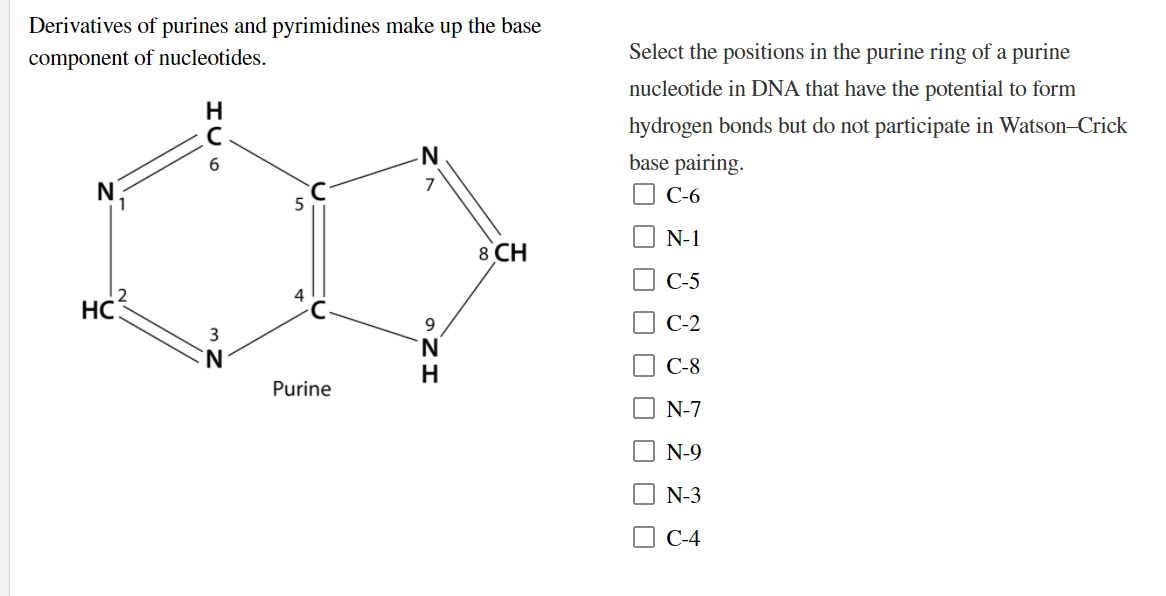
Derivatives of purines and pyrimidines make up the base component of nucleotides. Select the positions in the purine ring of a purine nucleotide in DNA that have the potential to form hydrogen bonds but do not participate in Watson-Crick 6. base pairing. 7 С-6 N-1 8 CH С-5 HC. 9. С-2 C-8 Purine N-7 N-9 O O O O O O
Q: The portion (the C-terminal end) of original substrate with the new amino terminus diffuses away.…
A: Introduction: Enzymes have a spectacular ability to accelerate the rate of a chemical reaction.…
Q: 1. Why is it important in Quantitative Analysis to postpone rounding until the calculation is…
A: A quantitative analysis is performed to detect the amount of specific substance in a sample…
Q: Some enzymes can be inhibited by high concentrations of their substrates. I expression for the rate…
A: In the biological systems , enzymes acts as catalysts . Enzyme help to accelerate the reactions.…
Q: Which of the following cause and effect relationships below is incorrect? higher ligand…
A: Which of the following cause and effect relationships below is incorrect? higher ligand binding:…
Q: 2. ( To the right is a schematic diagram of His the active site in the Michaelis complex of a-chy-…
A: Chymotrypsin is a protease that cleaves a peptide at the C-terminal of all aromatic amino acid.…
Q: estion properly and accordingl
A: Saponification is the process of forming a metallic salt of a fatty acid, which is referred to as a…
Q: 29. Here is a strand of DNA: TACCCGGTATAACGCTGGAAAGCTTAAGCAACATCGCCCGACCCC AAATCT…
A: DNA strand given here with directionality is as: 5’…
Q: Explain the biochemical consequences of ADA deficiency and explain them using the purine degradation…
A: The purine nucleotides are sequentially degraded by the removal of portions of nucleotides. The…
Q: -Inhibitor +Inhibitor [S] (mM) Vο&νβσπ: (μmol/sec). Vο&νβσπ:&νβσπ: (μmollsec) 0.0001 33 17 0.0005 71…
A: From the given data, I have calculated 1/S and 1/V0 in absence and presence of inhibitor. The plot…
Q: one more example of chemical (besides acids and alkalis) which can also affect DNA stability.…
A: DNA can be denatured by process of separating dsDNA into single strands, factors like temperature,…
Q: what is the chemistry of nucleosides?
A: Nucleosides are nothing but glycosamines whose analogues are used as anti cancer agents or antiviral…
Q: ssume that you have radioactively labeled an amino acid in a glass of milk. Also assume that you…
A: Introduction: The term digestion refers to the integration of complex nutrients into simple, soluble…
Q: Examine which of the following statement are FALSE about enzymes. * (Please choose one correct…
A: Enzymes are substances that enhance the rate of chemical reaction and facilitate the formation of…
Q: Choose two amino acid and explain the metabolism.
A: Introduction: Amino acids are molecules that contain an amine and carboxylic group with side-chain…
Q: reaction as a function of substrate concentration. Ex- plain why the maximal velocity can be…
A: Enzyme kinetics is the study of the rate of enzyme catalyzed bio chemical reactions and also we can…
Q: Why does it make sense that under conditions of low ATP levels in the cell the pyruvate carboxylase…
A: Pyruvate carboxylase (PC) is a ligase class enzyme which catalyze the irreversible carboxylation of…
Q: Starting from the O2 binding equilibrium of human hemoglobin written below, derive the Hb + nO2 2…
A: Hemoglobin is an oligomeric conjugated protein with four peptide chains joined by a non-covalent…
Q: Match the following descriptions to the given choices Synthesized from a steroid molecule A. Vitamin…
A: Vitamin is an organic substance and an essential micronutrient that an organism needs in small…
Q: 3. In what direction (toward the anode, toward the cathode, or toward neither) will the following…
A: As like amino acids, peptides, and proteins include both acidic and basic functional groups. The…
Q: Describe the mutational event that produces the MYC oncogene in Burkitt’s lymphoma. Why does the…
A: Burkitt lymphoma is a type of non-Hodgkin lymphoma that affects adults and children. NHL is a…
Q: но но- но он HN. он O Cerebroside O Sphingosine O Ganglioside O Ceramide
A: Cerebrosides are lipid complex , present in the sheaths of nerve fibers. Sphingosine forms a primary…
Q: Why does it make sense that under conditions of low ATP levels in the cell the pyruvate carboxylase…
A: Glucose is degraded to pyruvate through the process of glycolysis that occurs in the cytoplasm.…
Q: Break down this fatty acid. Show all the products made and the enzymes needed for any non-normal ß…
A: Fatty acids are building blocks of fats and composed of carboxylic acid with long aliphatic chain.…
Q: ection steps! Which of the following are proper disir ORemove organic matter O Disinfect only O…
A: Microscopic organisms such as bacteria, fungi (mold and yeast), protists, archaea, algae,…
Q: Please explain where you would find glycosaminoglycans and what is the importance of these…
A: GAGs are negatively charged linear polysaccharides that are frequently found conjugated with…
Q: Q1: Explain the effect of pH value on the amino acid ionization. Q2: Describe two reactions for…
A: Amino acids and their ionization: Amino acids are the basic building blocks of proteins, they are…
Q: Draw the Fischer projection formula for each sugar and give the importance/use of each. 1. D-…
A: Note : Hi ! Thank you for the question. We are authorized to answer three subparts at a time. Since…
Q: How many activation cycles, Initiation cycles, Elongation cycles and termination cycles are needed…
A: Protein synthesis occurs in four main steps such as activation or charging of tRNA, initiation of…
Q: CHALLENGE QUESTION I: Smurf hemoglobin has a p50 of 30 torr and has 8 subunits, instead of the usual…
A: Consider the Protein (P) is getting bound by the Ligand (L) to form the Protein-Ligand complex (PL)…
Q: A. Name the two proteins that induce a negative feedback in this pathway and explain why both…
A: Since, you have posted multiple questions we will solve the first question for you. If you want any…
Q: In the Haworth projection for Glucose the hydroxyl on carbon 1 is shown projected up or down. b In…
A: As you have asked multiple sub part questions we are supposed to answer only first 3 subparts for…
Q: Failed to follow
A: Waxes are a broad category of organic compounds that are lipophilic and bendable solids at room…
Q: TIONS (Continued)
A: 5) Hydrololysis is a reaction in which reactant react with water and decomposition of water and…
Q: What is the role of the prep phase in glycolysis? To convert G3P molecules into pyruvate and produce…
A: In glycolysis, the glucose molecules are broken down into two molecules of pyruvate along with the…
Q: Substrate cycling does not violate the laws of thermodynamics in making both directions of a…
A: The study of energy and energy transfer in physical matter is known as thermodynamics. A system is…
Q: List the key challenges in the biosynthesis phosphatidylcholine.
A: The most common PL identified in circulating VLDL is phosphatidylcholine (PC) . PC is produced in…
Q: Where does molecular oxygen (O2) get generated during photo-phosphorylation? Photosystem I 2…
A: The light reaction, also known as photolysis reaction, occurs in the presence of light. It mainly…
Q: Begining with 1 M concentrations of each reactant and product at pH=7 and 25.0 degrees C, calculate…
A: As given in the question, the concentration of each reactant, i.e., Pyruvate and NADH, and products,…
Q: Only one of the statements below is correct; which one? Two solutions are hypotonic when they have…
A: Tonicity is a parameter of the effective osmotic pressure gradient, which is the difference in water…
Q: Match the following descriptions to the given choices. Enzyme involved in the conversion of…
A: The above match the following is from an important pathway involving the biosynthesis of…
Q: 4. Complete the table below: RNA DNA Strand Sugar residue Nitrogenous bases Main Function
A: Nucleic acids are made up of nucleotides, which are the fundamental building blocks of DNA and RNA.…
Q: Derive the amino acid sequence that is coded for by each of the following mRNA sequence. 5' CAA…
A: Amino acid are molecules which are combine to form a protein molecules.Amino acid are synthesized by…
Q: A. Effect of pH with enzymatic activity. Include example of enzymes optimum pH. B. Effect of…
A: The enzymes are biological catalysts, that enhance the rate of biochemical reactions. The factors…
Q: A newly developed qPCR has an efficiency of 75%, and each cycle is pretty consistent. In this qPCR,…
A: The qPCR is generally useful to determine the actual value of PCR product present at provided…
Q: b. Two different liposomes have radii of 50.00 nm and 70.00 nm respectively. In a biopolymer…
A: Introduction: Liposomes are simple microscopic vesicles in which aqueous volume is enclosed entirely…
Q: Principle involved in the isolation of gluten * (Please choose one correct answer only) A.…
A: The protein component of wheat flour is called Gluten. This basic component gives elasticity and…
Q: (1) Describe the nitrogenous base pairing in DNA and RNA. (2) Determine the number of bonds in each…
A: DNA and RNA are polynucleotides. Monomers of nucleic acids are nucleotides. Nucleotides are made up…
Q: These are enzymes that can sustain in a high hydrostatic pressure. * (Please choose one correct…
A: Bacteria are classified into different classes based on the influence of different environmental…
Q: CH3 CH3 CH3(CH2)n: O sterol O Cholesterol O Lysophosphatidylcholine O Cholesteryl ester
A: Sterols are important sub groups of steroids that contain OH at 3rd position of A ring of structure.…
Q: Match the following descriptions to the given choices.…
A: A lipid is a biomolecules which include fats, waxes, oils, hormones, and certain components of…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Draw the structure of the following DNA sequence (5’-AG-3’) hydrogen bonded through Watson-Crick base pairing to the complementary PNA sequence (Cterminus-TC-Nterminus). Make sure to include both the sugar phosphate backbone of your DNA sequence, as well as the peptide backbone of your PNA sequence. DNA: 5’-AG-3’ PNA: (C)-TC-(N)What is the nucleotide sequence of the complementary strand of the DNA molecule: 5’-AATGCGATCTTCAT-3’? Indicate the 5’ and 3’ ends. Follow the same format as the given sequence.Which of the following cannot be said concerning base-pairing complementarity in DNA or RNA? Uracil forms 2 hydrogen bonds with Adenine. Thymine forms 2 hydrogen bonds with adenine. Purines pair with pyrimidines. Guanine forms 3 hydrogen bonds with cytosine. Pyrimidines pair with pyrimidines and purines pair with purines.
