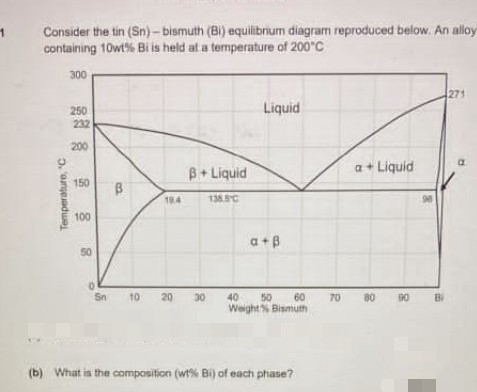
Consider the tin (Sn)- bismuth (Bi) equilibrium diagram reproduced below. An allo containing 10wt% Bi is held at a temperature of 200°C 300 271 250 Liquid 232 200 B+ Liquid a+ Liquid 150 184 1385C 100 a+B 50 Sn 10 20 30 40 60 70 80 90 Bi 50 WeightS Bismuth b) What is the composition (wt% Bi) of each phase? Temperature, C B.
Q: 14. 5 kg of liquid seawater with initial temperature 0°C and salinity 3 percent (by mass) is mixed…
A: Specific heat capacity is a characteristic of a particular pure liquid. It is a minimum heat…
Q: na H2O2 (hp) at 333.15 R, some liquid and vapor compositions and (total) vapor pressures are UIS OI…
A:
Q: (b) Surface sanitizers were prepared using ethyl alcohol/water and isopropyl alcohol/water…
A: According to Raoult's law, we have the following relation regarding vapour pressure : P=xP0 .. (1)…
Q: 10 50 -Hot 30 20 0.2 0.8 MOLE FRACTI IEmiPER ÀTURE (oc)
A: The mole fraction represents the number of molecules of a particular component in a mixture divided…
Q: A liquid mixture containing ether CH3-O-CH3 at a ratio of 0.58 and methano; CH3OH if the total…
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be…
Q: 4. One component exists in two phases at the same temperature, but the phases are not in…
A: One component exists in two phases at the same temperature, but the phases are not in equilibrium.…
Q: Carbon tetrachloride melts at 250 K. The vapor pressure of the liquid is 10539 Pa at 290 K and 74518…
A: (A) The final boiling point of carbon tetrachloride = Tf= 340 K The boiling temperature is = Ti =…
Q: For any composition other than pure components in a binary isomorphous system, the melting…
A: Binary isomorphous system: A system in which all components of solid have same structure is called…
Q: boiling point boiling point BPtof pure A vapour composition liquid composition Bpt of pure B O A 1.0…
A: Given data :-
Q: Figure 5.8 shows the phase diagram for the ternary system NH4Cl/(NH4)2SO4/H2O at 25 °C. Identify the…
A: The given ternary system is, NH4Cl/(NH4)2SO4/H2O. The first mole fraction is corresponds to NH4Cl,…
Q: boiling point boiling point BPtof pure A vapour composition liquid composition Bpt of pure B OA 1.0B…
A:
Q: a. What is the chemical formula of the solid represented by the vertical phase boundary passing…
A: The chemical formula find from mole fraction which is given below.
