub partsarrow_forwardIn each of the following classes of organic compounds, write a molecule with a total of 16 carbons (by express formula) with at least one aromatic group and one alkyl group in its structure. Name each structure you write according to the IUPAC system. a) Write down and name a 3,4-unsaturated aldehyde molecule. b) Type and name a carboxylic acid anhydride molecule.arrow_forward
- A.)illustrate the condensed structural formula of 1-chloro-2,4-dinitronapthalene. Use the dotted patterns as your guide in spacing and creating lines. B.) Illustrate the structure of the organic compound of 3-phenyl-2-ethyl-aniline. Used the dotted patterns as your guide.arrow_forward1. Draw structure of the following compounds? A) ethyldimethylamine B) N,N-dimethylbutanamide C) 2-chlorobutanoic acidarrow_forwardDraw the structure of each of the compounds listed in the picture.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



