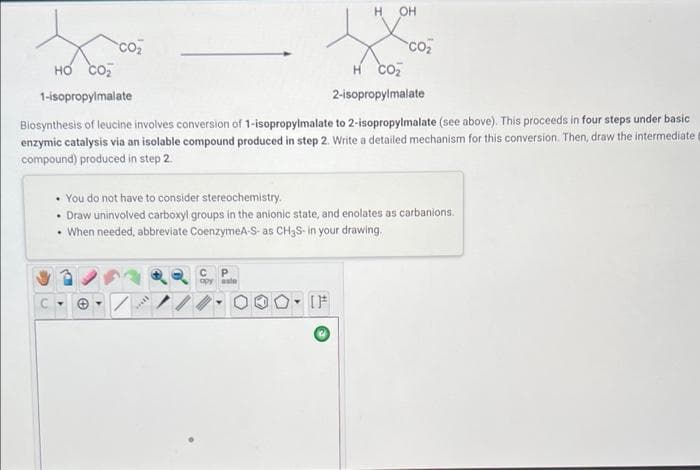
Biosynthesis of leucine involves conversion of 1-isopropylmalate to 2-isopropylmalate (see above). This proceeds in four steps under basic enzymic catalysis via an isolable compound produced in step 2. Write a detailed mechanism for this conversion. Then, draw the intermediate compound) produced in step 2.
Q: Given the active site diagram below, identify the numbered region corresponding to an acidic catalyt...
A: An acid is a molecule that can accept a proton from the solution and a base is a molecule that can d...
Q: Describe glucose oxidase's substrate, the substrate’s biological relevance, and identify the main in...
A: Glucose oxidase is an oxireductases class of enzyme which catalyzes the oxidation of glucose to hydr...
Q: 1. Draw the general structure of glycerophospholipids and designate the site for variation in head g...
A: Lipids are non-polar hydrocarbons, like fatty acids (FA), waxes, sterols, fat soluble vitamins (A, D...
Q: One amino acid loses an oxygen and hydrogen atom from its and the other amino acid loses a a-carbon ...
A: Any of a group of organic compounds composed of a basic amino group, an acidic carboxyl group, and a...
Q: Think of and identify a modern-day problem that can be solved by Genetic Engineering. If you were a ...
A: Genetic engineering is a branch of science which involves recombinant DNA technology to introduce fo...
Q: What is meant by reciprocal regulation ? Name one compound that reciprocally regulates glycolysis an...
A:
Q: ZHN. HN- HO HO HN, NH OH HO + H20 HO но NH2 lyase oxidoreductase hydrolase ligase isomerase transfer...
A: Enzymes and their classification: Enzymes are biological substances that catalyze biochemical reacti...
Q: Consider a glucose molecule that begins undergoing glycolysis. If the cell needs to make nucleotides...
A: Glycolysis is a catabolic pathway in which glucose is oxidized to two molecules of pyruvate. Glycoly...
Q: Can sucrose act as a reducing sugar? Explain the answer
A: Carbohydrates are composed of carbon, hydrogen, and oxygen in the ratio of 1:2:1. Carbohydrates act ...
Q: 1,3-bisphosphoglycerate is used to produce ATP. Which of the two phosphates of 1,3- bisphosphoglycer...
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabol...
Q: The common amino acid residues in B-turis are proline and lysine
A: Proteins are the polymers of amino acids. There are four levels of structural organizations for the ...
Q: draw an isomer of the triglyceride in sunflower seed oil. Show the reaction and write out the produc...
A: Hydrogenation is the process of chemical modification that is performed by addition of hydrogen to (...
Q: In Gel filtration chromatography, when will you stop collecting eluents if sample is not colored?
A: Gel-filtration chromatography is a type of partition chromatography that is used to separate molecul...
Q: Determine the catalytic power of jack bean urease in catalyzing the hydrolysis of urea given the fol...
A: ENZYMES : - Enzymes are defined as the organic molecules which helps in catalyzing the reaction....
Q: 3. Trace the course of the radioactive label in 2-[14C] glucose through glycolysis and the citric ac...
A: Glycolysis, also known as the Embden-Meyerhof-Parnas (EMP) pathway, occurs in the cytoplasm of all l...
Q: Which of the following is associated with pain relief? a. acetylcholine c. serotonin b. glutamate d....
A: Introduction: The term pain is an unpleasant sensory and emotional experience associated with actual...
Q: Consider the Ramachandran plot to answer both questions. Which observation most likely describes par...
A: 1) The observation of α helix can be most likely observed at: φ = –140° ; ψ = +130° ; many Val resid...
Q: What can be the interpretation for the results given below? Test/s Results Ninhydrin Xanthroproteic ...
A: Qualitative tests for amino acids help to detect the presence of specific amino acids and proteins i...
Q: 14. Subject this amino acid sequence under (1) trypsin (2) chymotrypsin (3) pepsin and (4) CNBR. Gen...
