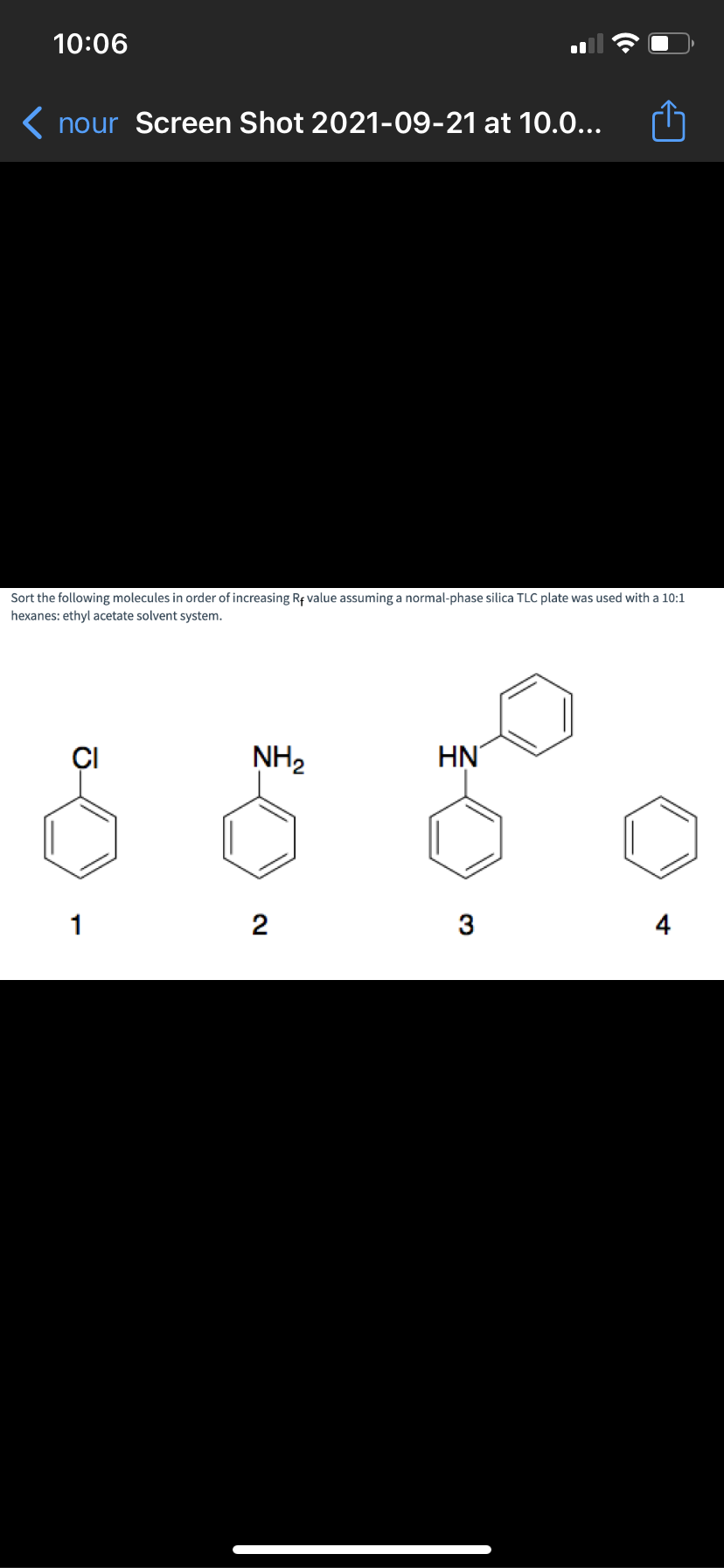
Sort the following molecules in order of increasing Rf value assuming a normal-phase silica TLC plate was used with a 10:1 hexanes: ethyl acetate solvent system. ÇI NH2 HN 1 2 3 4
Sort the following molecules in order of increasing Rf value assuming a normal-phase silica TLC plate was used with a 10:1 hexanes: ethyl acetate solvent system. ÇI NH2 HN 1 2 3 4
Chapter10: Tlc Analysis Of Analgesic Drugs
Section: Chapter Questions
Problem 2Q
Related questions
Question
Transcribed Image Text:10:06
( nour Screen Shot 2021-09-21 at 10.0..
Sort the following molecules in order of increasing Rf value assuming a normal-phase silica TLC plate was used with a 10:1
hexanes: ethyl acetate solvent system.
NH2
HN
1
2
3
4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
