Q: ive typed explanation not writte
A: IUPAC nomenclature is used for the systematic names of organic compounds. According to IUPAC…
Q: The most stable conformation of the compound shown has 1 CH3 mon H3C' 4 2 CH3
A: Given : structure of molecule Tip : groups at equatorial position are stable
Q: 5. In the lowest energy conformation of the compound below, how many alkyl substituents are axial…
A:
Q: Consider the following reaction at 298 K. C(graphite) + 2 H₂(g) → CH₂(g) AH' = -74.6 kJ Calculate…
A:
Q: The equilibrium constant, K, for the following reaction is 5.00×102 at 642 K. CoCl₂(g) CO(g) +…
A: The given equilibrium equation for the chemical reaction is COCl2(g) ⇔CO(g)+Cl2(g) K…
Q: How many micrograms of KMnO4 (Formula Weight: 158.04 g/mole) are contained in 50mL of 125mM KMnO4…
A: Given -> Formula weight of KMnO4= 158.04 g/mole Volume= 50 ml = 0.050 L (1L = 1000ml) Molarity=…
Q: A 73.0 kg patient being treated for leukemia is to receive an IV infusion of 5 cyclophosphamide over…
A: Given that, a 73 kg patient being treated for leukemia is to receive an IV infusion of 5 mg/kg…
Q: Identify the number of significant figures in each of the following a) 0.05040 m 6) 1400 ml. 2 c)…
A: ♧ Rules to find significant figures :1. All the non-zero digits 1 to 9 are significant .2. Zeros…
Q: 8. Identify the species that results from the movement of the following elelctron pairs. H N:
A: Answer:- This question is answered by using the simple concept of writing the resonating structure…
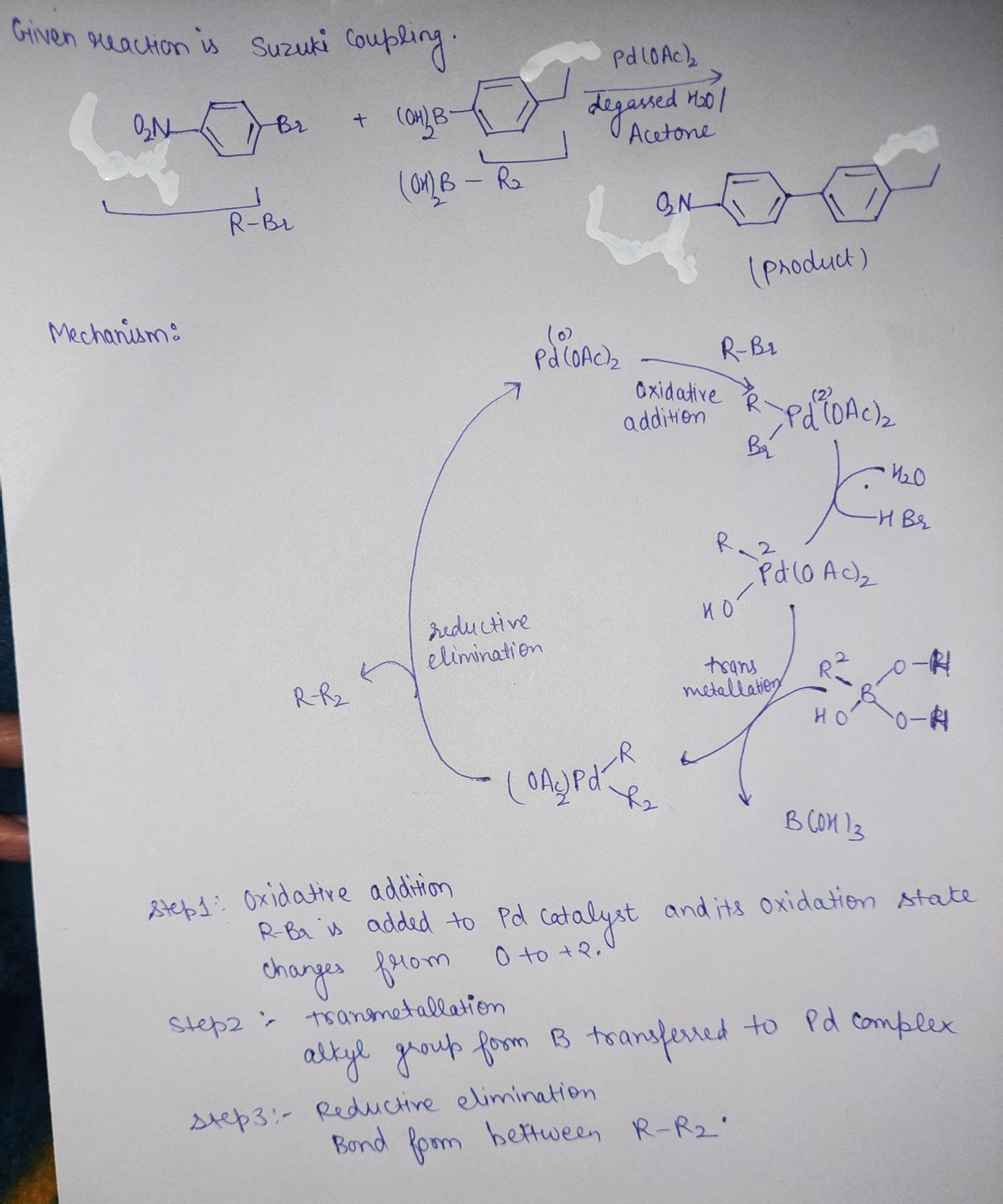
Q: Draw the products of the two step reaction sequence shown below. Ignore inorganic byproducts. HO…
A: The attack of PBr3 on alcohol follows SN2 reaction.
Q: The following chemical reaction is used to synthesize a flavouring agent that has an aroma similar…
A: Esterification Reaction: When primary alcohol is treated with a carboxylic acid in the presence of…
Q: The decomposition of ammonia, NH3, is zero order. The rate constant for this reaction at a…
A: The given data is as follows: Rate constant for zero order reaction (k) = 5.32×10-3 molL-1s-1…
Q: Which molecule is labelled with the correct R and S descriptors? Br., R Br... S в кон Br., R ОА OB…
A: The 'R' and 'S' configuration of the chiral carbon can be assigned by using the following steps ;…
Q: Choose the resonance structure that results from the arrow pushing scheme below: H سفی مشهد مع 00 O-…
A: We have to determine the resonance structure of the given molecule
Q: Which is an example of a chemical change? (Hint: Review the common signs of chemical reaction.) Salt…
A: A chemical change occurs when any substance combines with another to form a totally different…
Q: What is the molar mass (in g mol-1) of a gas which has a density of 0.88 g/L at a pressure of 0.84…
A: Given P = 0.84 bar T = 22.6°C Density = 0.88 g/L
Q: A chemical engineer is studying the two reactions shown in the table below. In each case, she fills…
A: Given that, Two different chemical reaction at temperature 69.0oc(342K). We have to calculate ΔG of…
Q: Hydrogen gas reacts with oxygen gas to produce water. When this reaction is written out and…
A: given reaction hydrogen gas ( H2 ) react with Oxygen ( O2 ) gas to produce water ( H2O )
Q: HO 17.8 Give the product. LOH Na₂Cr₂O7
A: we have to determine the product of the given reaction
Q: Can you help me with the explanation of the binary covalent (molecular) compound, ionic compound and…
A: Introduction Chemical bonding is the process of forming bonds between two atoms or molecules in…
Q: Convert the amount of spiperone in each sample to the concentration in the 1 ml total volume of…
A: Explanation The molar concentration is expressed as Molar concentration (M) = mole / Volume of…
Q: The following table contains some data showing the effect of molar concentration of a solution on…
A: Given: The data for concentration and optical density is: Concentration (M) Optical density…
Q: How many milligrams of solid MgCl2 (Formula Weight: 95.211 g/mole) are required to prepare 250mL of…
A: Given: Molarity of solution = 150 mM = 150 x 10-3 M = 0.150 M = 0.150 mol L-1 Volume of solution =…
Q: Consider the following reaction: 2HF(g) ⟷H2(g) + F2(g) (K = 1.00 x10-2) Given 1.00 mole…
A: The equilibrium reaction is as follows: 2HF(g) ⟷H2(g) + F2(g) (K = 1.00 x10-2) The initial…
Q: How many milligrams of solid MgCl2 (Formula Weight: 95.211 g/mole) are required to prepare 250mL of…
A: Given : formula weight = 95.211 g/mol Volume = 250 mL Molarity = 150 mM
Q: 3. Write the name for each of the following covalent compounds: (a) CBR4 (b) N₂O3 (c) NF3 (e) P₂Ss…
A: Note: As per the guidelines, solution of 3rd questions has been made. For the expert solution of 4th…
Q: What must you must pay attention to if you are using sodium bicarbonate (NaHCO3) as the basic wash?…
A: Sodium bicarbonate, also known as baking soda, is a white crystalline powder that is commonly used…
Q: The Ksp of magnesium hydroxide, Mg(OH)2, is 5.61 × 10-¹2. Calculate the solubility of this compound…
A: The Ksp expression for a salt is the product of the concentrations of the ions, with each…
Q: The Haber process is used to synthesize ammonia (NH3) on a large scale by the reaction of nitrogen…
A:
Q: A B. Write Br I ypac Names NH₂ CI Br L
A: The naming of molecules in IUPAC nomenclature can be done first by identifying the longest…
Q: Which is a product of this reaction according to the arrows? All lone pairs and formal charges are…
A: Nucleophilic substitution is a type of chemical reaction in which a nucleophile, a molecule with a…
Q: 21 Which statement is correct concerning the relative stabilities of the two conformations, A and B,…
A: Group present at axial position in cyclohexane chair conformation decrease the stability due to…
Q: Illustrate all possible stereoisomer geometries for the coordination compound, K[Cr(C₂O4)2(H₂O)2].
