Q: you kave ani Kal ion that has been ode Posited in the Porm of Ni(DMG) and in the form of NiS S…
A:
Q: What are the different tetrabasic forms of EDTA? b. Describe their individual reactivities
A: We have to describe the different tetrabasic forms of ETDA and their individual reactivities.
Q: 1. What is the molar concentration of a sulfuric acid (H,SO.) solution with 7.3g/dm' concentration?
A: Given, Concentration of the solution = 7.3 g/dm3 Molar concentration of sulphuric acid solution = ?…
Q: 1. Heterolysis `Br 2. Coordination involving NH3 3. Proton transfer involving H,O `NH2 2N° HO (a)…
A: Heterolysis happens when atoms have difference in electronegativity then one atom will gain positive…
Q: alaced in w nartial pre
A: 04. (a) True statement When sugar is added, the sugar molecules dissolve into the water. The causes…
Q: How many grems of AgCl cen dissolue in 2.8L ofa LiMNH Selulien ? CMW Agl = 143, 32 almol, Accl Kap =…
A: Given : Concentration of NH3 = 1.5 M Volume of solution = 2.0 L
Q: (a) (b) or Br₂ (1 equivalent) Page 3 of 20
A: Requirement from question: Total four questions and required products.
Q: rxn reaction So,Cl, (9)+ 2H,0(1) →H,SO,(1)+2HCI(g) AH° = -6 Calculate the AH', of HCI(g) in kJ/mol.…
A:
Q: но PhenoFluor, TMS-Imd он е. DEAD, PPh; H3C- -OH NCI он f. он Cu(CH3CN);OTf, TEMPO, Ph но, air, NMI,…
A: d. Inversion at chiral center (see below). e. Mitsnobu reaction (here p-methyl phenol is acting as…
Q: the 18e rule find frost now Aeuming doonutin ometal (M) parf en d Sketch the complex M(e0) (<zMy) (P…
A:
Q: For [PTCI(dien)]* (s = 0.65), the rate of substitution with I' is much greater than that with Br. *…
A: yes, the rate of substitution with I- is much greater than Br- . 1)the Pt2+ metal is soft metal . in…
Q: Br 17.
A: Solution
Q: Calculate the percent dissociation of trimethylacetic acid (C,H,CO,H) in a 0.52 mM aqueous solution…
A:
Q: A lUrouu ion a stable intermediate? If the reaction conditions were acidic , suggest the po A llkov1
A:
Q: What is the concentration of free Ag+(aq) in a solution that is initially 0.01254 M in AgNO3 and…
A: · Concentration of AgNO3 = 0.01254 M· Concentration of NH3 = 1.04842 M· Kf of AgNH32+ = 1.600×107
Q: (d) (е) HCI - 80°C
A: d) Alkenes reacts with alcohols in presence of strong acid like H2SO4, to form an Ether.\ Thus, the…
Q: 4. For CH24 Oor H2O: a. Which is on top? Why? b. Where does l; and KMNO, dissolve? Why? Type here to…
A:
Q: In the experiment, potassium permanganate is used to oxidize 2-chlorotoluene (o-chlorotoluene) to…
A: We have find out mechanism of oxidation of aromatic methyl group by KMnO4.
Q: the order of stabilitt opcarbocatin prarbocativ is 22 PrimartS Feriarts secendary a) b) sprimard…
A: Carbon atom has 4 valence electrons, therefore it is capable of form 4 bonds. When the carbon has…
Q: Part A The value of AS° for the catalytic hydrogenation of acetylene to ethene, C2H2 (g) + H2 (g) →…
A:
Q: The pK1 for germanic acid, H,GEO3, is 9.0. Account for the increase in pK,1 of germanic acid…
A: It is considered likely that germanium is present in the thermal waters in the form of Germanic…
Q: if what is the eguili m conceration of free Fe 24 Concentrdton of Fezt is for Fe[NHg]?t is 472×1014…
A: We have to predict the equilibrium concentration of the ferrous ion.