A: Trypsin, chymotrypsin, and pepsin are proteolytic enzymes, which help to cleave complex proteins int...
Q: Which of the following could be the DNA template for the following protein primary structum Methioni...
A: In order to form a polypeptide chain, amino acids are linked together in a sequence. During protein ...
Q: A small generic section of the primary structure of an a helix is given by -amino acid, -amino acid,...
A: Alpha-helix is an example of a secondary structure. Alpha-helix is formed by the hydrogen bonding be...
Q: What would be the best buffering agent to choose if you wanted to buffer an enzyme reaction or tissu...
A: pH is the measure of the strength of H+ ion or Hydronium ions in solution. pOH is the measure o...
Q: Using the provided data on the activity part, what is the acquired R value? Is it acceptable or not?...
A: Absorbance is the unit measure of amount of light that passes through a particular solution at a giv...
Q: Which would be an overall explanation for how a buffer works? O The conjugate base ions in the buffe...
A: A buffer resists acidic or basic components changing pH. Buffer solutions, which keep a pH, provide ...
Q: Identify the two buffer systems in the tubular fluid that carry excess hydrogen ions into the urine ...
A: The nephrons are microscopic structures and are the structural unit of the kidney that are constitut...
Q: explain how calvin cycle eznymes are indirectly activated by light
A: Calvin cycle occur at day time because it depends upon light reaction. The Calvin cycle refers to th...
Q: Below are several DNA sequences that are mutated compared with the wild-type sequence. Each is a sec...
A: Note - Hi ! Thank you for the question. We are authorized to answer three subparts at a time. Since ...
Q: 1 Active Site Reaction Mechanism Inhibitor HN- 2•Zn NH2 HO. HO, NH2 N N- NH `NH NH NH, N' 'N N 'N NH...
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer the ...
Q: Which statement below best describes the main difference between how scientists define data and hypo...
A: Scientific Hypothesis is a part of scientific method which consists of 6 different steps. Scientific...
Q: 3. Differentiate Enzyme Inhibition by filling the table below: Competitive Non-Competitive Uncompeti...
A: Enzyme inhibition is a process by which the activity of an enzyme is altered. Inhibitors are substan...
Q: Assume that a certain cell, the ratio products/reactants or Keq 560.6 (Keq dimensionless) reaction G...
A: A reversible reaction is one that can go both forward and backward. When the rate of the forward rea...
Q: The following reactions were catalyzed by an enzyme that follows the Michaelis-Menten mechanism in t...
A: The given enzyme kinetics is said to follow Michaelis-Menten mechanism. Here Substrate concentration...
Q: Assume that mitochondria contain 0.35 Molar KCL and 0.012 Molar NaCl. Calculate the grams per liter,...
A: Osmolarity is defined as the concentration of a solution expressed as the total number of solute par...
Q: 47. Protein Sequencing Applies changes in pH to separate amino acids Applies changes in pH to sepa...
A: "Since you have posted a multiple question, we will answer the first question for you. If you want a...
Q: 3. The rates of a carbonic anhydrase catalyzed reaction of CO2 and H2O, in the absence and presence ...
A: Carbonic anhydrases catalyze the interconversion between CO2 and H2O, maintain acid-base homeostasis...
Q: Why Sexual is the most important type of variation in human?
A: Sexual variation causes genetic diversity in humans . Genetic diversity is important because it give...
Q: . CTGATTCCG AATGS Given a part of DNA undergoing replication. Copy and write the corresponding bases...
A: DNA replication is a process by which each strand of DNA acts as a template and copies itself to pro...
Q: (a) if it is found that the number doubles in 6 hours, how many may be expected at the end of 18 hou...
A: Bacteria are microscopic organisms not visible with the naked eye. These are omnipresent (they prese...
Q: Can the iodine test distinguish between amylose and amylopectin? What would you look for?
A: Carbohydrates are an essential source of energy. It is found in almost all foods and is largely used...
Q: Explain what is meant by the term, “high energy compound”. Name a thioester molecule that is common...
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: Select the word from the dropdown menu that correctly completes each of the following statements.
A: Citric acid cycle is a cyclic metabolic chemical reactions in which energy (NADH, FADH, GTP/ATP) is ...
Q: Which has better emulsifying properties, soap or detergent. Explain your answer using the concept of...
A: Emulsifiers are those substances that decrease the surface tension between two phases which are comp...
Q: the different individuals involved in a clinical laboratory (their job specification -qualification,...
A: JOB SPECIFICATION Laboratory Director QUALIFICATION and TRAINING Doctoral degree (e.g., MD or ...
Q: The only sugar structure that does NOT contain chiral carbon atom. Erythrose Erythroluse Glyceraldeh...
A: Carbohydrates are composed of carbon, oxygen, and hydrogen which are connected by the glycosi...
