Туре of Reaction Catalyzed Classification 1. Oxidoreductases Oxidation-reduction reactions 2. Transferases Transfer of functional groups 3. Hydrolases Hydrolysis reactions 4. Lyases Group elimination to form double bonds 5. Isomerases Isomerization 6. Ligases Bond formation coupled with ATP hydrolysis (а) CO0 H-C-CH3 -H3C-C-H NH3 NH; (b) Ç00- H C=0 + + H* c=0 + 0=c=0 CH3
Туре of Reaction Catalyzed Classification 1. Oxidoreductases Oxidation-reduction reactions 2. Transferases Transfer of functional groups 3. Hydrolases Hydrolysis reactions 4. Lyases Group elimination to form double bonds 5. Isomerases Isomerization 6. Ligases Bond formation coupled with ATP hydrolysis (а) CO0 H-C-CH3 -H3C-C-H NH3 NH; (b) Ç00- H C=0 + + H* c=0 + 0=c=0 CH3
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter22: Gluconeogenesis, Glycogen Metabolism, And The Pentose Phosphate Pathway
Section: Chapter Questions
Problem 11P: Understanding the Mechanisms of Reactions Related to Transketolase The mechanistic chemistry of the...
Related questions
Question
Which type of enzyme (Table) catalyzes the following (given) reactions?
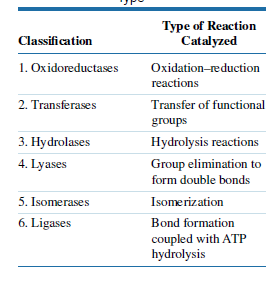
Transcribed Image Text:Туре of Reaction
Catalyzed
Classification
1. Oxidoreductases
Oxidation-reduction
reactions
2. Transferases
Transfer of functional
groups
3. Hydrolases
Hydrolysis reactions
4. Lyases
Group elimination to
form double bonds
5. Isomerases
Isomerization
6. Ligases
Bond formation
coupled with ATP
hydrolysis
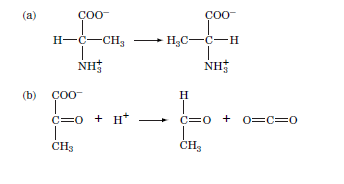
Transcribed Image Text:(а)
CO0
H-C-CH3
-H3C-C-H
NH3
NH;
(b) Ç00-
H
C=0 +
+ H*
c=0 + 0=c=0
CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning