
Concept explainers
(a)
Interpretation:
For the given species, the complete Lewis structure is to be completed by adding multiple bonds and/or lone pairs.
Concept introduction:
In order to draw a Lewis structure for a molecule, start by counting the total number of valence electrons in a molecule. The number of valence electrons by each atom is the same as its group number. For the given skeleton of the molecule, distribute the remaining electrons as lone pairs. In doing so, start with the outer atoms and work inwards. Try to achieve an octet on each atom other than hydrogen. If there is an atom with less than an octet, increase the atom’s share of electrons by converting lone pairs from neighboring atoms to bonding pairs thereby creating double or triple bonds. For an uncharged atom, carbon atoms will have a maximum of four bonds; Nitrogen will have three bonds and one lone pair, while oxygen will have two bonds and two lone pairs. Hydrogen always contributes to one bond. The number of bonds in case of halogen is one; while there will be three lone pair of electrons on halide atoms.
Answer to Problem 1.24P
The complete Lewis structure for the given species is:
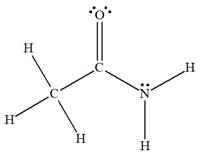
Explanation of Solution
The given species is:
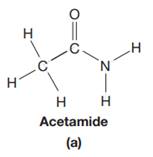
The formula for the species above is
The carbon atom on the left side has four bonds, thus, its octet is complete. The carbon atom in the middle has four bonds, hence, its octet is also complete. The nitrogen atom has three bonds, thus, its octet is not complete. There should be one lone pair of electron on nitrogen. The double bonded oxygen atom has got two bonds. Thus, in order to complete its octet, it should possess two lone pair of electrons. Thus, all the
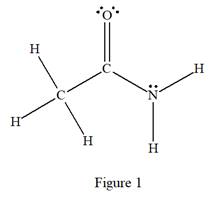
The complete Lewis structure for the given species including multiple bonds and lone pairs is shown in Figure 1 above.
(b)
Interpretation:
For given species, the complete Lewis structure is to be completed by adding multiple bonds and/or lone pairs.
Concept introduction:
In order to draw a Lewis structure for a molecule, start by counting the total number of valence electrons in a molecule. The number of valence electrons by each atom is the same as its group number. For a charged species, each negative charge increases the number of valence electrons by one while each positive charge decreases the number of valence electrons by one. For the given skeleton of the molecule, distribute the remaining electrons as lone pairs. In doing so, start with the outer atoms and work inwards. Try to achieve an octet on each atom other than hydrogen. If there is an atom with less than an octet, increase the atom’s share of electrons by converting lone pairs from neighboring atoms to bonding pairs thereby creating double or triple bonds. For an uncharged atom, carbon atoms will have maximum of four bonds. Nitrogen will have three bonds and one lone pair, while oxygen will have two bonds and two lone pairs. Hydrogen always contributes to one bond. The number of bond in case of halogen is one, while there will be three lone pair of electrons on halide atoms.
Answer to Problem 1.24P
The complete Lewis structure for the given species is:
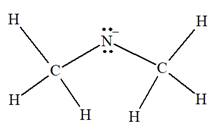
Explanation of Solution
The given species is:
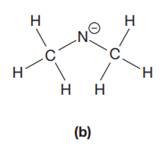
The formula for the species above is
The carbon atom on the left as well as on the right has four bonds, thus, their octets are complete. The nitrogen atom has two bonds and a negative formal charge. This suggests that the remaining four electrons should be present on the nitrogen atom so as to complete its octet and have a negative formal charge. Thus, the complete Lewis structure for the given species is:
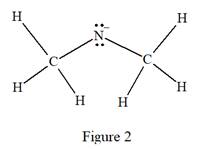
The complete Lewis structure for the given species including multiple bonds and lone pairs is shown in Figure 2 above.
(c)
Interpretation:
For given species, the complete Lewis structure is to be completed by adding multiple bonds and/or lone pairs.
