Q: Acetophenone is soluble in diethyl ether, isopropanol, and nhexane. Identify and justify the most…
A: The selection of the solvent for the UV-Vis characterization based on their UV cutoffs. The UV…
Q: The graph of temperature-component mole fraction for Benzene-Ethanol binary system is given in the…
A: Simple distillation is a technique of purification/ separation of liquids in which a liquid is…
Q: c) You decide to use ethanol in conjunction with another solvent to make a solvent pair. What two…
A: CRYSTALLIZATION: A process in which solid crystals are produced from other state of matter like…
Q: Given that a mixture of nitric acid (bp 86 °C) and water forms a maximum-boiling-point azeotrope…
A: Solution. Given That mixture of nitric acid = bp 86 °C…
Q: When performing a recrystallization of an impure solid, why is it important to allow the heated…
A: Recrystallisation is one of the most used technique in chemistry to obtain the pure compound from an…
Q: Construct an approximate boiling-point-composition diagram for a benzene– methanol system. The…
A: A benzene -methanol solution is an example of a minimum -boiling azeotrope. In the case of…
Q: For the liquid acetone, list all IMFs that have to be broken in order for it to mix with water
A: Solubility depends upon the polarity of the solvent. A polar solvent will be dissolved in the polar…
Q: In a steam distillation, the amount of water actually distilled is usually greater than the amount…
A:
Q: 1 g of organic matter dissolved 20 times more in ether than water is dissolved in 500 cm3 of water.…
A: Components are partitioned between an aqueous layer and the organic layer and at equilibrium, the…
Q: COMPARE the: a. phase diagram of eutectic point between binary system of menthol and camphor and…
A: The given theoretical diagram is:
Q: Consider the following two white solids below and their physical properties based on information…
A: From the given information we get the answer
Q: At some point, it can be get Eutectic point on two compounds (A and B) in the phase diagram?
A: In an equilibrium between phases in a multi-phase, multi-component system where all the phases are…
Q: measurement of mass and mole fraction
A:
Q: A. Provide an example of a boiling point composition curve and explain in simple words. Five…
A: In liquids, the interaction among the constituent particles is much weaker than the interaction in…
Q: A mixture of organic solvent (nitrobenzene) and water boils at 363K. The vapour pressure of water at…
A:
Q: Use a temperature-composition diagram to explain why fractional distillation of two components is…
A:
Q: Beer's law why does the procedure for measuring the concentration of a solution photometrically…
A: For the calibration of spectrophotometer blank solution is needed.
Q: Uranium tetrafluoride and zirconium tetrafluoride melt at 1035 °C and 912 °C respectively. They form…
A: The melting points of uranium tetrafluoride and zirconium tetrafluoride are given as 1035°C and…
Q: . After the reflux period, what substances are pre n Whu will the density of the organic phase be…
A: In organic chemistry, Reactants are mixed in a solvent media, and products are obtained. The final…
Q: - What is the ideal solvent behavior for crystallization? - You will probably hear me say…
A: Crystallization is defined as the process in which atoms in a given sample are arranged into a…
Q: Research the Eutectic point that is present in phase diagrams. How will you explain its importance…
A: Eutectic point is the composition at which a binary mixture of two components freezes at the lowest…
Q: Does your melting point obtained for your product indicate that your sample is indeed benzoin or…
A: By matching the literature and obtained value one can consider the product. The literature value of…
Q: explain why the boiling point of a two component mixture rises slowly throughout a simple…
A: A temperature is not absolute. When you measure a temperature, you are measuring a distribution of…
Q: What are the reasons why there are minimum and maximum azeotrope? How does this relate to the…
A: Minimum azeotrope is a solution whose boiling point is lower than the boiling point of the boiling…
Q: A student trying to determine if a white solid is a true hydrate heats the sample and finds that…
A: White solid is heated and it resulted in evolution of H2O. The anhydrous residue obtained waa…
Q: In order to obtain accurate data it is important to add hear slowly and steadily when the…
A: In order to obtain accurate data it is important to add heat slowly and steadily when the…
Q: Basis for recrystallization Nernst Distribution Law differences in solubilities of a solid and an…
A:
Q: A sample of naphthalene is contamined with a small amount of anthracene. The melting point would be…
A: Melting point the temperature where the solid starts to melt. Melting point is measure in oC or oF.…
Q: assuming that you distill a mixture of two liquids that initially contains 80% of a and 20% of b…
A: The phase diagram given is,
Q: When should we stop collecting distillate in ethanol-alcohol simple distillation process? Give me…
A: We are find out when to stop collecting distillate in ethanol distillation process.
Q: Give clear handwritten solution- What is the normal boiling point of this compound? (Using phase…
A: Before solving this question, in the phase diagram assignment of the phases and equilibrium curves…
Q: 3. What are su melting point using a capillary and a Thiele tube containing 4. Is it possible for a…
A:
Q: Which statement is incorrect concerning these two molecules with the same formula, C,H,O? H. 1. H. H…
A: Boiling point:The temperature at which the vapor pressure of the liquid becomes equal to the…
Q: 3. Draw (illustrate) a hypothetical phase diagram incorporating the following salient features: A.…
A: Since you have posted multiple questions, we have answered the first one for you. To get the…
Q: Graph of temperature versus fraction number for the distilled liquid and show the extrapolation of…
A: So, here, we need to plot the graph of temperature versus fraction number.
Q: An azeotropic mixture can be separated by fractional For azeotropic mixtures, the liquid and vapour…
A: Azeotropic mixtures are the ones that have same composition in liquid as well as in vapour phase and…
Q: 4. A 1:1 mixture of ethyl acetate and ethyl butyrate can be efficiently separated by careful…
A: #Q.4: We have to separate a 1:1 mixture of ethyl acetate and ethyl butyrate. The boiling point of…
Q: atch the following: Freezing point depression The effect of supercooling Freezing point of pure…
A: Let us discuss the phase diagram first and then find out the respective points.
Q: Is a mobile phase composition 40:60 of acetone:hexane more polar than a mobile phase composition of…
A:
Q: A student trying to determine if a white solid is a true hydrate heats the sample and finds that…
A: The explanation is about true hydrate or not given below.
Q: Fill in the blanks: a) The component of the mixture that becomes a water soluble salt by treating…
A: Solubility is the property of dissolution of a solute in a solvent to form a solution.
Q: What is the molar absorptivity of copper at λmax of copper?
A: The data given is,
Q: One of the components of nutmeg is an organic compound, Trimiristin, A. To extract this compound…
A: One of the components of nutmeg is an organic compound, Trimiristin, A. To extract this compound…
Q: A student obtained a solid product in laboratory synthesis. To verify the identity of the solid, she…
A: By merely looking at any compound we cannot identify what its structure is. Melting point is an…
Q: te the r
A: Given:
Q: A student is trying to perform a single solvent recrystallization of compound X using methanol. He…
A: Ans.(i) Option (c) is right. No, you need a solvent which dissolves the sample only at boiling…
Q: Why is it dangerous to attempt a distillation in a completely closed system
A: Distillation : a process of separating mixture of compounds based on difference in their boiling…
Q: Which statement is NOT correct for an azeotropic mixture of two volatile liquids? O a. These two…
A: An azeotropic mixture is a mixture of two volatile liquids in a specific ratio. The azeotropic…
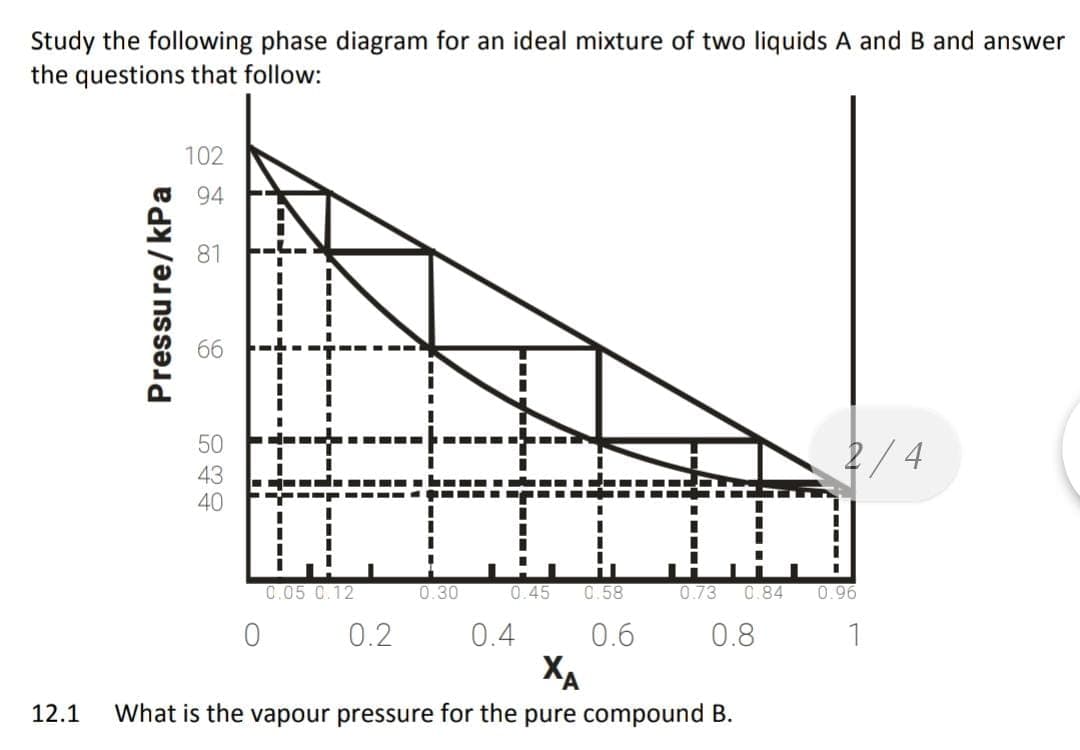
please find the attatched graph in order to help.
1. What is the vapour pressure for the pure compound B.
Step by step
Solved in 2 steps with 1 images

- Give clear handwritten solution- What is the normal boiling point of this compound? (Using phase diagram given)Consider the phase diagram below for Na, K, and the compound Na2K. For each region 1-6 give the number and identity of the phases present. Then describe the sequence of phase changes that would occur cooling of a solution of composition A (indicated on the diagram) to –30°C. Graph attachedWhy is it important to maintain the rock salt/ice mixture at a temperature of -10oC? A. Because it can be done B. Due to the limitations of the temperature probes or thermometers being used in lab. C. Because that is as cold as a salt/water mixture can get D. So all 3 solutions being measured will freeze
- When performing a recrystallization of an impure solid, why is it important to allow the heated solution to cool slowly? Group of answer choices For the molecules of the pure solid to arrange in organized crystal lattices. To encourage solubility. To avoid the need for ice. To trap particles in the crystal lattice.7.59. Starting from in figure in the liquid region, describe what happens as the temperature is decreased until the entire solution is solid.7.18. The vapor pressures of pure liquid A and pure liquid B are and , respectively. Determine the vapor pressure of: (a) a solution composed of of liquid A and of liquid B (b) a solution composed of of liquid A and of liquid B
- Glacial acetic acid has a melting point of 16.0C and enthalpy of fusion of 11.7kJ/mol. a Calculate the cryoscopic constant of glacial acetic acid. b What is the freezing point of a solution of 27.6g of I2 dissolved in 73.3g of HC2H3O2?1. What limitations are encountered in the application of sublimation as a general method of purification? 2.Explain how a process of fractional sublimation might be applied to a mixture of two substances which sublime closely together.Describe a binary eutectic diagram and show all phase regions with all the relevant information for each.
- 1. What characteristics must a substance possess for it to be separated from a heterogeneous mixture by steam distillation? 2. Explain the role of steam in facilitating the distillation of essential oils. 3. Compare and contrast simple and steam distillation in terms of the type of mixtures separated by each technique. 4. What is/are the difference(s) between a simple distillation and steam distillation?Describe how fractional distillation can better purify a mixture of liquids by discussing a liquid vapor phase diagram. Draw a liquid vapor phase diagram and use this in your answer.- What is the ideal solvent behavior for crystallization? - You will probably hear me say frequently: “minimum amount of hot solvent” during this lab. Why isthat?

