
Concept explainers
(E) What does the number (+Z) at the center of each atom in Figure 1.1 represent, and whatnumber would you expect at the center of a representation of a bromine atom (Br)?
Interpretation:
The purpose of the representation of number (+Z) at the centre of each atom in given figure 1.1 should be determined along with the number which is present at the centre of a representation of bromine atom.
Concept Introduction:
In a planetary model of an atom, negative charged electrons are arranged around the positive charged electron in a series of shells which is like orbits.
The electrons present in the outermost energy level or shell is known as valence electrons. These electrons are available for bonding and outermost shell is known as valence shell.
Answer to Problem 1CTQ
+Z represents the nuclear charge of an atom which is equal to the number of protons or atomic number.
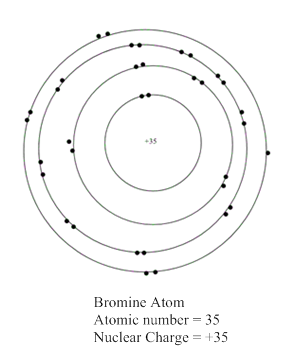
The representation of bromine atom:
Explanation of Solution
Given information:
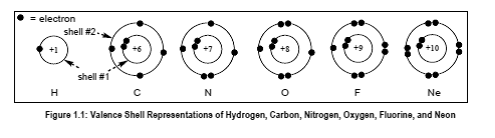
In the given figure, the number Z represents the atomic number of an atom such as atomic number of carbon is 6. +Z represents the nuclear charge which is nothing but atomic number.
The total charge present on all the protons in the nucleus is known as nuclear charge. The value of nuclear charge is equal to atomic number.
The number of protons present in nucleus of an atom is known as atomic number.
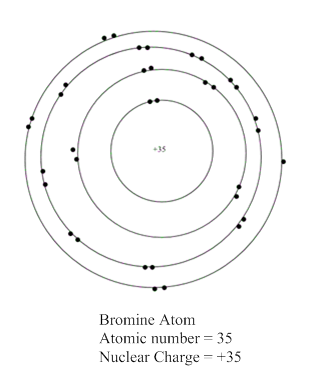
Now, atomic number of bromine atom is equal to 35 which is equal to the number of protons present in bromine atom. In the valence shell representation, +35 is present at the centre of a representation of bromine atom.
Hence, the representation of bromine atom is shown as:
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry: A Guided Inquiry
Additional Science Textbook Solutions
Chemistry: A Molecular Approach (4th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Chemistry In Context
Chemistry: The Central Science (14th Edition)
- #11 - I need help with only the second row. Row 1 and 3 are correect. The answer is not -11 for row 2. Please help.arrow_forwardMost shielded and least shielded protons?arrow_forwardIn the first picture, give the absolute configuration (Use D/L for left molecule and R/S for the right molecule. In the second picture, rank the groups (A,B,C,D) according to how they would be ranked when determining absolute configuration with R/S.arrow_forward
- Could someone show me how the following molecule can be cut into fragments for a mass spectrum and help me determine the m/z numbers? I don’t understand it, thank you. :)arrow_forwardhelp with my organic chemistry homework please. these 2 questions go together you have to answer the one with the NMR spectrum first to answer the second onearrow_forwardwhy is this S not R(I means the one that I circle in red color) 123 or 134???if go 123 it would be R,if it goes 134,it would be s?so confusing?Can anyone make it clear?Please help and explain in detailsarrow_forward
- How do you assign R or S to a molecule when the lowest-priority group is not oriented toward the back, on a dashed wedge?arrow_forwardIdentify the remaining three fragments from the table. (m/z=22,16,12) (hint: for m/z=22, the value of Z isn't necessarily 1)arrow_forward[E] *refer to the (2) photos below* (dont forget to put your answer on its corresponding table)arrow_forward
- Can someone explain what letter g is asking in relation to "k value", what exactly do they mean by the k value of letter f compared to the original value of k? I dont understand it at all, plz explainarrow_forwardSolve. Match a-e with 1-6arrow_forwardHi, please show the work for letter carrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