Concept introduction:
In order to draw a Lewis structure for a molecule, start by counting the total number of valence electrons in a molecule. The number of valence electrons by each atom is the same as its group number. For a charged species, each negative charge increase the number of valence electrons by one while each positive charge decrease the number of valence electrons by one. For the given skeleton of the molecule, distribute the remaining electrons as lone pairs. In doing so, start with the outer atoms and work inwards. Try to achieve an octet on each atom other than hydrogen. If there is an atom with less than an octet, increase the atom’s share of electrons by converting lone pairs from neighboring atoms to bonding pairs thereby creating double or triple bonds. For an uncharged atom, carbon atoms will have maximum of four bonds. Nitrogen will have three bonds and one lone pair, while oxygen will have two bonds and two lone pairs. Hydrogen always contributes to one bond. The number of bond in case of halogen is one, while there will be three lone pair of electrons on halide atoms.
Answer to Problem 1.24P
The complete Lewis structure for the given species is:
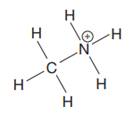
Explanation of Solution
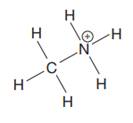
The given species is:
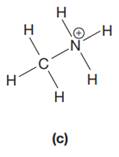
The formula for the species above is
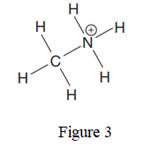
The complete Lewis structure for the given species including multiple bonds and lone pairs is shown in Figure 2 above.
(d)
Interpretation:
For given species, the complete Lewis structure is to be completed by adding multiple bonds and/or lone pairs.
Concept introduction:
In order to draw a Lewis structure for a molecule, start by counting the total number of valence electrons in a molecule. The number of valence electrons by each atom is the same as its group number. For a charged species, each negative charge increase the number of valence electrons by one while each positive charge decrease the number of valence electrons by one. For the given skeleton of the molecule, distribute the remaining electrons as lone pairs. In doing so, start with the outer atoms and work inwards. Try to achieve an octet on each atom other than hydrogen. If there is an atom with less than an octet, increase the atom’s share of electrons by converting lone pairs from neighboring atoms into bonding pairs thereby creating double or triple bonds. For an uncharged atom, carbon atoms will have maximum of four bonds. Nitrogen will have three bonds and one lone pair, while oxygen will have two bonds and two lone pairs. Hydrogen always contributes to one bond. The number of bond in case of halogen is one, while there will be three lone pair of electrons on halide atoms.
Answer to Problem 1.24P
The complete Lewis structure for the given species is:
Explanation of Solution
The given species is:
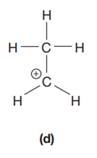
The formula for the species above is
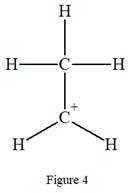
The complete Lewis structure for the given species including multiple bonds and lone pairs is shown in Figure 4 above.
Want to see more full solutions like this?
Chapter 1 Solutions
ORG. CHEM. LL W/SSM+SG+ACCESS
- See image and include any lone pairs and any charges as wellarrow_forwardBelow are two sets of resonance structures. Where applicable provide the missing curved arrow notation, lone pair electrons, and nonzero formal charge. Do not delete or add any bonds or atoms.arrow_forwardProblem. Calculate the dipole moment of Glycine molecule using partial charge and positions of atoms. In this problem, How can I get the partial charge of the atoms? Should I find the reference or appendix?arrow_forward
- Draw the major resonance structure for the compound shown; include lone pairs of electrons, formal charges, and condensed hydrogen atoms (located in the More menu). Then draw curved arrows to show how this can be converted to the Lewis structure givenarrow_forwardFor each of the following compounds, identify any polar covalent bonds by indicate δ+ and δ- symbols in the appropriate locations.arrow_forwardDraw the curved arrows and the resulting resonance structure for the following structure. Include lone pairs and charges in your structure.arrow_forward
- Expand the following condensed formula so as to show all of the bonds and all of the unshared electron pairsarrow_forwardPlease draw all possible resonance structures, provided with an explanationarrow_forwardThe molecule shown here has quite a large dipole, as indicated in its electrostatic potential map. Explain why.Hint: Consider various resonance structures.arrow_forward
- For the following molecule, give, according to IUPAC standards, it's name. Follow the rules for commas, spaces and dashes.arrow_forwardDraw all reasonable resonance structures for the following compounds. Be sure to show the proper arrows to indicate electron movementarrow_forwardPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Write the resonance structure that would result from pushing the electrons as indicated by the curved arrow.Zarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