Q: Malonyl-ACP is a precursor in fatty acid synthesis in the acyl carrier protein pathway. Draw the str...
A: Fatty acid synthesis is the formation of fatty acids from acetyl co A and NADH using fatty acid synt...
Q: Modified true or false. Write the correct answer if the underlined word is false. 1. A prosthetic g...
A: Proteins' principal role is to act as enzymes—catalysts that help cells speed up nearly all chemical...
Q: MRI What does the "induced free decay" curve describe?
A: The term "free induction decay" refers to a short-lived sinusoidal electromagnetic signal that devel...
Q: 23. In the concerted model, the most active enzyme form will be when a. All subunits are in the R st...
A: Enzyme can have it's subunits in 2 conformations : Low-activity state -(Tense)T state High-activity...
Q: _____________ is a tranquilizer that is also known as the “date rape” drug. a. Halcion b. Librium c....
A: Tranquilizer is a drug which is used to reduce anxiety and stress. Dopamine is a neurotransmitter wh...
Q: In DNA extracting. What is the purpose of clear shampoo in the DNA extraction buffer?
A: DNA is extracted from different sources to analyze and study the DNA sequence and diseases caused du...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Distinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in animal muscle is a class 1 aldolase, which forms a Schiff base intermediate between substrate (for example. fructose-1, 6-bisphosphate or dihydroxyacetone phosphate) and a lysine at the active site (see Figure I8.12). The chemical evidence for this intermediate conies from studies with aldolase and the reducing agent sodium borohydride, NaBH4. Incubation of the enzyme with dihydroxyacetone phosphate and NaBH4 inactivates the enzyme. Interestingly, no inactivation is observed if NabH4 is added to the enzyme in the absence of substrate. Write a mechanism that explains these observations and provides evidence for the formation of a Schiff base intermediate in the aldolase reaction.Examine the ActiveModel for alcohol dehydrogenase and describe the structure and function of the catalytic zinc center.Using the ActiveModel for enoyl-CoA dehydratase, give an example of a case in which conserved residues in slightly different positions can change the catalytic rate of reaction.
- The Reactions and Meehanisms of the Leloir Pathway Write the reactions that permit galactose to be utilized in glycolysis. Write a suitable mechanism, tor one of these reactions.Using the ActiveModel for aldose reductase, describe the structure of the TIM barrel motif and the structure and location of the active site.The catalytic efficiency of many enzymes depends on pH. Chymotrypsin, which has a well-known catalytic mechanism, shows a maximum value of kcat/Km at pH 8.0. A) Draw a pH curve of chymotrypsin activity over the pH range of 5 to 10 and briefly explain the rationale within the context of catalysis for your depiction. In particular, note how kcat and Km may change over this pH range. B) Enzymes of the a-amylase family catalyze a reaction by forming a covalent intermediate analogous to chymotrypsin, but to a conserved aspartate residue. Illustrate a catalytic mechanism containing a tetrahedral intermediate for a glycogen debranching enzyme based upon its potential membership in the a-amylase family. (don’t need to draw a whole glycogen)
- Behenate (C22H44O2) can be obtained by chain elongation from palmitate. How many ATP equivalents are produced in the degradation of behenate to eleven acetyl-CoA?Enolase catalyzes the reversible reaction shown below. Draw the detailed mechanism for the reaction that occurs in gluconeogenesis, namely the reaction of phosphoenolpyruvate to form 2-phosphoglycerate (the reverse direction in the reaction below).Galactose 1-phosphate: UDP-glucose uridyltransferase (GALT) is an enzyme important for the catabolism of galactose. Patients with GALT deficiency present within days of birth with feeding difficulty, fatigue, jaundice and liver problems, and failure to thrive. In addition, a buildup of galactose and its metabolites are present in the bloodstream. GALT catalyzes the following reaction in galactose catabolism. NOTE that GALT is reversible!! Classify this enzyme and explain why you classified it in this way.
- In working skeletal muscle under anaerobic conditions, glyceraldehyde 3-phosphate is converted to pyruvate (the payoff phase of glycolysis), and the pyruvate is reduced to lactate. Write balanced biochemical equations for all the reactions in this process, with the standard free-energy change for eachreaction. Then write the overall or net equation for the payoff phase of glycolysis (with lactate as the end product), including the net standard free-energy change.Draw a schematic illustration of the hydrolysis of N-acetylphenylalaninamide catalyzed byalpha-chymotrypsin in which you indicate the important catalytic residues in the active site and how thesubstrate undergoes transformation to products through two different tetrahedral intermediates.The high phosphoryl transfer potential of 1,3-bisphosphoglycerate is explained by the resonance stability of the product, 3-phosphoglycerate. The same explanation applies to the reaction that generates resonance stabilized pyruvate from phosphoenolpyruvate. Show the resonance structures for both products, 3-phosphoglycerate and pyruvate, and state why the reactant in both reactions is not resonance stabilized.

