ezto.mneduca .com/ext/map/index.html?_con=conočexternaI_browser=UQlaunchuri=nttps7o25 7252Fewconn /activity/q... Apps My STC O Mail - Janeth Soto ... * CengageNOW | Onl. Hidalgo County O Yahoo Mail M HBO Max * ScheduleConnect cp Colonia Self-Help C... E Reading Electrochemistry lab alternative O Save & Exit Submit Saved Help 15 A spontaneous galvanic cell consists of a Pb electrode in a 1.0 M Pb(NO3)2 solution and a Cd electrode in a 1.0 M Cd(NO3)2 solution. What is the standard cell potential for this galvanic cell? Pb2*(ag) + 2e Pb E° = -0.13 V Ca2*(ag) + 2e → Cd E° = -0,40 V E° cell=
ezto.mneduca .com/ext/map/index.html?_con=conočexternaI_browser=UQlaunchuri=nttps7o25 7252Fewconn /activity/q... Apps My STC O Mail - Janeth Soto ... * CengageNOW | Onl. Hidalgo County O Yahoo Mail M HBO Max * ScheduleConnect cp Colonia Self-Help C... E Reading Electrochemistry lab alternative O Save & Exit Submit Saved Help 15 A spontaneous galvanic cell consists of a Pb electrode in a 1.0 M Pb(NO3)2 solution and a Cd electrode in a 1.0 M Cd(NO3)2 solution. What is the standard cell potential for this galvanic cell? Pb2*(ag) + 2e Pb E° = -0.13 V Ca2*(ag) + 2e → Cd E° = -0,40 V E° cell=
Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.15QAP
Related questions
Question
Transcribed Image Text:Courses
Question 15 - Electrochemistry la X
b My Questions | bartleby
X * physical chemistry - Positive or N X +
ezto.mheducation.com/ext/map/index.html?_con=con&external_browser=0&launchUrl=https%253A%252F%252Fnewconnect.mheducation.com%252F#/activity/q...
2
Аpps
My STC
Mail - Janeth Soto -...
CengageNOW | On..
Hidalgo County
Yahoo Mail
M HBO Max
ScheduleConnect
Colonia Self-Help C..
E Reading list
Electrochemistry lab alternative i
Help
Save & Exit
Submit
Saved
15
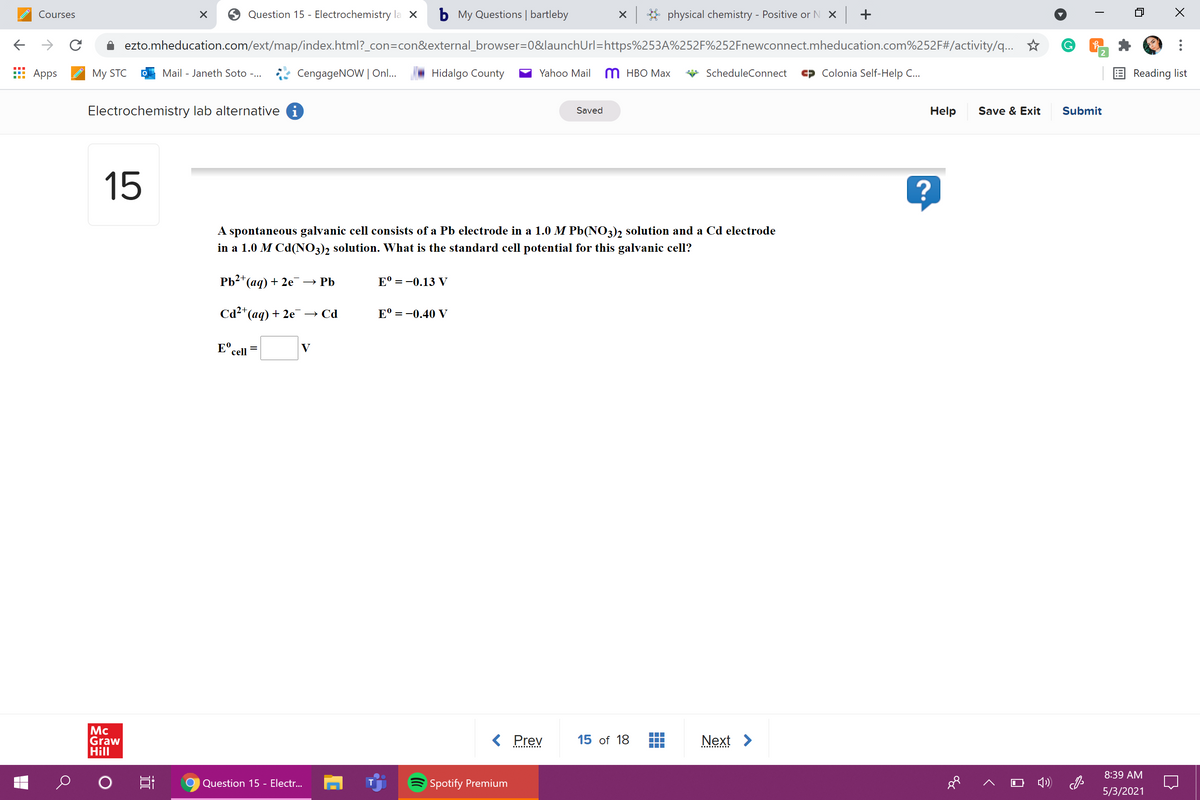
A spontaneous galvanic cell consists of a Pb electrode in a 1.0 M Pb(NO3)2 solution and a Cd electrode
in a 1.0 M Cd(NO3), solution. What is the standard cell potential for this galvanic cell?
2+
Pb+* (aq) + 2е — РЬ
Е 3 -0.13 V
Cd2*(ag) + 2e→ Cd
Е 3 —0.40 V
E°.
V
%3D
cell
Mc
Graw
Hill
< Prev
15 of 18
Next >
8:39 AM
Question 15 - Electr...
Spotify Premium
5/3/2021
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning