
Concept explainers
Practice Problem H.5
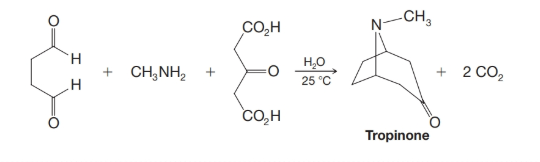
Many alkaloids appear to be synthesized in plants by reactions that resemble the Mannich reaction (Section 19.8). Recognition of this (by R. Robinson in 1917) led to a synthesis of tropinone that takes place under “physiological conditions,” that is, at room temperature and at pH values near neutrality. This synthesis is shown here (and discussed in “Why These Topics Matter” at the end of Chapter 19). Propose reasonable mechanisms that account for the overall course of the reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter H Solutions
ORGANIC CHEMISTRY-ETEXT REG ACCESS
Additional Science Textbook Solutions
Fundamentals of Heat and Mass Transfer
Organic Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry: The Central Science (14th Edition)
Organic Chemistry (9th Edition)
Organic Chemistry (8th Edition)
- Problem 5: Arrartge the following isomeric amines in the order öf increasing boiling point. Provide a succinct rationale A picture or a scheme is worth significantly more than words... NH2 (X) () (Z)arrow_forward2 Question: a) Show the mechanism of synthesis of an amide from an ester and a primary amine: b) The ester responsible for the aroma of the rum has the following structural formula: CH3 H-C-CH2-0-C-CH3 || CH3 Write down the complete chemical reaction (detailed mechanism) that makes it possible to obtain the ester described above, starting from the carboxylic acid (use SOCI, to obtain the acyl chloride).arrow_forwardHO Craven a Structure indicate. in the structure which is the most acidic frotton. Show efficient bose that can be cen tised to abstract each proton and generate the enolate without complications ( Shove the mechanism) Don't we out as a base. مارا GIVE HAND WRITTEN ANSWER WITH EXPLANATIONarrow_forward
- 3) Predict the products of a proton transfer reaction between the two species below and draw the mechanism. NaNH,arrow_forwardCan you please draw and show the mechanism for the changes to help with understanding how phenolpthalein changes rather than just words? As little hard to follow.arrow_forwardProvide detailed (arrow pushing) mechanism for the following reaction. Whereappropriate indicate Lewis acid and base for each step, and whether they are alsoBronsted acids and bases (LB/BA, LA/BA etc.) Indicate the number of steps in themechanism and the number of intermediates.arrow_forward
- Which starting materials will result in the below product through a reductive amination reaction? The correct answer is C, but please explain why:)arrow_forwardRank the Basicity of the following sets of compounds.arrow_forwardElectrostatic potential maps of anisole and thioanisole are shown. Which do you think is the stronger acid, p-methoxybenzoic acid or p-(methylthio)benzoic acid? Explain.arrow_forward
- answer IS Predict the major product of the following process. 1) LDA i ? 2) 3) H3O+arrow_forwardPROBLEM Predict the product(s) and propose a mechanism for each of the following reactions: 19-31 (a) (b) + HO H+ catalyst + OH HO OH H+ catalystarrow_forwardHello, May get help with this organic chemistry please? Question: Synthesize the following aromatic from benzenearrow_forward