Q: For the ZrO2 -CaO system identify and write all eutectic and eutectoid reactions, and also for each…
A: Find the complete detailed explanation given below
Q: In the system NaCl-H2O, a simple eutectic is observed at −21.1°C for a solution containing 23.3% by…
A:
Q: Consider the given liquid-vapor diagram: 375 370 365 360 355 350 345 340 0.1 02 0.8 03 04 05 0.5 0.7…
A: The given liquid-liquid phase diagram states the composition of the vapor and corresponds to the…
Q: A vapor mixture containing 150 kg of benzene and 350 kg of toluene is initially at 115°C. What is…
A:
Q: In an experimental study of membrane-like assemblies of synthetic materials, a phase diagram like…
A: First, consider At point a, the mixture is in liquid form. Suppose the liquid mixture reaches the…
Q: a)Write a balanced equation for the dissolution of PbBr2. Express your answer as a chemical…
A:
Q: The table below shows temperature/composition data collected for a mixture of methylbenzene (M) and…
A: Given: Boiling point of methylbenzene (M) = 110.60C Boiling point of octane (O) = 125.60C
Q: sing the lead-lin phase diagram in gure or a nominal composition of 90 % Sn and 10 % Pb at 204 °C.…
A: Solution -
Q: Atomic weights in g/mole: H = 1.0, C = 12.0, N = 14.0, O = 16.0, F = 19.0, Cl = 35.5, Cu = 63.5 =…
A: Entropy is basically the measure of randomness of the particles in a system. Its units are joules…
Q: eter to Fig. 5.15. Suppose you have a mixture containing 20 wt% acetic acid, 60 wt% water and the…
A: Solution - According to the question - Given -
Q: TIK |210 Compositon of one phase P.1 Vapour 292 290 P+2 Composton fsecond phase P2 Liquid 273 P.1…
A:
Q: For alloys of two hypothetical metals A and B, there exist an a, A-rich phase and a ß, B-rich phase.…
A:
Q: At 25°C a binary system containing components A and B is in a state of liquid-liquid vapor…
A: A binary mixture consists of two types of molecules for example A and B and X is the mole fraction…
Q: At 25°C a binary system containing components A and B is in a state of liquid-liquid vapor…
A: A binary mixture consists of the two types of molecules for example of A and B and X is the mole…
Q: Lead and tin are mixed to make solder for use in circult boards and plumbing applications. A…
A: Given that: Teutectic = 80.2 oC = 353.2 K ΔHofus (Pb) = 4.77 kJ/mol Tofus (Pb) = 327.46 oC = 600.46…
Q: At 183.1°C, just above the eutectic line, what phase(s) is (are) present?
A:
Q: Determine the following from the Txy diagram of a two-component (A and B) solid-liquid system:…
A: The given phase diagram is:
Q: (h) Consider the binary phase diagram below. i. What is the eutectic composition and temperature? At…
A: 1. A eutectic mixture is a homogeneous mixture which melts at a single melting temperature , which…
Q: 1000 912 sio 700 s00 400 Weighs Pere Siver For an alloy containing 20 wt % Ag, refer to the Cu-Ag…
A:
Q: Water boils at 100 oC at 1 atm (101325 Pa). Tom dissolve 574.4 g of sugar (MW = 342.3 g/mol) in…
A: Raoult's law: According to this law pressure of solution is equal to the product of mole fraction of…
Q: 5. The following temperature/composition data were obtained for a mixture of octane (0) and…
A: The temperature/composition data has to be plotted on a graph for a mixture of methylbenzene(M) and…
Q: At 90 degrees celsius the vapor pressure of 1,2-dimethylbenzene is worth 20kPa and the vapor…
A: The given information is: Temperature = 90°C Vapor pressure of 1,3 dimethylbenzene(P01) = 18 kPa…
Q: a.) A 12 kmol liquid mixture with XSi = 0.8, xca = 0.2 at 1500oC was cooled to 1030oC. Determine the…
A: A question based on phase diagram, which is to be accomplished.
