
Organic Chemistry
12th Edition
ISBN: 9781118875766
Author: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter H, Problem 6PP
Practice Problem H.6
Papaverine has been synthesized by the following route:
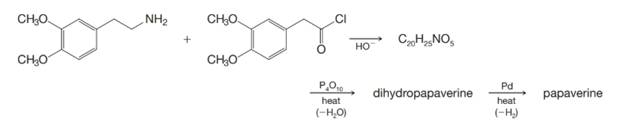
Outline the reactions involved.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Problem B: Dicholorodiflouromethane, CCl2F2, a known refrigerant, can be produced by the chemical equation shown below. Consider the following: atomic mass of C is 12, Cl is 35, and F is 19.
CH4 + Cl2 -> CCl4 + HCl (not balanced)
CCl4 + HF -> CCl2F2 + HCl (not balanced)
1. Assuming that all the CCl4 produced in the first reaction is consumed in the second, how many moles of Cl2 must be consumed in the first reaction to produce 2.25kg CCl2F2 in the second? Show the complete solution
A. 56.24 mol Cl2
B. 56.25 mol Cl2
C. 56.26 mol Cl2
D. 56.27 mol Cl2
E. 2250g Cl2
F. None of the above
Which of the following species should not be present in a base-catalyzed mechanism? (select all that apply)
Identify the products that are expected in the given reaction. (select all that apply)
Chapter H Solutions
Organic Chemistry
Ch. H - Prob. 1PPCh. H - Prob. 2PPCh. H - Prob. 3PPCh. H - Practice Problem H.4
In 1891 G. Merling...Ch. H - Practice Problem H.5
Many alkaloids appear to be...Ch. H - Practice Problem H.6 Papaverine has been...Ch. H - Practice Problem H.7
One of the important steps in...Ch. H - Prob. 8PPCh. H - Practice Problem H.9 Gramine has been synthesized...
Additional Science Textbook Solutions
Find more solutions based on key concepts
For each of the following, (i) give the systematic name of the compound and specify the oxidation state of the ...
General Chemistry: Atoms First
A circuit board with a dense distribution of integrated circuits (ICs) and dimensions of 120mm120mm on a side i...
Fundamentals of Heat and Mass Transfer
39. Consider the reaction:
Kp = 28.4 at 298 K
In a reaction mixture at equilibrium, the partial pressure...
Chemistry: Structure and Properties
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
a. Draw the resonance forms for SO2 (bonded OSO). b. Draw the resonance forms for ozone (bonded OOO). c. Sulfur...
Organic Chemistry (9th Edition)
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following reaction sequences will produce the product below? Select ALL that apply.arrow_forwardOchem prelab question I"ve attached part of the lab, but basically we are reducing m-nitroacetophenone to m-aminoacetophenone using granular tin. I've also attached one of my workups for the prelab, which require us to show the half reaction equation for reduction, the half reaction equation for oxidation and the overall balanced equation. Then determine the limiting reactant and theoretical yield Can you please check my work... especially my half reactions and see if those are correct (I'm not sure about the added e- and H+ Also, is the limiting reactant tin or should it be m-nitroacetophenonearrow_forwardQuestion b) calculate deltaH for the overall reaction using the following data for the indicated bond dissociation energies:arrow_forward
- Select all possible products of the following questionarrow_forwardWhat advantages and disadvantages of N-formylation? Why do we make it? please 1. abstract 2. introduction 3. methodology 4.result and discuss 5. conclusionarrow_forwardHello please explain step by step how to calculate theoretical and % yield of product isoquinoline 9. schiff based used = 4g, 12.73mmol Mol wt. (C17H19CIN2O4) = 350.79676g/molProduct isoqunioline (C17H19CIN2O4) obtained = 3.07garrow_forward
- Which of the following reactions would produce this product? Select ALL correct answers.arrow_forwardPractice problem 1: Show the correct major organic products. Thank you !arrow_forwardHello! quick question, when a chem problem starts with "refer to the unbalanced equation below" , and theres a unbalanced equation below (lol), do i have to first balance the equation first and then solve for problem? or am i supposed to just solve assuming its a 1 for the coefficent of each reactant and product? thanks in advance!arrow_forward
- Nitration experiment. Considering the structure of these hydrocarbons, will the following react with the nitrating mixture? If so, describe the result of the reaction. (a) Cyclohexane (b) Cyclohexene (c) Toluenearrow_forwardare the reactants correct to produce N-Ethyl-2-phenylethanamine and show mechanismarrow_forwardanswer 3,4,5,6,7 only. choices are: bromination, baeyer's reagent, ammoniacal AgNO3, ACETYLENE GASarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License