Bial's test uses concentrated HCI as a dehydrating acid and orcinol + traces of ferric chloride as condensation reagent. The test reagent dehydrates pentoses to form furfural. Furfural further reacts with orcinol and the iron ion present in the test reagent to produce a O bluish or green product red precipitate yellow solution O light pink or pink solution O Other:
Q: Based on the information given, what separation technique(s) is/are most likely feasible? Please…
A: The selective boiling and subsequent condensation of a part in a liquid mixture is referred to as…
Q: The reaction in Fohl’s test will cause the formation of what compound? What type of reaction happens…
A: Fohl's test is the biochemical test of proteins for the detection of presence of Aminoacids with…
Q: A 100 mL solution of 0.1 M amino acid (AA) at pH 1.0 was titrated with 1.0 M NaOH solution. The pH…
A: Amino acids are composed of a central chiral carbon, hydrogen, an alpha-carboxylic group, an…
Q: You want to set up a 1:10 dilution series, so that you can plate a 10-1, 10-2, and a 10-3dilution;…
A: It is basically done for counting the microorganisms like viruses, bacteria etc from an unknown…
Q: The table below summarizes the results for Millon's test. Provide the correct remarks from the…
A: Millon’s test is an analytical test used for the detection of the hydroxy phenol groups. Since the…
Q: 11. ● Possible methods for the quantitative determination of sodium bicarbonate for injection are:…
A: We'll answer the first question since the exact one wasn't specified. Please submit a new question…
Q: What is the purpose of doing the silver nitrate test? Write the chemical equation involved here
A: Introduction: Silver nitrate is an inorganic compound and its formula is AgNO3 with a molar mass…
Q: A carbohydrate sample reacts positively with all test reagents except for Lugol's. The sample could…
A: The sample could be possibly Sucrose.
Q: Provide a chemical test/reagent that would differentiate each of the following pairs of compounds.…
A: Asked : Reagent and indication of positive test
Q: Results: Tub Ranking of reaction Iodine Test Benedict's Test e rate 1 2 3 4
A: The most important thing to remember is that chemical reactions rely on reactant particles colliding…
Q: Explain the chemical basis (reaction) of Millon’s test. (Generic reaction and explanation) What is…
A: Proteins are made up of amino acids joined together by peptide bond. Proteins can be estimated by…
Q: Which of the following is the test for Methionine A. Biuret Test B. Ninhydrin Test C.…
A: since you've posted multiple questions, only the first one will be answered. before addressing the…
Q: Provide a schematic diagram for the Iodine test methodologies in determining the presence of…
A: Biomolecules are formed inside living organisms. They can be simple or complex depending on the…
Q: The BSA stock solution from the previous problem was then diluted to generate a set of standard…
A: CONCENTRATION OF BSA (mg/ml) ABSORBANCE 0 0 0.1 0.048 0.2 0.095 0.4 0.191 0.6 0.290 0.8…
Q: Provide the Principles and detailed procedures of the following tests for the "Color Reaction of…
A: Hopkins - cole test Principle -In this test , concentrated H2So4 cause dehydration of tryptophan…
Q: Give and explain the principles of the following tests for lipids: Spot Test Acrolein Test Test for…
A: Your question has multiple subparts. I will answer 1st three of the subparts, as per the guidelines.…
Q: Which of the following below will show a NEGATIVE result in Huble’s test? Group of answer choices…
A: Lipids are carboxylic groups is attached to a long hydrocarbon chain. On the basis of the presence…
Q: What are the possible interferences or complications in detecting and differentiating aldehydes and…
A: Before scientists developed tools such as x-ray crystallography and other spectrophotometric…
Q: Discuss the properties of potassium iodide solution, and how it results in the detection of starch.…
A: Starch is a complex polysaccharide that is made of glucose units joined together by beta glycosidic…
Q: What are the chemical reaction of the following Blood Additives/Anticoagulants? 1. None 2. Clot…
A: medicines that helps to prevent blood clot are known as anticoagulants they are also known as blood…
Q: A vial containing 400 mg hydrocortisone sodium succinate powder for injection is to be reconstituted…
A: GIVEN: Amount of hydrocortisone sodium succinate powder for injection = 400 mg Volume of injection =…
Q: Give at least two (2) chemical tests for amino acids/ proteins other than what were performed in…
A: There are several other tests to determine properties of proteins and amino acids present in a…
Q: REAGENTS. Match the name of the reagent with the physiological test. Tests may require more than one…
