
Concept explainers
(a)
Interpretation:
The name of the following structure is to be determined by using the word amino.
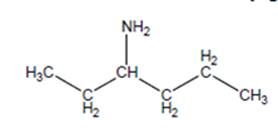
Concept Introduction:
The chemical name of the compound can be defined as the name of each elements which are present in chemical composition of the compound.
(b)
Interpretation:
The name of the following structure is to be determined by using the word amino.
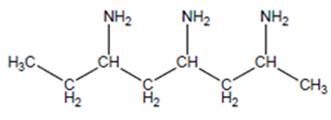
Concept Introduction:
The chemical name of the compound can be defined as the name of each elements which are present in chemical composition of the compound.
(c)
Interpretation:
The name of the following structure is to be determined by using the word amino.
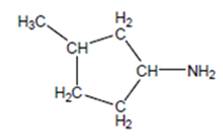
Concept Introduction:
The chemical name of the compound can be defined as the name of each elements which are present in chemical composition of the compound.
(d)
Interpretation:
The name of the following structure is to be determined by using the word amino.
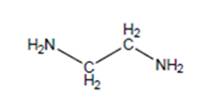
Concept Introduction:
The chemical name of the compound can be defined as the name of each elements which are present in chemical composition of the compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter NW2 Solutions
Organic Chemistry: A Guided Inquiry
- does methanol and methanol amine overlap each other are Scheme 1 Model A. Methanol: Model B. Methanol amine + red ball- no need of the H yellow red ball H- yellow ball blue ball NH2arrow_forwardAnswer the question below. Please go through each step in great detail.arrow_forwardAnswer the following problems and explain your answers for better understanding. ILLUSTRATE YOUR ANSWER CLEARLY.arrow_forward
- Which formula represents a heterocyclic amine? Match the correct answer to the letter below. SCH CH₂ ထ ၃ .. ဝဒ O b Od H d.arrow_forwardGive the following1. an amino acid that has an aryl side group chain 2. an amino acid that contains a side chain with a thioether bond3. an amino acid that reacts with HCL through acid-base reaction4. an amino acid that contains a sulfhydryl side chain5. an amino acid with a side chain that can form hydrogen bonds with water6. an amino acid that contains an amide side chainarrow_forwardThe answer is D (not C). The explanation given is "This compound is the only one of the choices that does not contain a base." What does it mean by "does not contain a base"? Isn't Br- a base?arrow_forward
- Organic Functional Groups Identifying positions labeled with Greek letters in acids and derivatives If possible, replace an H atom on the a carbon of the molecule in the drawing area with a methyl group substituent, and replace an H atom on the ß carbon with a hydroxyl group substituent. If one of the substituents can't be added for any reason, just don't add it. If neither substituent can be added, check the box under the drawing area. HO. Oneither substituent can be added. D X 1/5 0:0 Tyler V ola Ararrow_forwardOrganic Functional Groups Identifying positions labeled with Greek letters in acids and derivatives If possible, replace an H atom on the a carbon of the molecule in the drawing area with a methyl group substituent, and replace an H atom on the ß carbon with a hydroxyl group substituent. If one of the substituents can't be added for any reason, just don't add it. If neither substituent can be added, check the box under the drawing area. HO Oneither substituent can be added. OH 0/5 Xarrow_forwardIdentify the functional groups in the following molecules. (Use names from the table below. List each class of functional group only once. If there are fewer than 3 functional groups, leave an appropriate number of answer boxes empty.) HO CHS a) b) OH amine Carboxylic Acid Give detailed Solution with explanation needed, don't give Handwritten answer arene NH₂ phenylalaninearrow_forward
- 1. C is basic and is also known as sulfonamide group. * TRUE FALSE 2. A is a an amide and imparts water solubility to the molecule. * TRUE FALSE 3. D is an example of a heterocyclic aromatic ring. * TRUE FALSE 4. B is a little more hydrophilic than D. This is because B does not contain a heteroatom in its ring. * TRUE FALSE 5. B is an aromatic ring and imparts lipid solubility to the molecule. * TRUE FALSEarrow_forwardOne way of naming amines is to name in alphabetical order the alkyl groups attached to the nitrogen atom, using the prefixes di- and tri- if the groups are the same. An example is isopropylamine, whose formula is shown above. What are names for (a.), (b.), (c.), and (d.)? H NMe, (a) (b) (c) Me (d) Write bond-line formulas for: (e.) propylamine, (f.) trimethylamine, and (g.) ethylisopropylmethylamine Which amines in Problem 16 are (a.) primary amines, (b.) secondary amines, and (c) tertiary amines?arrow_forwarda. Classify each of the following amines as (1) primary, secondary or tertiary and as (2) aliphatic or aromatic. b. Classify each of the following amines as primary, secondary or tertiary.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
