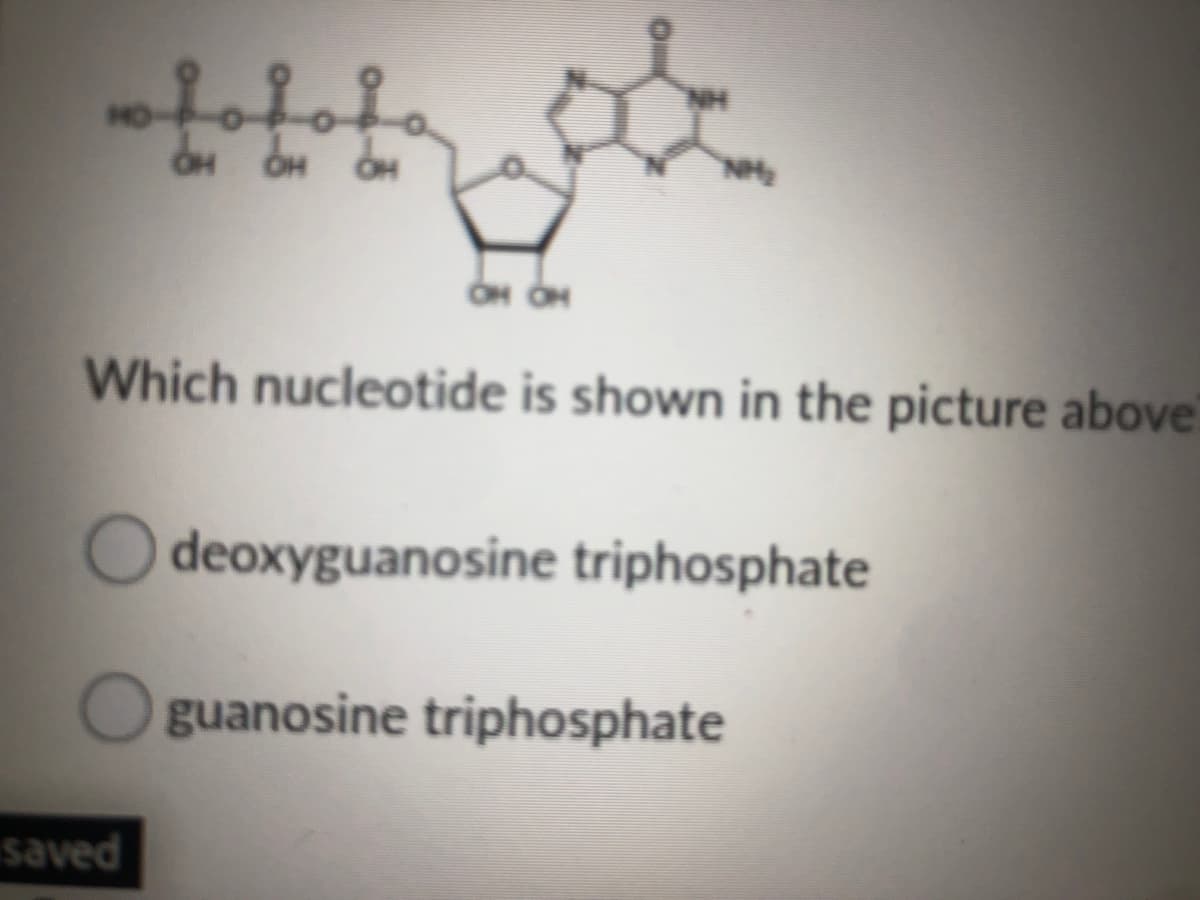
NH OH OH Which nucleotide is shown in the picture
Q: 1 0.8 0.6 Mutant y 0.4 Wild-Type 0.2 40 60 80 [ligand] 20
A: According to the given information, the wild type protein structure has aspartate in its primary str...
Q: Prepare 5 liters of a 0.2 M dextrose solution using 5 liters of 5 M dextrose.
A: The equation used to make dilute solutions from the concentrated stock solution is as follows, C1V1=...
Q: Draw the product of the reaction below. (Upload your answer here) H- но ? NaBH4 HO -H H- ČH2OH
A: Given structure :- D-GALACTOSE (monosaccharide) In presence of NaBH4 , REDUCTION reaction oc...
Q: Evaluate the special traits of microbes in bioremediation of oil spill and their mechanism in degrad...
A: Process of removal of hramful/toxic substance using microbes or plant is called Bioremediation. Such...
Q: 1. What is the name we give to structures which are not complete polypeptide chains but form superse...
A: "Since you have asked multiple questions, we will solve the first four questions for you. If you wan...
Q: b. What is happening in panel B? You are looking at the southern blot image. What is the radioactive...
A: This is an experimental data where experimenter is trying to look on function of PHT4;2 gene allele....
Q: If a liver extract capable of carrying out normal metabolic reactions (including gluconeogenesis) is...
A: Gluconeogenesis is the metabolic route that leads to glucose production from non-carbohydrate carbon...
Q: How to use the sterile filter? Show the different sizes and specifications of sterile filters.
A: Sterilization is the method that is used to eliminate all forms of life and biological agents presen...
Q: Below is the primary sequence of a viral protein. MSVVNTEIKFPTHLRSGDFAIIDGMVVEVTSVEYKPVEQAVYLKYRYHL...
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen but there are other element...
Q: Given the following question on the image identify the following:1. Total number of acetyl coA molec...
A: Maltose is a carbohydrate and it gets oxidized through glycolysis in cytoplasm and through the TCA c...
Q: What risks are involved in genetic engineering of crop plants? How do these risks compare with other...
A: Genetic engineering is a technique where the genes are edited or newly genes are inserted using gene...
Q: Why do you think DNA is the genetic material used by eukaryotes instead of RNA?
A: Deoxyribonucleic acid (DNA): Nucleic acids are molecules that store hereditary information for perf...
Q: Name the experiment shown above and briefly describe how it is set up as well as the role of each co...
A: Gel electrophoresis is a method of separation of protein (SDS-PAGE, polyacrylamide gel electrophores...
Q: ENDOSYMBIOTIC THEORY application to this subject (BIOCHEMISTRY)?
A: It is believed that life arrived on earth some four billion years ago and the first cells were proka...
Q: Compound A below is a key intermediate in the synthesis of keto-myo-inositol B. Suggest a synthetic ...
A: Here compound C is D-Glucose D-Glucose is converted into Glucose-6-Phosphate in presence of Hexokina...
Q: The extracellular sodium [Na+]0 is reduced in the saline bath. Following another current injection ...
A: Polar large size molecules (glucose, amino acids) and Ions (Na+, H+,k+, Ca+) can't pass...
Q: Convert the hydrogen ion concentration (moles per liter) of a solution to a pH value and describe ho...
A: Suppose a solution has a hydrogen ion concentration of 20 mM. One needs to calculate the pH of the g...
Q: 6. Celiac disease is a disorder of the small intestine characterized by autoimmune response to glute...
A: Gluten protein is commonly found in many cereals like Wheat, barley, rye etc and is difficult to dig...
Q: Regulation of glycolysis pathway involves. . Allosteric inhibition by ATP Allosteric stimulation by ...
A: The important regulatory enzyme of Glycolysis is PFK-1 and at this stage one ATP is generated. In ca...
Q: Complete the following paragraph describing the fate of the second 14CO-acetyl-CoA molecule incorpor...
A: The glyoxylate produced in the isocitrate reaction combines with 2nd molecule of acetyl coa in the m...
Q: Which of the following vitamins that commonly activates more different enzymes? *
A: Vitamins are organic molecules and it is a vital micronutrient that is required in small quantities ...
Q: kinetically perfect enzymes a) Using the Gibbs energy formula, explain why engergonic reactions can ...
A: Most enzymes follow Michaelis -Menten (MM)kinetics and hence obeys the MM equilibrium assumption tha...
Q: All of the following are considered weak interactions in proteins, except:
A: Proteins are the macromolecules composed of amino acids bound together by peptide bond between amino...
Q: fof NH HO-P-O-P-O-F `NH2 ÓH ÓH OH OH OH Which nucleotide is shown in the picture above?
A: A nucleotide is an organic molecule containing a nucleoside and a phosphate. It is the basic buildin...
Q: How do you prepare 42:1 chloroform/isoamyl alcohol 70% ethanol?
A: Preparation of 42:1 Chloroform / Isoamyl alcohol Add 42ml of Chloroform and later add 1ml of Isoamy...
Q: What three components does a nucleotide consist of?
A: Nucleotides are the basic building block of nucleic acids. DNA and RNA are polymers of long chains o...
Q: Which of the following peptides will likely adopt an alpha helix structure? (idk) A-D-I-E-L-Y-F-H-I...
A: α-helix is a secondary structure of protein in which the polypeptide chain twists to form a helical ...
Q: Why can't we store Vitamin C in our bodies?
A: Vitamin C- Ascorbic acid It is vital for tissue repair , collagen synthesis and it's deficiency- cau...
Q: 1mL of the stock solution acetaminophen (1mg/mL) is used for the estimation of PC using n-octanol. A...
A: Acetaminophen is also commonly known as a Paracetamol that is a medicine used to treat the fever and...
Q: Some of the test(s) you tried from this virtual lab are important basis for determining glucose leve...
A: The sugar present in the blood or urine sample is glucose.
Q: 26. Dietary triglycerides are hydrolyzed by pancreatic lipase, which is related to but distinct from...
A: The pancreas is a heterocrine gland because it secretes digestive juices and hormones. Pancreatic am...
Q: Task 1: DNA Extraction To begin work on the bacterium, you begin by extracting its genomic DNA (gDNA...
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: Flame Test (Unknown) View the Unknowns to see the results of the flame test. Record Your Observation...
A: As per our honor code, we are not allowed to give you a handwritten solution, however here is the an...
Q: The OH group at which carbon atom is involved in ring formation in aldohexose? *
A: Monosaccharides are represented as linear molecules but many also adopt cyclic structures and this ...
Q: Define triad
A: Those organic molecules are mainly comprised of two functional groups; an amino group and a carboxyl...
Q: Explain how the carbonate-bicarbonate buffer system works in balancing acid-base in the blood.
A: Buffers are solutions that have weak acid and its conjugate base. They nullify small changes in the...
Q: Propose TWO (2) industrially important bacterial products for human needs with their respective proc...
A: Bacterial products been proven to be very beneficial for several industrial processes in manufacturi...
Q: Which of the following cells is solely dependent on glucose as energy source? Muscle cells Kidney ce...
A: Inside the body, varying energy sources get exploited by different cells of various organs and tissu...
Q: What are functions of chromosomes?
A: Nucleic acids are macromolecules comprised of carbon, hydrogen, oxygen, nitrogen, and phosphorus. Th...
Q: If a given double stranded DNA undergoes enzymatic hydrolysis targeting only the "a" side in the pho...
A: Here in question, I believe “a” side in Phosphodiester bond refer to the side bond to 3’-C atom of t...
Q: A hypothetical electron-transport chain contains an electron donor (1), an electron carrier (2), and...
A: All biological reactions that take place in a cell are referred to as the metabolism. It is divided ...
Q: A conjugate was incubated with a mixture of lysosomal enzymes and no drug release was observed in th...
A: Lysosomes contain a wide variety of hydrolytic enzymes including proteases, nucleases, glycosidases,...
Q: A protein with a positive charge at physiologic pH (7.4) most likely contains amino acids with Basic...
A: Protein is essential for the body. Without proteins, the organs and functions of the body cannot su...
Q: The ionizable side change (-OH) of a synthetic amino acid has a pKa of 6.54. Find the amino acid's i...
A: The pH at which the amino acid has no net charge and is electrically neutral is called isoelectric p...
Q: 1. A ligand binds more tightly to the folded state (N) of a protein than to the unfolded state (U). ...
A: "Since you have asked multiple questions, we will solve the first question for you. If you want a sp...
Q: Create 5 water molecules with appropriate bond formation. You must Include images of the water molec...
A: The main constituent of Earth's hydrosphere is water and all Known living organisms' fluid ...
Q: Suppose a mutant Fatty Acid synthase only synthesizes a FA 14 Carbons long. How many ATP will the sy...
A: Acetyl CoA is a hybrid molecule that is involved in a variety of metabolic processes in the body. As...
Q: Your supervisor asks you to carry out an in vitro transcription reaction. Unfortunately, they are ca...
A: The genome of an organism is composed of DNA. The DNA is replicated during the time of cell division...
Q: How are blood glucose concentration affected by heavy alcohol consumption? Please explain with extre...
A: Alcohol plays major role on health. Alcohol can effect every organ of the body. In an average person...
Q: A solution is prepared by dissolving 13.5 g of glucose in 0.100 kg of water. What is the mass percen...
A: The mass percentage is one way of representing the concentration of an element or a compound in a mi...
Step by step
Solved in 2 steps with 1 images

- Using Figures 8.7 and 8.9 as a guide, draw a dinucleotide composed of C and A. Next to this, draw the complementary dinucleotide in an antiparallel fashion. Connect the dinucleotides with the appropriate hydrogen bonds. FIGURE 8.9 The two polynucleotide chains in DNA run in opposite directions. The left strand runs 5 to 3, and the right strand runs 3 to 5. The base sequences in each strand are complementary. An A in one strand pairs with a T in the other strand, and a C in one strand is paired with a G in the opposite strand. FIGURE 8.7 Nucleotides can be joined together to form chains caled polynucleotides. Polynucleotides are polar molecules with a 5 end (at the phosphate group) and a 3 end (at the sugar group). An RNA polynucleotide is shown at the left, and a DNA polynucleotide is shown at the right.Draw the dinucleotide AT and label the following: 5’ and 3’ ends Phosphodiester bond N-glycosidic bondsThe best definition of an endonuclease is that it hydrolyzes A. nucleotide from only the 3' -end of an oligonucleotide . B. nucleotide from either terminal of an oligonucleotide. C. phosphodiester bond located in the interior of a polynucleotide . D. bond only in a specific sequence of nucleotides. E. bond that is distal co the base chat occupies the 5' position of the bond
- Draw the dinucleotide CU. Show structures of the basesWhen cytosine is treated with bisulfite, the amino group is replaced with a carbonyl group. Identify the resulting base.Draw the strucures of Adenosine Triphosphate (ATP) and Cytidine Triphosphate and Show how they combine to form the polynuleotide (i.e. only the end product). Show at any one of these structures where the glycosidic bond occurs
- Which of the followinga.B-D-riboseb. thyminec. phosphatewhich is not a component of deoxyribonucleotide?The formation of deoxyribonucleotide from ribonucleotide requires all the following except: a.Nucleoside diphosphate (NDP) b. Nucleoside triphosphate (NTP) c.Thioredoxin d. Ribonucleotide reductaseGive me nucleotide sequence with pairing. Like this ATC TCA TGA GCC TAG AGT ACT. CGG
- Translate the following opposite strand of DNA based on the nucleotide bases. ATTGACTGGDraw the following oligonucleotides: a. d(pAACGTCTC) b. pGUAACUGCIn the image attached, there are 4 nitrogenous bases plus ribose used to generate strands of DNA. They are oriented with hydrogen bonding regions aligned. Diagram the enol form of cytosine following a tautomeric shift. Include in the diagram how this change shifts hydrogen bonding characteristics and a new binding partner for this nucleotide.