- A strand of nucleic acid is defined by its sequence of nucleotides: A, C, T, and G. How many different sequences are possible for a nucleic acid that is 200 nucleotides long? How does that number compare to the estimated number of atoms in the universe, which is approximately 1080?Indicate whether each of the following base-pairing situations (1) involves two DNA strands, (2) involves a DNA strand and an RNA strand, or (3) could involve either two DNA strands or a DNA strand and an RNA strand? a. A G T U C A b. A C T T G A c. A G U T C A d. C G C G C G a. A G T U C A b. A C T T G A c. A G U T C A d. C G C G C GIn the Watson-Crick structure of DNA, the: a. adenine content of one strand must equal the thymine content of the same strand. b. nucleotides are arranged in the A-form. c. purine content (fraction of bases that are purines) must be the same in both strands. d. two strands are parallel. e. the strands are complementary to each other.
- Number of hydrogen bonds that form between U and A in a Watson-Crick base pair interactions?a) 0b) 1c) 2d) 3Given the following mismatch (highlighted in red), which base will be replaced after mismatch repair occurs? Mutant sequence (CH3 = methyl): CH3 I 5'-GATCTCAGGC-3' 3;-CTAGAGGCCG-5' a. G b. AThe specificity of Watson-Crick nucleobase pairing enables the biological information to be encoded and passed down through generations of cells and organisms. This specificity arises from the chemical structure of the base pairs, namely the pattern of hydrogen bond donors and acceptors that stabilize only two pairing interactions] relative to the others. How many possible pairwise interactions could potentially exist at a single position within a sequence, including the correct pairs, and all possible other combinations of DNA bases?
- The specificity of Watson-Crick nucleobase pairing enables the biological information to be encoded and passed down through generations of cells and organisms. This specificity arises from the chemical structure of the base pairs, namely the pattern of hydrogen bond donors and acceptors that stabilize only two pairing interaction relative to the others. How many possible pairwise interactions could potentially exist at a single position within a sequence, including the correct pairs, and all possible other combinations of DNA bases?The base composition for one of the strands of a DNA double helix is 19% A, 34% C, 28% G, and 19% T. What is the percent base composition for the other strand of the DNA double helix?An exonuclease is an enzyme that sequentially cleaves nucleotides from the end of a polynucleotide strand. Snake venom phosphodiesterase, which hydrolyzes nucleotides from the 3′ end of any oligonucleotidewith a free 3′-hydroxyl group, cleaves between the 3′ hydroxyl of the ribose or deoxyribose and the phosphoryl group of the next nucleotide. It acts on single-stranded DNA or RNA and has no base specificity. This enzyme was used in sequence determination experiments before the development of modern nucleic acid sequencing techniques. What are theproducts of partial digestion by snake venom phosphodiesterase of an oligonucleotide with the sequence (5′)GCGCCAUUGC(3′)—OH?

