Fill out the last column of this table: Component 1X concentration 20X (Provide unit)
Fill out the last column of this table: Component 1X concentration 20X (Provide unit)
Basic Clinical Laboratory Techniques 6E
6th Edition
ISBN:9781133893943
Author:ESTRIDGE
Publisher:ESTRIDGE
Chapter1: The Clinical Laboratory
Section1.8: Laboratory Math And Reagent Preparation
Problem 12RQ
Related questions
Question
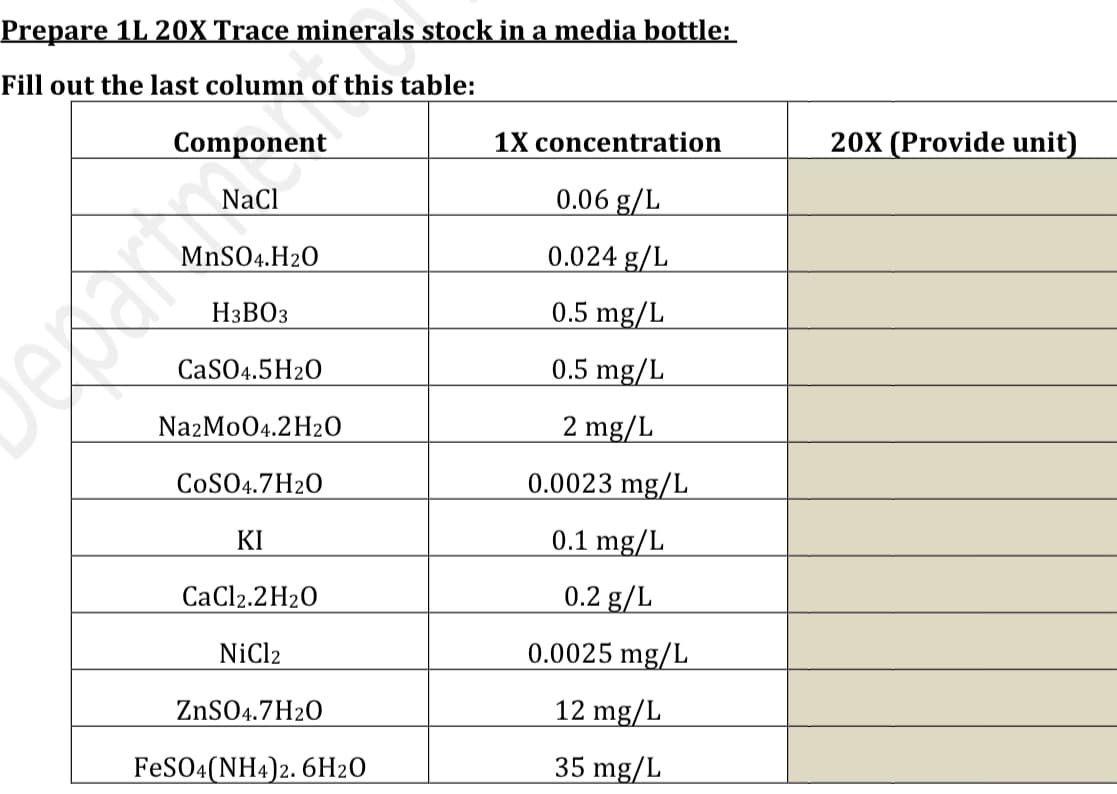
Transcribed Image Text:Prepare 1L 20X Trace minerals stock in a media bottle:
Fill out the last column of this table:
Component
NaCl
MnSO4.H₂O
H3BO3
CaSO4.5H2O
NazMo04.2H₂O
COSO4.7H2O
KI
CaCl2.2H₂O
NiCl2
ZnSO4.7H2O
FeSO4(NH4)2. 6H20
1X concentration
0.06 g/L
0.024 g/L
0.5 mg/L
0.5 mg/L
2 mg/L
0.0023 mg/L
0.1 mg/L
0.2 g/L
0.0025 mg/L
12 mg/L
35 mg/L
20X (Provide unit)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you







Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning


Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College