Extension Questions 1. A sample with an unknown concentration of Cr(NO3)3 has an absorbance of 0.68 at A of 426 nm. A calibration curve was produced for Cr(NO3)s and is shown below in Figure 1. Absorbance 0.5 4.63E-03*x + 4.4E-03 0.4 0.3 0.2 0.1 0 25 50 75 100 Concentration (mM) Figure 1. Calibration curve for Cr(NO) collected at 426 nm a. Determine the molar absorptivity (E) ifa 1.5 cm path length cuvette was used to collect data. Utilize the slope of the calibration curve above to determine the molar absorptivity value. b. Determine the concentration (mM) of Cr(NO3) in the unknown solution. n 9 c. A different lab group used a 1.0 cm path length cuvette in the preparation of a calibration curve using the same concentrations of Cr(NO3)3 space below, quantitatively draw out what the new calibration curve would look like. as in Figure 1. In the Include the axes labels and numerical values. Absorbance
Extension Questions 1. A sample with an unknown concentration of Cr(NO3)3 has an absorbance of 0.68 at A of 426 nm. A calibration curve was produced for Cr(NO3)s and is shown below in Figure 1. Absorbance 0.5 4.63E-03*x + 4.4E-03 0.4 0.3 0.2 0.1 0 25 50 75 100 Concentration (mM) Figure 1. Calibration curve for Cr(NO) collected at 426 nm a. Determine the molar absorptivity (E) ifa 1.5 cm path length cuvette was used to collect data. Utilize the slope of the calibration curve above to determine the molar absorptivity value. b. Determine the concentration (mM) of Cr(NO3) in the unknown solution. n 9 c. A different lab group used a 1.0 cm path length cuvette in the preparation of a calibration curve using the same concentrations of Cr(NO3)3 space below, quantitatively draw out what the new calibration curve would look like. as in Figure 1. In the Include the axes labels and numerical values. Absorbance
Chapter4: Least-squares And Calibration Methods
Section: Chapter Questions
Problem 11P
Related questions
Question
Please answer all the parts of the question that correspond to number 1 (a, b and c). Please show all work and explain all the steps.
Thank you.
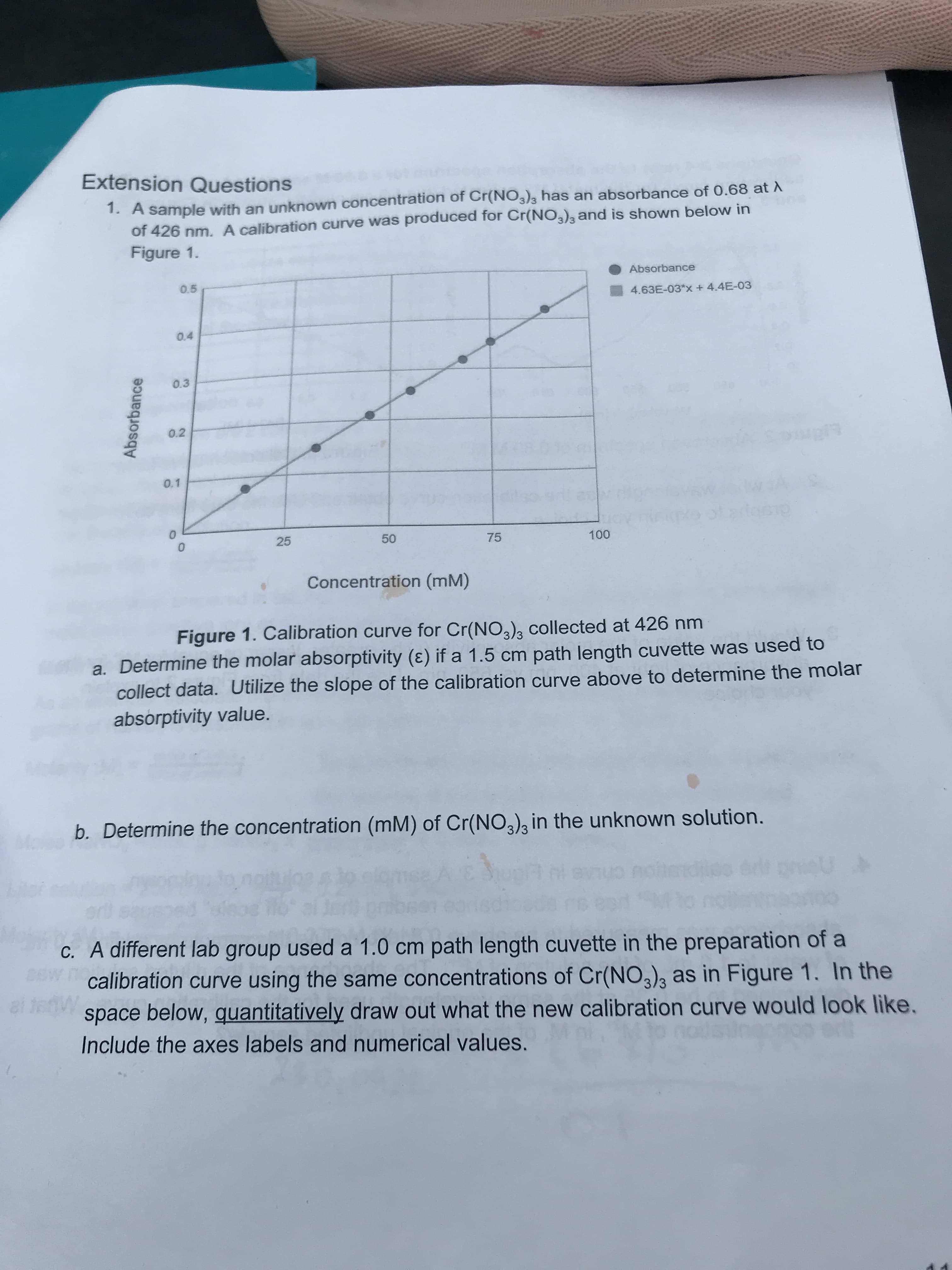
Transcribed Image Text:Extension Questions
1. A sample with an unknown concentration of Cr(NO3)3 has an absorbance of 0.68 at A
of 426 nm. A calibration curve was produced for Cr(NO3)s and is shown below in
Figure 1.
Absorbance
0.5
4.63E-03*x + 4.4E-03
0.4
0.3
0.2
0.1
0
25
50
75
100
Concentration (mM)
Figure 1. Calibration curve for Cr(NO) collected at 426 nm
a. Determine the molar absorptivity (E) ifa 1.5 cm path length cuvette was used to
collect data. Utilize the slope of the calibration curve above to determine the molar
absorptivity value.
b. Determine the concentration (mM) of Cr(NO3) in the unknown solution.
n
9
c. A different lab group used a 1.0 cm path length cuvette in the preparation of a
calibration curve using the same concentrations of Cr(NO3)3
space below, quantitatively draw out what the new calibration curve would look like.
as in Figure 1. In the
Include the axes labels and numerical values.
Absorbance
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning