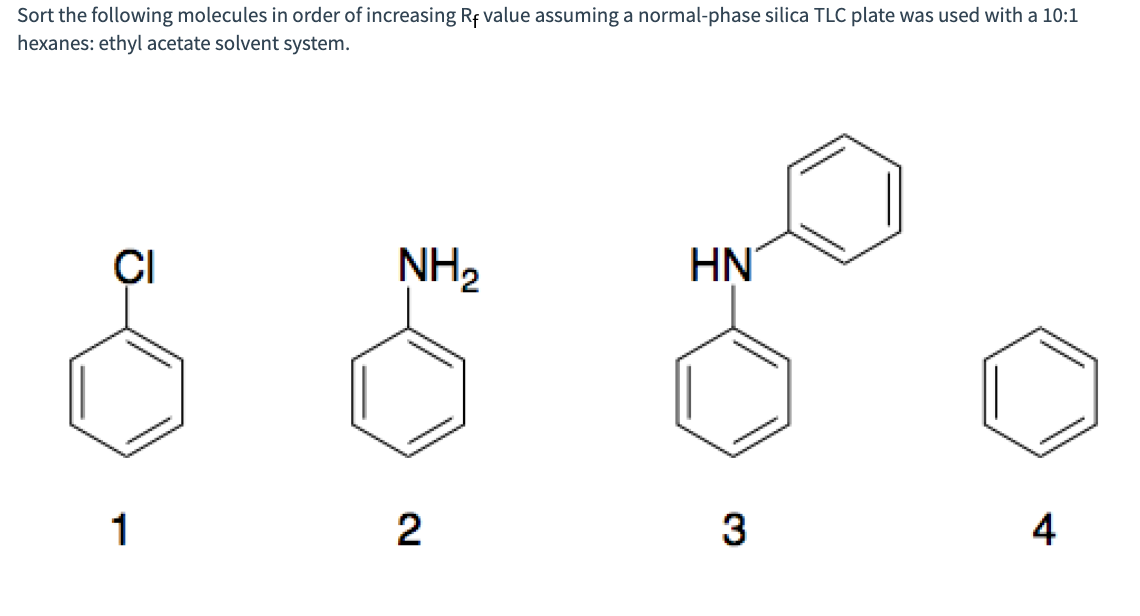
Sort the following molecules in order of increasing Rf value assuming a normal-phase silica TLC plate was used with a 10:1 hexanes: ethyl acetate solvent system. ÇI NH2 HN' 1 2 4
Q: Two species A and B are known to have water/hexane partition coefficient of 5.99 and 6.16. They are…
A: Hey, since there are multiple subparts posted, we will answer first three subparts. If you want any…
Q: VGR 9. This is the TLC plate for the solvent: Ethyl acetate 50% / acetone 50% V is crystal violet, G…
A: Retention factor (Rf) is the distance travelled by the compound divided by distance travelled by the…
Q: Chromatographic analysis using gas-liquid chromatography with a 40-cm packed column gave the…
A: Given, Compound Retention time, min…
Q: The following data were obtained by GC on a 40 cm packed column. ( VM: 1.28 mL, Vs: 0.65 mL)…
A: Solution:
Q: Which of the following is/ are adsorption chromatography: Select one: a. TLC O b. Column…
A: Adsorption chromatography Adsorption Chromatography involves the analytical separation of a…
Q: Substances A and B have retention times of 6.40 and 7.20 min, respectively, on 15.0 cm liquid…
A: (a) The column capacity factor or say the retention factor is an important experimental quantity…
Q: The TLC shown above, similar to the silica (SiO₂) TLC plates you used in lab, w developed in a…
A: For the better understanding you need to go through the following concepts first.
Q: The following data were obtained by gas-liquid chromatography using a 40.0 cm packed column: Air i.…
A: Since you have asked multiple questions, we will answer the first one for you. To get the remaining…
Q: Rank the following compounds in terms of decreasing theoretical retention factors if a reverse-phase…
A: Retention factor is the ratio of distance travelled by the sample to that of distance travelled by…
Q: föllowing analysis of a compounds (A-D) by were obtaine HPLC: Peak Retention time (min) Width of…
A: The full form of HPLC is high performance liquid chromatography. It is used to identify or separate…
Q: Differentiate between isocratic elution and gradient elution. Why is gradient elution sometimes a…
A:
Q: A chromatogram gives an ideal Gaussian peak with tr = 8.70 min and win = 0.19 min. How many…
A: 1. The number of theoretical plates (N) = 11616 2. The plate height of the column (H) = 0.00938 mm
Q: What controls the mobile phase flow rate in ascending TLC or paper chromatography? Do you think it…
A: The answer is given below
Q: What mode of chromatographic separation (RPLC, NPLC, HILIC, SEC, IEX) is best used for Analysis of…
A: a). HILIC. Because there will be occuring hydrophilic intraction chromatography also it can partly…
Q: Methanol and ethanol are separated in a capillary GC column with retention times of 370 and 385 s,…
A: The solution of the question is given below:
Q: Arrange the following compounds from highest Rț to lowest Rf on a TLC plate after elution with 50%…
A: Compounds A, B and C are ketone, acid and ether respectively. Polarity order is : acid > ketone…
Q: wo components in an HPLC separation have retention times that differ by 22 s. The first peak elutes…
A:
Q: density
A: According to the question, the readings are: 0.24, 0.35, 0.67, 0.89. We need to determine the…
Q: Two components in an HPLC separation have retention times that differ by 22 s. The first peak elutes…
A: Soln
Q: PART A: Substance A was found to have a retention times of 3.15 minutes on a 70.00-cm column. An…
A: Hello. Since the question contains multiple subparts, the first three parts are solved. In case the…
Q: Consider a sample that is a mixture composed of biphenyl, benzoic acid and benzyl alcohol. The…
A: Given, Biphenyl, Benzyl alcohol, and Benzoic acid.
Q: The result of a TLC experiment showed that Compound X has an Rf values of 0.25 on a silica plate…
A: The Rf value of a compound on a TLC plate is calculated by the formula, Rf=Distance travelled by the…
Q: i need to caluclate a rf value for the tlc plate and i really need help with showing the steps…
A: TLC can easily tell whether a reaction is complete or not. On a TLC plate, a polar compound takes…
Q: ii) What are the adjusted retention times for components A and B? iv) What is the relative retention…
A: 3 . For component A = 8-1.2=6.8 For component B = 10-1.2 = 8.8 4. Separation factor = 1.29
Q: Arrange the following compounds from highest Rț to lowest Rf on a TLC plate after elution with 50%…
A: In TLC, distance travelled by analyte species divided by distance travelled by solvent front is…
Q: Describe how a TLC study could be used to determine an appropriate solvent system for a column…
A: TLC is thin-layer chromatography and its a technique to separate non-volatile mixtures and identify…
Q: In a HPLC separation of two compounds, A and B, the retention times observed are 3.5 min. for A and…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: What mode of chromatographic separation (RPLC, NPLC, HILIC, SEC, IEX) is best used for Analysis of…
A: a). HILIC. Because there will be occurring hydrophilic interaction chromatography also it can…
Q: The separation of APAP-L,(12 = 0.28 min) from APAP-D (W12 = 0.35 min) on a chiral stationary phase…
A: APAP-L W12 = 0.28 min has a retention time of 4.78 min and APAP-D W12 = 0.35 min has aretention…
Q: The following figure indicated the separation 5-hydroxymethylfurfural (HMF), patulin (PAT),…
A: To predict the order of polarity of the given compounds.
Q: The following data were obtained by gas-liquid chromatography on a 40-cm packed column: Compound tr,…
A: To solve this problem, we will need the following relations regarding gas-liquid chromatography : 1.…
Q: 1) Study the chromatograph (below) of a mixture of Compounds A and B, run on the GCs in the teaching…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Blandard Normal... A T Distribution Table A Chi-Squared Distr... %23 VGR This is the TLC plate for…
A: Retention factor is the distance travelled by compound divided by distance travelled by the solvent.…
Q: How complexation of diclofenac sodium with hydroxy propyl beta-cyclodextrin by high pressure liquid…
A: Diclofenac sodium is a drug used to reduce pain, reduce swelling and joint stiffness (due to…
Q: Rank the following compounds in terms of decreasing theoretical retention factors if a reverse-phase…
A: Reverse-phase chromatography is a chromatographic technic in which the stationary phase is…
Q: b. Our ability to separate compounds like the four above are dependent on the properties of the…
A: Given data: Length of the column = 250mm since you have posted multiple questions, but we will able…
Q: The table contains data for a liquid chromatography separation of four compounds, A, B, C, and D, on…
A: Compound Retention time (min) Width at the Base (min) A 5.44 0.439 B 13.3 1.09 C 14.2 1.18 D…
Q: chromatogram below show a separation of a mixture of 2 components injected in a 25
A: The separation technique in which the liquid is a mobile phase where the molecules and sample ions…
Q: Two components in an HPLC separation have retention times that differ by 15 s. The first peak elutes…
A: The base number of hypothetical plates under given conditions ought to be resolved to utilize the…
Q: -24. The following data are for a liquid chromatographic column: Length of Packing Flow rate VM Vs A…
A: Given, Chromatic mixture of species A,B,C and D. Length of packing , L =24.7 cm
Q: is hexane or 70:30 hexane: ethyl acetate a better eluent based on their TLC plates and rf value in…
A: Misciblity is main factor which determine that which solvent are best eluent based on their TLC…
Q: A mixture of three compounds was chromatographed using a silica gel column with diethyl…
A:
Q: In TLC, point of origin refers to O The bhottom edge of the TLC plnto.
A: Question 1: Option 1 is the correct answer. The point of origin was sketched very lightly with…
Q: For a reverse phase TLC separation, which of the following molecules will elute first? a. 1 b. 2…
A: The organic compounds given are
Q: culate the selectivity factor (a) for the peak pairs of 1,2 and 3,4 and 5,6 in the chromatogram…
A:
Q: Why are peak areas used instead of peak height in Reverse Phase HPLC of Caffeine and Benzoic Acid?
A: In case of carrying out HPCL of caffeine and benzoic acid, peak heights are better option for…
Q: In a HPLC separation of two compounds, A and B, the retention times observed are 3.5 min. for A and…
A: Length of the column = 15 cm Retention time for A = 3.5 min Retention time for B = 4.5 min
Q: Sort the following molecules in order of increasing Rf value assuming a normal-phase silica TLC…
A: In thin layer chromatography is a technique to separate different components in a mixture based on…
Q: Arrange the following compounds from highest Rț to lowest Rç on a TLC plate after elution with 50%…
A: The given compounds are A = Ketone B= Carboxylic acid C=Ether. We need to predict the polarity…
Q: 2) Consider the following compounds. Which rankings list these compounds in terms of Ry from lowest…
A: Thin layer chromatography.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- in what order (from lowest to highest Rf) would you expect to find compund A, B, and C on a silica gel TLC plate with dichloromethane? Draw flowchart for separation with org solvent: ether, methylene chloride, aq: HCl, NaOH, NaHCO3In TLC, why does a 3:1:1 ratio of the developing solvent of n-butanol/acetic acid/water move up the TLC plate so slowly?In TLC, why does the developing solvent 3:1:1 n-butanol/acetic acid/water climb up the TLC plate so slowly?
- For a reverse phase TLC separation, which of the following molecules will elute first?a. 1 b. 2 c. 3A mixture of biphenyl, benzyl alcohol, and benzoic acid is spotted on a silica gel TLC plate and developed using Ethyl Acetate with 0.5% acetic acid as the solvent. Predict the relative Rf (highest to lowest).HPLC VFA analysis - why do I get so different peaks for very similar samples?
- During a Thin layer chromatography (TLC) analysis of crystallized napthalene, What would be the effect of adding acetic acid to the mixture in regards to the separation/spot shape?Thin Layer Chromatography why the non-polar compounds move up the plate most rapidly (higher Rf value), whereas polar substances travel up the TLC plate slowly or not at all (lower Rf value)? Please help me explain this question! Thank you so much!i need to caluclate a rf value for the tlc plate and i really need help with showing the steps please! Reduction of 4-Nitrobenzaldehyde to 4-Nitrobenzyl Alcohol and Thin Layer Chromatography
- is hexane or 70:30 hexane: ethyl acetate a better eluent based on their TLC plates and rf value in column chromatography separation of ferrocene-acetylferrocene.Two compounds A and B from GC on two open tubular columns contains a XY-1 (100% methyl) and a XY-2 (50% methyl, 50% phenyl), with the same diameter, thickness and length under the same experimental conditions. The retention times and peak widths are show in the Table 1.Hint: phenyl groups interact more strongly with polar groups than methyl groups. Which compound is more polar? Explain your answerTwo components in an HPLC separation have retention times that differ by 22 s. The first peak elutes in 10.5 min and the peak widths are approximately equal. Calculate the minimum number of theoretical plates needed to achieve an acceptable resolution



