From this structure, what is the predominant secondary structure of the protein? Based on what you can observe from the protein, what is its highest level of organizational structure? If applicable, how many subunits does this protein have and are they identical? . If electrostatic interactions primarily stabilize this structure, propose a method to test your observations in 4.2 and 4.3.
From this structure, what is the predominant secondary structure of the protein? Based on what you can observe from the protein, what is its highest level of organizational structure? If applicable, how many subunits does this protein have and are they identical? . If electrostatic interactions primarily stabilize this structure, propose a method to test your observations in 4.2 and 4.3.
Human Biology (MindTap Course List)
11th Edition
ISBN:9781305112100
Author:Cecie Starr, Beverly McMillan
Publisher:Cecie Starr, Beverly McMillan
Chapter2: Chemistry Of Life
Section: Chapter Questions
Problem 7RQ: Describe the four levels of protein structure. How do a proteins side groups influence its...
Related questions
Question
3
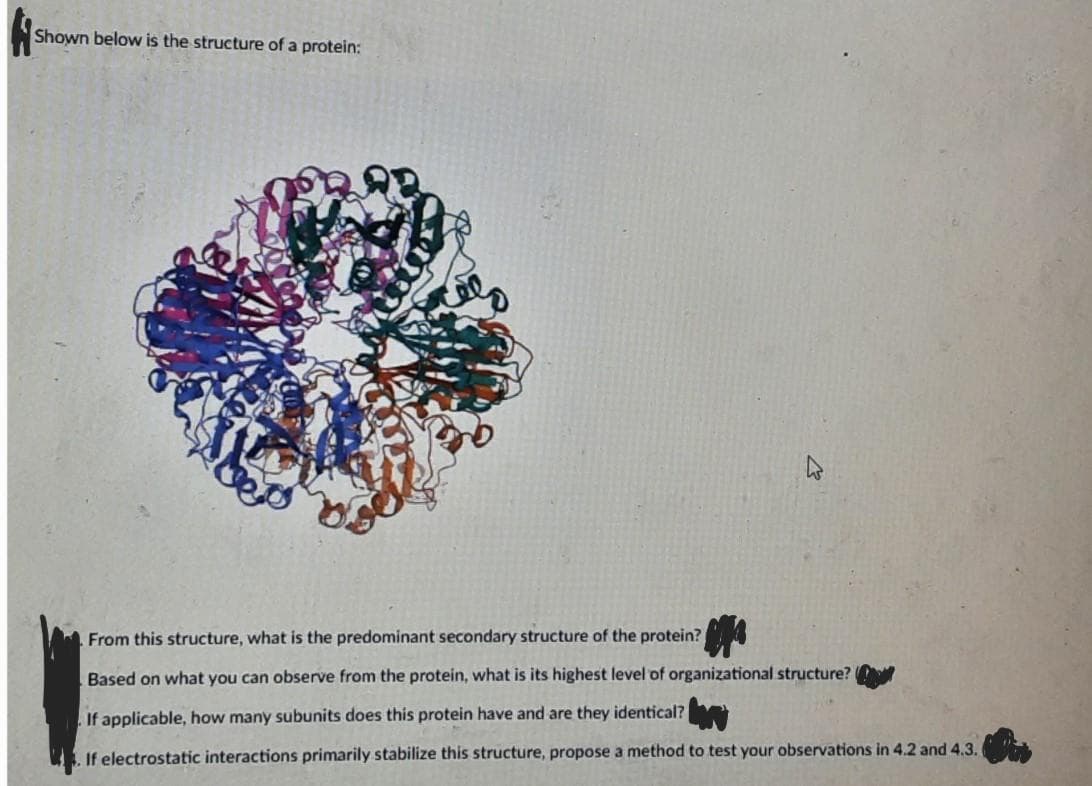
Transcribed Image Text:Shown below is the structure of a protein:
From this structure, what is the predominant secondary structure of the protein?
Based on what you can observe from the protein, what is its highest level of organizational structure?
If applicable, how many subunits does this protein have and are they identical?
If electrostatic interactions primarily stabilize this structure, propose a method to test your observations in 4.2 and 4.3.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:
9781305112100
Author:
Cecie Starr, Beverly McMillan
Publisher:
Cengage Learning