
Concept explainers
In one experiment, 12.0 g of PCl5 was slowly added to 15.0 g of H2O according to the following balanced chemical equation:
Pcl5(s)+4H2O(l) ---> H3PO4 (aq) + 5HCl (g)
The molar masses for each compounds in the equation are as follows;
PCl5: 208.224 g/mol
H2O: 18.015 g/mol
H3PO4: 97.994 g/mol
HCl; 36.45 g/mol
What is the limiting reagent in this scenario?
Note! Make an accurate claim: The limiting reagent in this scenario is _____. Provide additional details and use subject specific language.
Cite evidence from what's given to you in the problem that supports your answer, hint: look at that equation!. Lastly! Fully connect the evidence to the claim/your answer to the question. Include subject specific language in your reasoning.
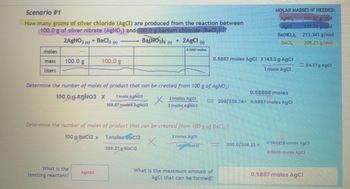
Your written response should LOOK exactly like the response attached. The other image is there to show how you would figure out the limiting reagent.
Trending nowThis is a popular solution!
Step by stepSolved in 5 steps

- The fizz produced when an Alka-Seltzer tablet is dissolvedin water is due to the reaction between sodium bicarbonate(NaHCO3) and citric acid (H3C6H5O72)3 NaHCO3(aq) + H3C6H5O7(aq)-------->3 CO2(g) + 3H2O(l) + Na3C6H5O7(aq) In a certain experiment 1.00 g of sodium bicarbonate and1.00 g of citric acid are allowed to react. The actual yield of CO2 was 0.436 g. a.) What is the theoretical yield of CO2? b.) What is the % yield?arrow_forwardIf 50.0 g of water is combined with 155.3 g of PCl3, PCl3(l) + 3H2O (l) --> H3PO4 (aq) + 3HCl (aq) what is the limiting reagent?arrow_forwardBalance the following reaction. If a reactant or product is not present, put a zero (0) in the blank. H+(aq) + H2O(l) + ClO-13(aq) + e-1 <--> Cl-1(g) + H+(aq) + H2O(l)arrow_forward
- 2+ Natural waters often contain relatively high levels of calcium ion, Ca²+, and hydrogen carbonate ion (bicarbonate), HCO3¯, from the leaching of minerals into the water. When such water is used commercially or in the home, heating of the water leads to the formation of solid calcium carbonate, CaCO3, which forms a deposit ("scale") on the interior of boilers, pipes, and other plumbing fixtures. Ca(HCO3)2 (aq) → CaCO3 (s) + CO2(g) + H₂O(1) If a sample of well water contains 0.0016 mg of Ca(HCO3)2 per milliliter, what mass of CaCO3 scale would 1.0 mL of this water be capable of depositing? Mass= garrow_forwardArsine, AsH3, was once a product of a test used in forensic analysis. Tissues from a possible poisoning victim were treated with zinc and sulfuric acid. If arsenic was present (usually as diarsenic trioxide, As203), arsine would form: As₂03 (s) + 6 Zn(s) + 6 H₂SO4(aq) ----> 2 AsH3(g) + 6 ZnSO4(aq) + 3 H₂O(1) a) In a lab exercise, a forensic technician reacts 19.8 g of AS₂O3 with 32.7 g of Zinc in the presence of excess sulfuric acid. What mass of AsH3 will be produced? b) Use the same chemical reaction, create a question which needs to be solved by percentage yield.arrow_forward???arrow_forward
- 16) According to the balanced chemical equation 5 H2C20 4(aq) + 2 MnO4-(aq) + 6 H+(aq) →10 CO 2(g) + 2 Mn2+(aq) + 8 H2O() 0.3500 grams of oxalic acid, H2C2O4 will react with_ _moles of permanganate, MnO4". A) 0.009718 B) 0.001554 C) 0.003887 D) 0.007774arrow_forwardDteremine the limiting reaganet (SHOW THE calculations for determinig the limiting reagent ) and calculate the theoretical mass of PbI2 by using limiting reagent. show your calculations. Balanced Equation: Pb(NO3)2 (aq) + 2KCL(aq) ---> 2KNO3(aq) + PbI2(s) No of moles of Pb(NO3)2 = 0.003 No of moles of KI = 0.007arrow_forwardThe reaction for producing glucose in plants, called photosynthesis, is 6CO2+6H2O−→−−lightC6H12O6+6O2 6CO2+6H2O→lightC6H12O6+6O2 If a plant produces 7.56 mol C6H12O6,7.56 mol C6H12O6, how many moles of CO2CO2 are needed?arrow_forward
- Assuming you started with 3.23 g of sodium bicarbonate and your actual yield of solid product is 1.20g, which reaction is more likely to be the correct one?arrow_forward4.106. Aspirin (acetlsalicyclic acid) may be formed from salicyclic acid and acetic acid as follows:C7H6O3 (aq) + CH3COOH (aq) → C9H8O4(s) + H2O(l)Salicyclic acid Acetic Acid Aspirin b. How many mol of aspirin may be produced from 1.00*102 mol mol salicylic acid? c. How many g of aspirin may be produced from 1.00*102 mol salicylic acid? d. How many g of acetic acid would be required to completely with the 1.00*102 mol salicylic acid? e. For the conditions in part (d), how many g of aspirin would form?arrow_forwardIn a particular experiment 0.0039 moles of AgNO3 is added to 0.001875 NH4Cl and 0.156g of AgCl are recovered. What is the percent yield? AgNO3(aq) + NH4Cl(aq) --> AgCl(s) + NH4NO3(aq)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