Q: that apply) Mg?+ PO? Mg?+ PO- O H O H Mg2+ C-Ċ-Ċ–H Mg2+ C=C-C-H H OH нон H-N-H H-N-H но. Lys345 H.…
A: Metal ion catalysis acid catalysis base catalysis Electrostatic catalysis electron sink…
Q: 2) OH (е) ationoy (d) (f)
A:
Q: Predict the solubility and explain why: a) CdCO3 b) Na2S c) PbSO4 d) (NH4)3PO4 e) Hg2Cl2
A: The maximum amount of solute that can be dissolved in the definite amount of solvent at a fixed…
Q: The two structures for Iron - penta - carbonyl: Ço "Fe" oc co oc-FeCO co čo (A) (B) .Structure A has…
A: IR spectroscopy is also referred to as vibrational spectroscopy used for identifying the functional…
Q: New Tab Metallic tantal C Metallic Tants C The Subitance C The Subitanci C The Subitarice C Wich Of…
A: In chemical kinetics, the rate of the reaction (the speed at which it occurs) is determined by the…
Q: Calculate the lutiopy malest enbiespychaye sbnd allowed to kflom. a peuesive an idedl aue…
A: Entropy is the randomness or disorderness in a system. Higher the randomness, higher the entropy.…
Q: [deprotonation of triple bond] [rka 25] 0 & 0 t NHs [vka 38] CHs by &
A:
Q: ОН 1. TBSCI а. imidazole, CH2Cl2 A DMF, -18 °C to rt 2. NalO4, CH2Cl2 80% НО OH large excess
A: We have to give the product of the given reaction. We know that the TBSCl is used to protect the…
Q: Acetic acid (CH3COOH, abbrev. HA) is a very common weak acid with pKA = 4.75 (at 25oC and low ionic…
A:
Q: प्खर जो०त ककवे टागल्वेर क्रेमए pocodurd with mechanis mA 2504 major 2. - १entamos fhरSoप
A:
Q: An oogone hoving MF Corompound Tell ens eeduces lo dutien and The - lo」子リ5444 41 and 29 at…
A: Given: The organic compound can reduce tollen's reagent as well as fehling's solution, this implies…
Q: Cu(OAc)2 (10 mol%) NazS203 (2 equiv.) CO2ET + KS OEt DMSO, 110 °c Ph H3C S=0--1---I H3C %3D Cu(OAc)2…
A: The domino reaction or cascade reaction involves the intermediate that produces by the combination…
Q: Draw the organic products formed in each reaction.
A:
Q: CHEM 321 Addifional Prsblems for Exam4 : aculale eS after 30. 00mk of o.02000M EDTH s added to…
A: Given, Volume of EDTA = 30 mL Concentration of EDTA = 0.02000 M Volume of Sr2+ solution = 50 mL…
Q: In a complex like IrPh(CO) (PMe3)2. the v(CO) frequency would be - performing oxidative-addition…
A:
Q: solubility in: compounds H2O Et20 5% NaOH 5% NaHCO3 5% HC1 Conc. H2SO4 comments 1. CH3CO2H; acetic…
A: Hello. Since your question has multiple subparts, the first three subparts shall only be solved in…
Q: In a 0.20 mM aqueous solution of trimethylacetic acid (CH,CO,H), what is the percentage of…
A: Given : Concentration of C4H9CO2H = 0.20 mM = 2.0 X 10-4 M…
Q: deltaHf
A:
Q: + AIATS For Two Year Medic... 90 /180 (02:53 hr miny Mark for Review HC = CH red hot Fe tube 873K…
A: When acetylene gas is passed through red hot iron tube, it results in the formation of benzene…
Q: XIDIZED Cr+ + Sn4+ 72+ our answer
A: The given reaction, Cr + Sn4+ -----> Cr3+ + Sn2+ Half reaction splitting Cr ----> Cr3+ + 3e…
Q: hohat stoichiom etuic and intimati are mechanisms for heautrion 4 substitution complises ype metal…