A: Introduction to coordination compound A coordination compound, also known as a coordination…
Q: What concentration of barium hydroxide is needed to give an aqueous solution with a pH of 12.430?…
A: Answer:- This question is answered by using the simple concept of calculation of molarity using the…
Q: Nitrogen gas (N2) reacts with hydrogen gas (H₂) to form ammonia (NH3). At 200°C in a closed…
A:
Q: A pure sample of Substance S is put into an evacuated flask. The flask is then heated steadily and…
A:
Q: Rank the elements or compounds in the table below in decreasing order of their boiling points. That…
A: Introduction At the boiling point, the vapor pressure of the liquid is equal to the atmospheric…
Q: Give the complete electronic configuration for Mn²+ and V4+.
A: Electronic configuration refers to the arrangement of electrons in an atom or ion. It is a way of…
Q: At 1 atm, how much energy is required to heat 55.0 g H₂O(s) at -16.0 °C to H₂O(g) at 161.0 °C? Use…
A: Answer: Converting ice from -160C to steam at 1610C is 5 step process: 1. Heating of ice 2. melting…
Q: Standard enthalpy of formation of N(g) is 472 kJ/mol. From this information, estimate the bond…
A: Introduction Bond enthalpy is an important physical quantity used to describe the strength of a…
Q: Perform a confirmational analysis on 2-bromo-3-methylbutane.
A: Given 2-bromo-3-methylbutane
Q: H H
A: Electrons move from electron rich center towards electron deficient center. Here arrow notation is…
Q: Can you help me with the explanation of the noncovalent interaction, hydrogen bonding, dipole-dipole…
A:
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Pb(s) + O₂(g) → 2PbO(s)…
A: Recall the given reactions, 1 2 Pb s + O2 g → 2 PbO s…
Q: The decomposition reaction is a second order reaction. Initial concentration of MH is 0.135 M, and…
A: The given data is as follows: The initial concentration of reactant A° = 0.135 M The rate…
Q: Consider the fictional element A (which has a molar mass of 7.00 g/mol) and the fictional element D…
A: Given :- Molar mass of A = 7.00 g/mol. Molar mass of D = 13.00 g/mol. Mass of hydrate compound of…
Q: Name each of following compounds:
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Beryllium-10 changes to boron-10 . write a balanced nuclear equation and identify the radiation…
A: Given -> Beryllium-10 ----> Boron-10
Q: In the laboratory, a general chemistry student measured the pH of a 0.392 M aqueous solution of…
A:
Q: Convert the following from mg/L as the ion or compound to mg/L as CaCO3 a. 265.00 mg/L HCl b.…
A:

Step by step
Solved in 2 steps with 1 images

- When 1.40 moles of Br2 are put in a 0.960−L flask, 1.10 percent of the Br2 undergoes dissociation. Calculate the equilibrium constant Kc for the reaction.19.Which explanation below best describes why the following reaction occurs? 3Zn + H33PO44 --> 3H22 + Zn33(PO44)2 A.H is higher on the activity series than Zn B.Zn3(PO4)2 is insoluble C.Zn has higher energy than H. D.PO4 has higher energy than H2What is the missing product from this reaction? 244/96Cm -> + 4/2HE
- Which of the following steps performed in the Chemical Changes experiment is INCORRECTLY matched to the observed change? Select one: a. Formation of CuSO4: formation of blue solution b. Addition of H2SO4 to CuO: dissolution of black solid c. Addition of Zn dust: blue solution turns colorless d. Formation of Cu(OH)2: formation of black precipitateWhich compound is most likely to undergo oxidative addition of H2? a. RhCl(PPh3)3 b. Fe(CO)5 c. [RhI4(CO)2]− d. [HFe(CO)4]−Which of the reagents would oxidize Zn to Zn2+, but not Pb to Pb2+? Ca2+ Co Co2+ Br2 Br− Ca
- Can Ag+ , Pb2+ and Hg22+ be separated if ammonia is first added to an aqueous mixture of these three cations instead of hydrochloric acid? If so, how?Which reaction requires a metal catalyst? Explain. A. hydrohalogenation B. halogenation C. dehydration D. hydrogenationWhat is the oxidizing agent (oxidant) when acidified potassium permanganate reacts with hydrogen peroxide KMnO4 + H2O2