A: A reagent is a substance added to a system to cause a chemical reaction or test if a reaction…
Q: Which of the following tests is used to distinguish glucose from fructose?* A. Molisch test B.…
A: Carbohydrates are polyhydroxy aldehydes or polyhydroxy ketones, or compounds that can be hydrolyzed…
Q: explain what the results of this experiment are and the purpose Table 2 Absorbance vs CoCl2…
A: The Beer-Lambert Rule: The concentration of the absorbing species and the route length of light…
Q: Ninhydrin test for amino acids or protein requires that sample test solution should be at initially…
A: Proteins are one of the essential macronutrients for a human body. The building blocks of proteins…
Q: What is the clinical significance of the following tests? Discuss what constituent is being detected…
A: Heller's test is a clinical test that was discovered by Austrian chemist Johann Florian Heller. It…
Q: Which Limit test is not found in the USP Calcium Gluconate drug substance monograph? A. Phosphate…
A: Calcium Gluconate is the anhydrous or if hydrous it only contains one molecule of water. Calcium…
Q: Create an illustrated step-by-step schematic diagram of the following Qualitative test for Lipids:…
A: Lipids are substances that are insoluble in water and soluble in organic solvents like ether and…
Q: Finally, draw the standard curve for BSA. Reagents Blank Tube Tube Tube 2 3 Tube Tube Tube Tube 5 4…
A: BSA is the bovine serum albumin that is a protein concentration used as a reference in the protein…
Q: Section 1: Flame test for Na*, K* and Ca* ions. Your conclusion for each test will be that the metal…
A: The presence of carbonate in a compound can be tested using a dilute acid. Upon addition of a…
Q: Give at least two (2) chemical tests for amino acids/ proteins other than what were performed in…
A: In biochemistry , a chemical test is performed in qualitative or quantitative procedure to identify…
Q: 1.000-g sample containing bromide was dissolved in sufficient water to give 100.0 mL. A 50.00 mL…
A: Asked : Cell notation and Nernst equation
Q: What test reagent is most sensitive in detecting sugar in urine? What are the possible positive…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: need to dissolve cholesterol in Chloroform in the Liebermann-Burchard Test
A: The reaction of acetic anhydride in chloroform with unsaturated steroids (e.g. cholesterol) in the…
Q: Which of the following tests gives bluish green product due to the presence of carbohydrates?*…
A: Carbohydrates are polyhydroxy aldehydes or polyhydroxy ketones. Monomers of carbohydrates i.e.,…
Q: Explain the color changes observed throughout the peroxide value of oil experiment. Materials: 1.…
A: The unsaturated fat when undergoing oxidation forms peroxide so the peroxide value denotes the…
Q: You are asked to prepare Murashige & Skoog (MS) medium according to concentration shown the Table.…
A: We are having the concentration of the ammonium nitrate and potassium nitrate at 1 litre or 1000 ml…
Q: Laemmli buffer, also referred to as SDS-reducing buffer, is added to each of the samples. It is a…
A: Buffer is defined as any solution that resists small change in its pH. Laemmli buffer is formulated…
Q: Answer the following questions (not more than 5 sentences/question). Discuss the correct…
A: Qualitative analysis is the process of identification and detection of elements or groups of…
Q: Is this an drawing of the chemical reaction involved in Lugol’s test?
A: Carbohydrates are molecules that include carbon, hydrogen, and oxygen and have the formula (CH2O)n.…
Q: This test gives red color, indicating the presence of cysteine.* A. Lead acetate test B.…
A: Cystein is a sulphur containing amino acid that is capable of forming disulphide bridges in the…
Q: Make the following prescription isotonic with lacrimal fluid: 0.06 g Ephedrine sulfate (E = 0.23)…
A: Asked : Volume of sterilized distilled water (in milliliters) that should be added to the…
Q: Calculate the amount of each solution needed to make 750 mL if a dextrose 25% solution given only a…
A: Given- We make 25% dextrose of 750ml by using 60% and 5% dextrose. Use alligation method for such…
Q: Check which test (s) will yield a positive result or negative result for the given compounds.…
A: The different protein tests carried out in this experiment are: Biuret test is for peptide bonds in…
Q: The table below summarizes the results for Hopkins-Cole test. Provide the correct remarks from the…
A: The amino acid tryptophan has a side chain containing an indole ring.
Q: A positive result for the Ninhydrin test yields a deep blue or violet-blue color of the soluti more…
A: Ninhydrin test is a chemical test performed to detect the presence of amines or amino acids.
