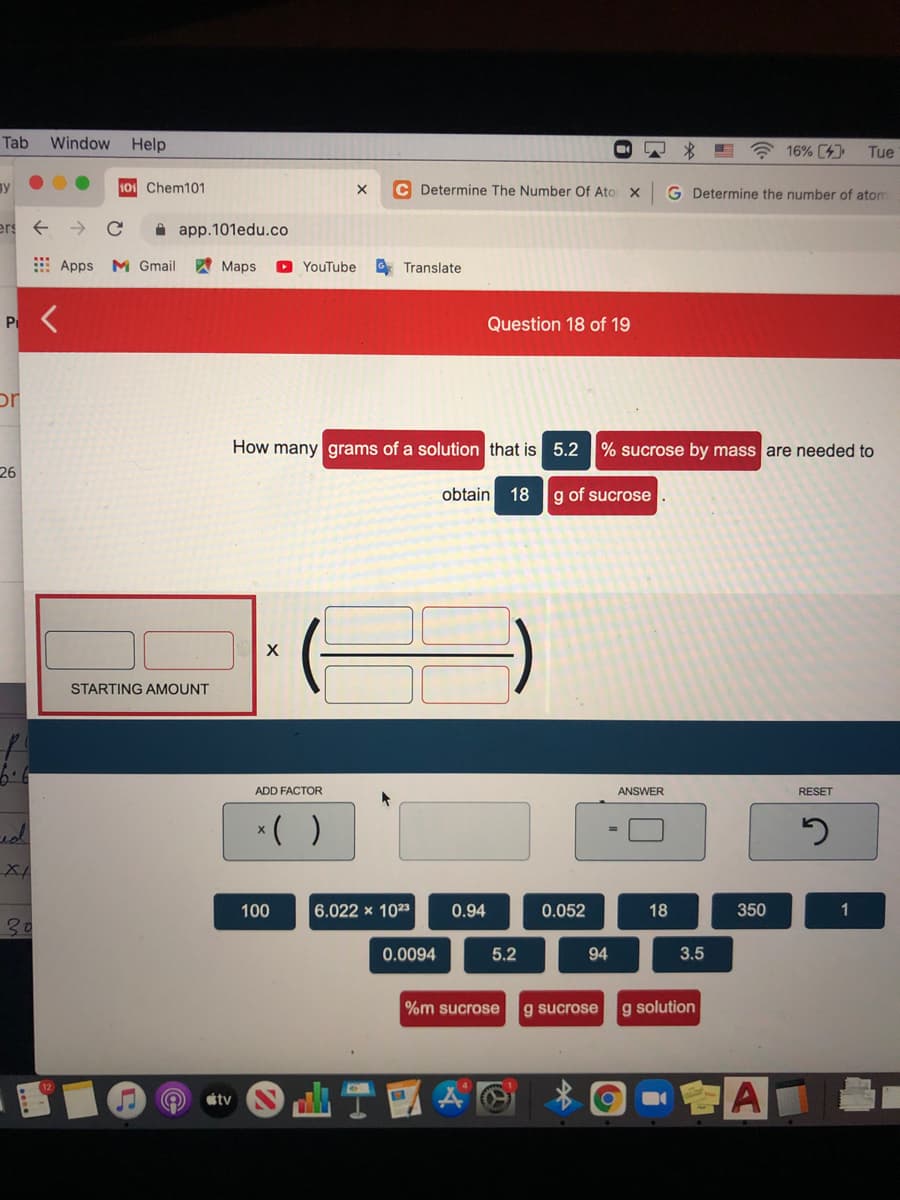
How many grams of a solution that is 5.2 % sucrose by mass are needed to obtain 18 g of sucrose STARTING AMOUNT ADD FACTOR ANSWER RESET *( ) 100 6.022 x 1023 0.94 0.052 18 350 1 0.0094 5.2 94 3.5 %m sucrose g sucrose g solution
How many grams of a solution that is 5.2 % sucrose by mass are needed to obtain 18 g of sucrose STARTING AMOUNT ADD FACTOR ANSWER RESET *( ) 100 6.022 x 1023 0.94 0.052 18 350 1 0.0094 5.2 94 3.5 %m sucrose g sucrose g solution
Chapter13: Isolation Of Eugenol From Clov
Section: Chapter Questions
Problem 9Q
Related questions
Question
Please help fill in the blanks
Transcribed Image Text:Tab
Window
Help
A 16% C4)
Tue
ay
101 Chem101
C Determine The Number Of Ato
G Determine the number of atom
ers
A app.101edu.co
E Apps M Gmail
A Maps
O YouTube
O Translate
P
Question 18 of 19
or
How many grams of a solution that is 5.2 % sucrose by mass are needed to
26
obtain
18
g of sucrose
STARTING AMOUNT
6.6
ADD FACTOR
ANSWER
RESET
*( )
100
6.022 x 1023
0.94
0.052
18
350
30
0.0094
5.2
94
3.5
%m sucrose
g sucrose
g solution
會A
tv
…个
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co