A: The mechanisms for the substitution reaction has two types of mechanisms. A substitution reaction is…
Q: diluted H2SO4 "ОН ОН checkpoint-08v02-toc-ay21-22fa.pdf (page 18 of 18) OH OH 'OH "OH racemic…
A: The first reaction occurs via acid hydrolysis reaction in which addition of H-OH takes place while…
Q: कना १०मaडडापल श्ुल्कांवेर ( EcN ) vrल्वलङ्ड ज्क वतंवेe , व वटववाय P० 3cnu&4& भp8, सुdसमा पूुकलांवर…
A: Amount of KCN= 0.410 gram KCN(aq)+HCL(aq) ->HCN(g) + KCl(aq)
Q: Compound Addition of FeCl3 Observations Dark ben on top botton Separation of both colors Phenol…
A: ASPIRIN: Aspirin is also known as acetylsalicylic acid. The molecular formula of aspirin is C9H8O4.…
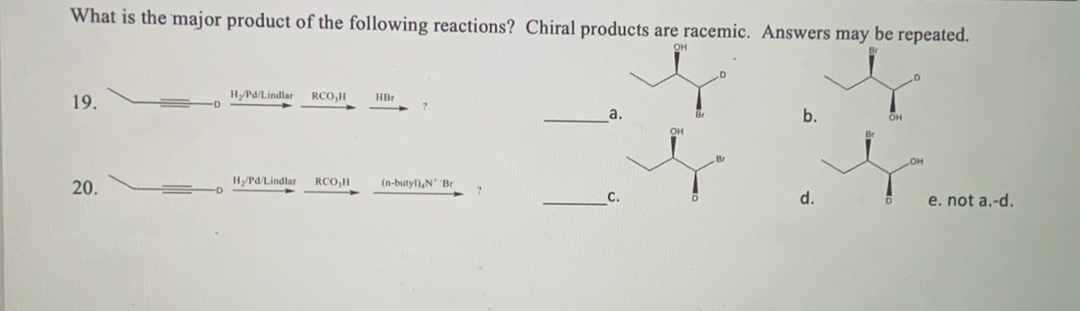
Step by step
Solved in 2 steps with 2 images

- The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?Please fill out for each reaction i.Fill in the missing starting materials, products, or reagents as necessary.If no reaction occurs, write "N.R." and explain why this is the case. ii. Label each transformation as SN1, SN2, or acid/base. iii. Indicate if the product is racemic or a single enantiomer.SN1 is always a competing reaction of E1. True or False?
- Draw the products of the SN1 reaction below and use wedge-and-dash bonds to indicate the stereochemistry of any stereogenic centers.i. Fill in the missing starting materials, products, or reagents as necessary. ii. Label each transformation as SN1, SN2, or acid/base. iii. Indicate if the product is racemic or a single enantiomerThe use of curved arrows is a powerful tool that illustrates even complex reactions. a.Add curved arrows to show how carbocation A is converted to carbocation B. Label each new σ bond formed. Similar reactions have been used in elegant syntheses of steroids. b.Draw the product by following the curved arrows. This reaction is an example of a [3,3] sigmatropic rearrangement, as we will learn in Chapter 25.
- What are the products from the following reactions? Please help with part e,f,gArrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, IQ4 Which statement below about Sn1 reactions is incorrect? (A) SN1 reactions are stepwise and have intermediates. (B) The slow step in a SN1 reaction is formation of the carbocation intermediate. (C) SN1 reactions have first order kinetics which means only the alkyl halide is involved in the rate limiting step. (D) The products of a SN1 reaction will be a pair of enantiomers. (E) An aprotic solvent is best for Sn1 reactions as they tend to help stabilize carbocation intermediates.
- Draw the reaction products for each SN1 reaction and indicate the appropriate stereochemistry.Draw the SNl or SN2 reaction with CN for the following molecules t butylchloride , n - butylbromide , 2 -bromobutane . Explain in which mechanism it will react and why ?Based on your knowledge in biological chemistry reaction in biomolecules, what do you think is the best type and product for the above reaction? a. SN1 b. SN2 c. E1 d. E2 e. None of the above