Q: Lead nitrate Stock Solution— Dissolve 159.8 mg of lead nitrate in 100 mL of water to which has been…
A:
Q: Give the ideal observation for the positive results of the following samples to complete the table…
A: Proteins or peptides are composed of twenty standard amino acids attached via peptide bonds. These…
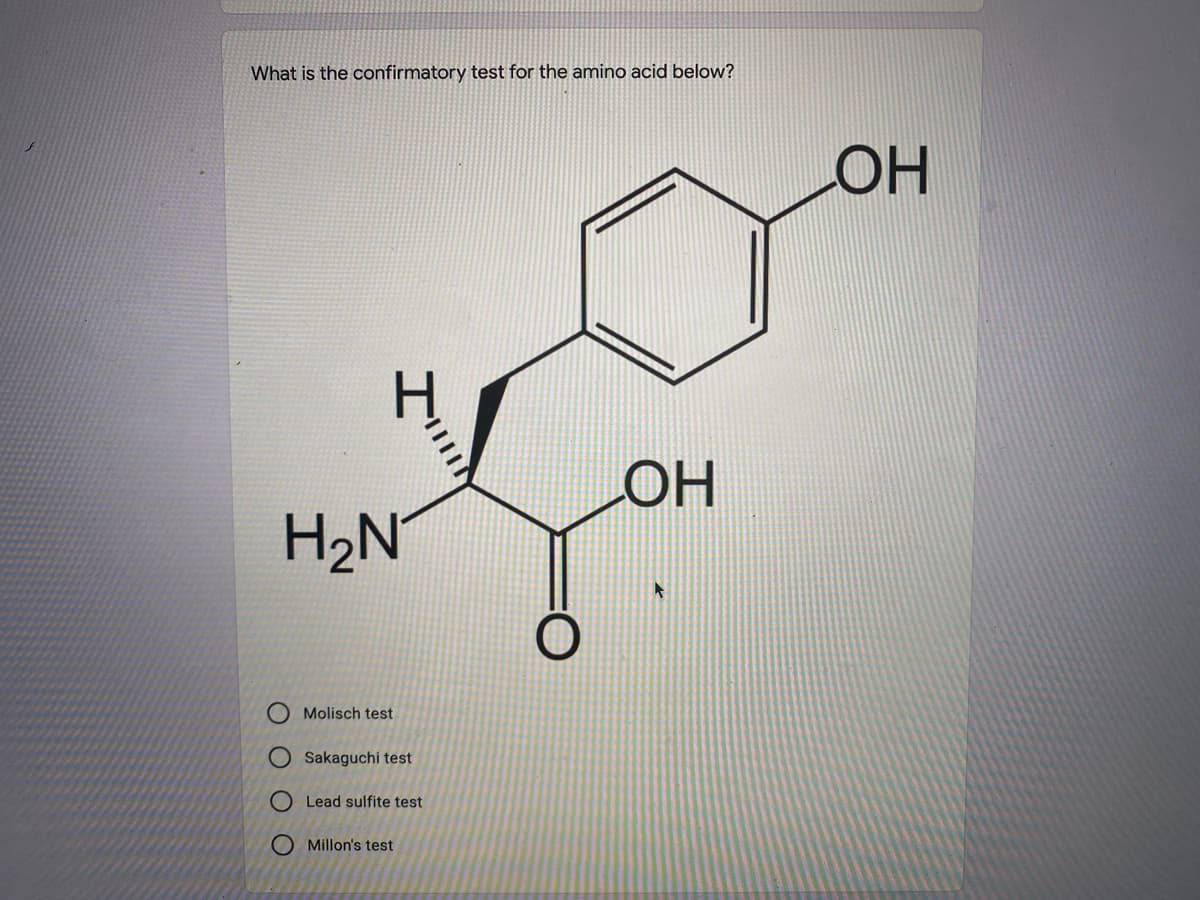
Q: The above molecule was subjected to acid hydrolysis then was analyzed thru qualitative tests.…
A:
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- Laemmli buffer, also referred to as SDS-reducing buffer, is added to each of the samples. It is a blue solution and the recipe for 2X Laemmli buffer is: 0.125 M Tris HCl, pH 6.8, 20% glycerol, 4% SDS and 10% β-mercaptoethanol (βME). What is the purpose of each of these ingredients?Your supervisor asks to make 350 mL of a 0.75% (w/v) starch solution using a 5% (w/v) stock solution. You have calcuated how much stock solution you need. But, how much water (in mL) will you need to add to the stock to achieve the correct final concentration?The above molecule was subjected to acid hydrolysis then was analyzed thru qualitative tests. Indicate whether the given molecule will give a positive (+) or negative (-) result in each given test. Identify whether (+/-) Test for phosphate: Dische Test: Bials Test: Murexide test: Wheeler-Johnson Test: 6. What is the transitory temperature?: Higher or lower?
