Give the IUPAC name for the following complexes: (i) (ii) Fe. OC- CO C OEC CEO Tc OC OEC (ii) CH3 (iv) CH3 H3C CH CH3 As (OC),Os CH3 CH3 H3C Zr (OC),Os Os(CO)3 P-Ph PhP Ph H2 Ph OEU-
Give the IUPAC name for the following complexes: (i) (ii) Fe. OC- CO C OEC CEO Tc OC OEC (ii) CH3 (iv) CH3 H3C CH CH3 As (OC),Os CH3 CH3 H3C Zr (OC),Os Os(CO)3 P-Ph PhP Ph H2 Ph OEU-
Chapter26: Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 26.30QAP
Related questions
Question
Please give answer first three with explanation
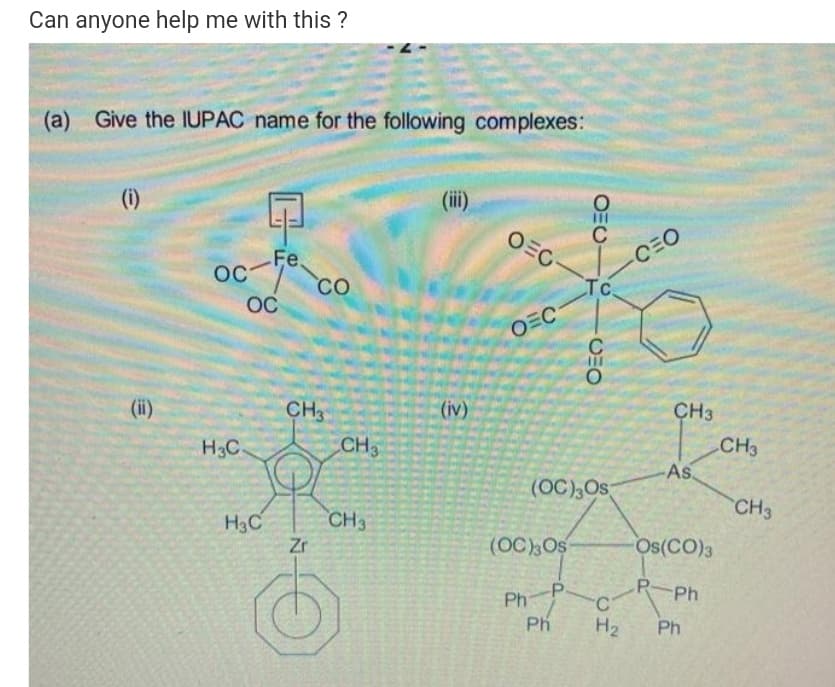
Transcribed Image Text:Can anyone help me with this ?
(a) Give the IUPAC name for the following complexes:
(i)
(ii)
OEC-
C
oc
Fe.
OC
CO
CEO
Tc
OC
OEC
(ii)
CH
(iv)
CH3
H3C
CH3
CH3
As.
(OC),Os
CH3
CH3
H3C
Zr
(OC),Os
Os(CO)3
-P
P-Ph
Ph
Ph
C'
H2
Ph
OEU
CHO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

