The value of Kp (@ 25 °C) for compound H when partitioned between water and chloroform is 2.1. Starting with a solution of 30 g of compound H in 250 mL of water what quantity of compound H (in grams) would remain in the water layer after two successive extractions with chloroform each using 125 mL of chloroform? Please carry maximum number of significant figures through to final answer then round to one decimal place. Please do not include units.
The value of Kp (@ 25 °C) for compound H when partitioned between water and chloroform is 2.1. Starting with a solution of 30 g of compound H in 250 mL of water what quantity of compound H (in grams) would remain in the water layer after two successive extractions with chloroform each using 125 mL of chloroform? Please carry maximum number of significant figures through to final answer then round to one decimal place. Please do not include units.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.26QAP
Related questions
Question
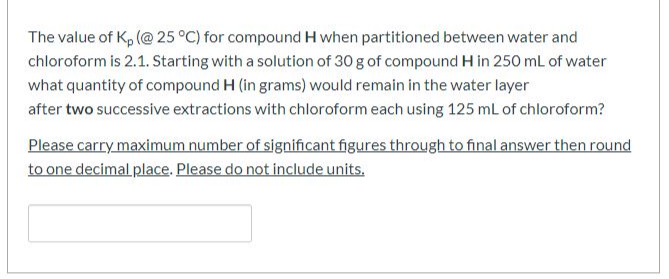
Transcribed Image Text:The value of Kp (@ 25 °C) for compound H when partitioned between water and
chloroform is 2.1. Starting with a solution of 30 g of compound H in 250 mL of water
what quantity of compound H (in grams) would remain in the water layer
after two successive extractions with chloroform each using 125 mL of chloroform?
Please carry maximum number of significant figures through to final answer then round
to one decimal place. Please do not include units.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