Q: Calculate mole fraction of each component in a reaction vessel of volume 24.4 dm containing 1.0 mol…
A: Mole fraction of a particular component in a solution is the ratio of number of moles of that…
Q: a) What is relative volatility? What happens when the relative volatility is 1) equal to "one" ii)…
A:
Q: Make schematic sketches of the microstructures that would be observed for conditions of very slow…
A:
Q: K.m 10,20,30 and 40 К. ol /hr of a mixture containing mol % of propane, n-butane, n-peutane, and…
A: I think some values are missing,as i have tried this problem.please look it once,I will solve for…
Q: Laboratory works were carried out to measure the composition of vapor and liquid of chloroform (1)…
A:
Q: We discussed the temperature-composition phase diagram for a binary system given below. Mole…
A: The number of phases that exists in equlibrium in a system depends on the variables temperature,…
Q: 4. Various mixtures of ethanol and ethyl acetate were heated to different temperature at a pressure…
A: Phase diagram Phase diagram tells about the different states of matter under different conditions…
Q: The composition of the liquid and the vapor that are in equilibrium is not necessarily the same. T…
A: Given statement is : The composition of the liquid and the vapor that are in equilibrium is not…
Q: Table 2. Gibbs Free Energies of formation (kJ), ∆G°f, for Ions in 1M Solution and Ionic Solids…
A:
Q: Comelli and Francesconi examined mixtures of propionic acid with various other organic liquids at…
A: (a) Component 1 is taken as propionic acid. Component 2 is taken as THP. The expression for molar…
Q: en that must be added at 35 C to change the volumetric analysis to 50% of each component.
A: We have, V=3m3 ,P=101kPa,T=35o C+273k=308k,YCH4 =60%100% =0.60YO2 =40%100% =0.40
Q: 1. Solid-Liquid Equilibrium 1500 Liquid 1314 1324 1300 1268 1100 1040 1030 90아 700 0.2 0.4 0.6 0.8…
A: A question based on phase diagram, which is to be accomplished.
Q: The following table gives the vapor pressures of liquid chloroform(1) + acetone(2) solutions at 35…
A: In volatile substance that is in liquid state, some amount of liquid evaporates that the…
Q: A simple distillation flask (V=2L) is initially charged with 320 moles of a solution consisting of…
A: Distillation is a process by which one can separate out two miscible liquids (one of them is…
Step by step
Solved in 2 steps with 1 images

- 10.11/ A 1.5-kg specimen of a 90 wt% Pb-10 wt% Sn alloy is heated to 250°C (480°F), at which temperature it is entirely an a-phase solid solution . The alloy is to be melted to the extent that 50% of the speci- men is liquid, the remainder being the a phase. This may be accomplished either by heating the alloy or changing its composi- tion while holding the temperature con- stant. (a) To what temperature must the speci- men be heated? (b) How much tin must be added to the 1.5-kg specimen at 250°C to achieve this state? 10.15) A magnesium-lead alloy of mass 5.5 ka consists of a solid a phase that has a compo- sition that is just slightly below the solubility limit at 200°C (390°F). (a) What mass of lead is in the alloy? (b) If the alloy is heated to 350°C (660°F). how much more lead may be dissolved in the a phase without exceeding the solubility limit of this phase? 10.27 A hypothetical A-B alloy of composition 55 wt% B-45 wt% A at some temperature is found to consist of mass…Polyethylene glycol 400 (PEG400) is a nonvolatile liquid polymer, which when mixed with water canbe used as a base for parenteral formulations. The designation “400” means that the molecular weightof PEG molecules is approximately 400 g/mol. Preparation with water (ΔHv = 40.7 kJ/mol) requires that the two be mixed at 56 oC, at which the equilibrium vapor pressure of pure water is 16.5 kPa. a. If 105 g of PEG400 are added to 500 g of water at 56 oC, what will the boiling temperature of the solution be?In a system ABC, a ternary alloy with composition of 30 wt. % B and 30 wt. % C consists at a particular temperature of three phases of equilibrium compositions as follows:Liquid phase: 50 % A, 40 % B, 10 % Calfa solid solution: 85 % A, 10 % B, 5 % Cbeta solid solution: 10 % A, 20 % B, 70 % Ca. Plot the composition of the alloy.b. Calculate the proportion by weight of liquid, and present in this alloy.c. For the same temperature, find the composition of the alloy which will consist of equal proportion of and phases of the compositions stated above, but with no liquid phase.