- You are supplied with the following: / Jy word voorsien van die volgende: NaCl (Mr= 58,443 g /mol) 2.5M Tris-Cl, pH 8 solution /oplossing (1 Litre) EDTA (Mr= 380,2g/mol) 10% sodium dodecyl sulphate solution / natriumdodecyl sulfaat oplossing Proteïnase K solution / oplossing (50 mg dissolved / opgelos in 1 ml ddH2O) You need a digestion buffer consisting of the following: / Jy moet 'n verteringsbuffer op maak wat uit die volgende bestaan: 15m M NaCl 75 mM Tris-Cl,pH 8 16 mM EDTA,pH 8 0.8% sodium sulphate / natrium dodecyl sulfaat 0,75 mg/ml proteïnase K How will you prepare 500 ml of the digestion buffer? Show all your steps and calculations. Remember to explain exactly how you will make it up. / Hoe sal jy 500 ml van die verteringsbuffer voorberei? Wys al jou stappe en berekeninge. Onthou om te verduidelik presies hoe jy dit sal op maak.Determine the weight/volume of the chemicals needed to prepare the following solutions: a) 100 ml of 0.9% (w/v) saline (NaCl) b) 30 ml of 50% glycerol (v/v) c) Electrophoresis requires TAE, which is a specific mixture of Tris base, acetic acid, and EDTA. TAE is normally made as a 50X concentrated stock. Provide a recipe to make 40 ml of 50X TAE. The recipe for one liter of 50X TAE is as follows: 242g Tris base, 57.1 ml glacial acetic acid, 100 ml 0.5 M EDTAAn aqueous antifreeze solution is 45.0% ethylene glycol () by mass. The density of the solution is 1.05. Calculate the molality, molarity, and mole fraction of the ethylene glycol
- Complete the table below regarding the different laboratory tests done for Amino Acids and Proteins Tests Procedure (Chemical Reagents Added) Positive Results/ Observations (ex: color of solution, precipitate formation, etc) Positive for (provide details like functional group responsible) Molisch Test Benedict’s Test Barfoed’s Test Moore’s Test Bial’s Test Seliwanoff’s Test Osazone Test Fehling’s Test Tollen’s Test Mucic Acid Test Lugol’s Test/ Iodine Test Hydrolysis w/ Benedict’s Test Hydrolysis w/ Iodine Test Provide the expected results with the samples given below: For negative results just leave it blank, for positive results write “(+) then the expected observation” ex: + blue solution Samples Molisch Test Benedict’s Test Barfoed’s Test Moore’s Test Bial’s Test Seliwanoff’s Test Glucose Galactose Fructose Ribose…. A prescriber ordered 240 mL of ¾ strength Sustacal PO B.I.D. Explain how you would use water as a diluent to mix this solution. Show your work with units and describe how much water and how much Sustacal you would use to create this mixture..Solution Absorbance mg/ml aspirin Standard solution - 1.6 mg/mL A 0.638 0.08 mg/mL B 0.504 0.064 mg/mL C 0.376 0.048 mg/mL D 0.259 0.032 mg/mL E 0.126 0.016 mg/mL A = -log T where T = %T ÷ 100 Construct a callibration curve using the above data. Absorbance should be on the vertical axis and "mg/mL of acetylsalicylic acid" on the horizontal axis. The line should go through the origin. Using the data provided, the graph you have generated, and the procedure that was used to generate the solutions which were examined by spectroscopy, calculate the amount of acetylsalicylic acid per tablet. Commercial tablet 1 labelled as 100 mg enteric coated Absorbance = 0.16 Commercial tablet 2 labelled as 300 mg Absorbance = 0.45 Student prepared tablet from practical 5 Absorbance = 0.19 Using the data provided, the graph you have generated, and the procedure that was used…
- Qualitative and Quantitative Tests for Amino Acids and ProteinsComplete the table: Test Reagents Positive Result Positive Indication Ninhydrin test Xanthoproteic acid test Millon's test Hopkins Cole test Pauly's diazo Test Lead sulphide test Histidine test Isatin test Folin’s McCarthy Sullivan Test Nitroprusside TestLipids from an organic sample are extracted separately by column chromatography using the ff eluants: 1st eluant - chloroform: methanol: water (6:3:1) 2nd eluant - methanol: chloroform (9:1) 3rd eluant – petroleum ether: ethyl ether (1:1) Test results for each eluate are tabulated below. Identify the possible lipid present in each eluate. Test Reagents 1. KHSO4 2. HNO3,(NH4)3MoO4 3. CHCl3, acetic anhydride, conc. H2SO4 4. -naphthol, conc. H2SO4 5. Hydroxylamine HCl, FeCl3 6. ninhydrin reagent POSSIBLE LIPIDS: Triacylglycerol, cholesterol, sphingomyelin, lecithin20 mL solution is 2 M NaOH. If 0.5 M HCl is to be added to this solution, calculate the pH of the resulting solution: A. when 10 mL of the 0.5 M HCl is added to the original solution of the base (initial pH). B. When 50 mL H20 is added to the original solution of the base.