- A 90 wt% Ag-10 wt% Cu alloy is heated to a temperature within the β + liquid phase region. If the composition of the liquid phase is 85 wt% Ag, determine:(i) The temperature of the alloy(ii) The composition of the β phase(iii) The mass fractions of both phasesFor a Copper-Nickel (Cu-Ni) alloy of composition 25 wt % Ni and 75 wt % Cu at 1200 OC (Point B), refer to the phase diagram provided in Figure 2 and answer the following. (i) What is composition of liquid phase? (ii) What is composition of solid phase? (iii) What fraction of the system is liquid for a 25% Ni alloy at 1250 OC? (iv) What fraction of the system is solid for a 25% Ni alloy at 1250 OC?GRAVIMETRIC ANALYSIS The addition of dimethylglyoxime, H2C4H602N2, to a solution containing nickel(Il) ion gives rise to a precipitate: Nickel dimethylglyoxime is a bulky precipitate that is inconvenient to manipulate in amounts greater than 135 mg. The amount of nickel in a type of permanent-magnet alloy ranges between 22% and 38%. Calculate the sample size that should not be exceeded when analyzing these alloys for a nickel.
- The composition of the liquid and the vapor that are in equilibrium is not necessarily the same. T or FIn the Deacon process for the manufacture of chlorine, HCl and O2 react toform Cl2 and H2O, Sufficient air (21 mol% O2, 79% N2) is fed to provide 35% excess oxygen and the fractional conversion of HCl is 85%. (a) Calculate the mole fractions of the product stream components, usingatomic species balances in your calculation. (b) Again calculate the mole fractions of the product stream components,but use the extent of reaction in the calculation.A monolayer of CO molecules is adsorbed on the surface of 1.00 g of an Fe/AI2O3 catalyst at 77 K, the boiling point of liquid nitrogen. Upon warming, the carbon monoxide occupies 4.25 cm3 at O oC and 1.00 bar. What is the surface area of the catalyst?
- Given the solubility of ferrihydrite (Fe(OH)3) and goethite (FeOOH), with dissolution reactions shown below: [1] Fe(OH)3 3 H+ à Fe3+ + 3 H2O Ksp = 8.4 x 10^4 ∆ Gr0 = -28 KJ/mol Solubility of Fe(OH3) = 7.468 [2] FeOOH + 3 H+ à Fe3+ + 2 H2O Ksp = 8.8 x 10^-2 ∆ Gr0 = 6 KJ/mol Solubility of FeOOH = 0.353 QUESTION: [A] How much Fe3+ from ferrihydrite would dissolve at pH 2 ? [B] How much Fe3+ from ferrihydrite would dissolve at pH 7 ? [C] How much Fe3+ from goethite would dissolve at pH 2 ? [D] How much Fe3+ from goethite would dissolve at pH 7 ?Phase Behavior of Elemental Gallium: Gallium (Ga) is one of the few metals that is liquid near room temperature (Tm = 302 K). a) The vapor pressure for Ga has been measured at 100 Pa at 1620 K and 10 kPa at 2125 K. What is the average metal-metal bond strength (in kJ/mol) for Ga assuming a liquid state coordination number of 6? b) What is the boiling point ( in K) for Ga in a standard lab at sea-level given the above informationThe table below shows temperature/composition data collected for a mixture of methylbenzene (M) and octane (O) at 1 atm. Recall that x stands for the mole fraction in the liquid and y stands for the mole fraction in the vapor in equilibrium. The boiling points for methylbenzene (M) and octane (O) are 110.60C and 125.60C, respectively. Construct the phase diagram with Temperature vs. xM. What is the composition of the vapor in equilibrium with the liquid of composition (a) xM = 0.250 and (b) xO = 0.250. T (0C) 110.9 112.0 114.0 115.8 117.3 119.0 121.1 123.0 xM 0.908 0.795 0.615 0.527 0.408 0.300 0.203 0.097 yM 0.923 0.836 0.698 0.624 0.527 0.410 0.297 0.164

