
Concept explainers
(a)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. Each non-hydrogen atom is written explicitly followed immediately by the number of hydrogen atoms that are bonded to it. Multiple bonds are not shown explicitly in the condensed structure. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis, in the condensed formula, represents that the repetitive unit is attached to the previous carbon atom. The
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
![]()
Explanation of Solution
The given line structure is:
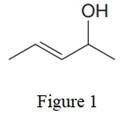
In the given line structure, there is a five carbon chain, with a carbon-carbon double bond and with a hydroxyl group. Each carbon atom is written explicitly followed immediately by the number of hydrogen atoms that are bonded to it. Multiple bonds are not shown explicitly in the condensed formula. Each carbon atom has a maximum of four bonds while each oxygen atom should have a maximum of two bonds. A parenthesis must be used for the carbon atom having three different groups attached. Thus, the hydroxyl group attached to the carbon atom must be shown in the parenthesis.
Thus, the condensed structure for the given line structure is:

The condensed structure for the given line structure is shown in Figure 2 above.
(b)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. Each carbon atom is written explicitly followed immediately by the number of hydrogen atoms that are bonded to it. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. The
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
![]()
Explanation of Solution
The given line structure is:
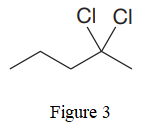
In the given line structure, there is a five carbon chain, with two chlorine atoms attached to one of the carbon atoms of the chain. Each carbon atom should have a maximum of four bonds while each oxygen atom should have a maximum of two bonds. A parenthesis must be used for the carbon atom having three different groups attached. Thus, the
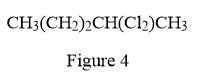
The condensed structure for the given line structure is shown in Figure 4 above.
(c)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. Each non-hydrogen atom is written explicitly followed immediately by the number of hydrogen atoms that are bonded to it. Multiple bonds are not shown explicitly in the condensed structure. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. The
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
Explanation of Solution
The given line structure is:
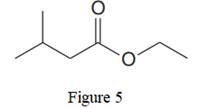
In the given line structure, there is a chain of four carbon atoms on the left side of a singly bonded oxygen. Each carbon atom is written explicitly followed immediately by the number of hydrogen atoms that are bonded to it. Multiple bonds are not shown explicitly in the condensed structure while an ethyl fragment is present at the right side of the singly bonded oxygen atom. The
Thus, the condensed structure for the given line structure is:
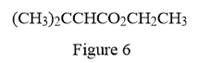
The condensed structure for the given line structure is shown in Figure 6 above.
(d)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. Rings are generally now shown in their condensed formulas, but they are commonly shown in their partially condensed form. Multiple bonds in the ring and outside the ring are shows as they are. Parentheses are also used to clarify when two or three groups are attached to the same carbon atom.
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
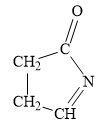
Explanation of Solution
The given line structure is:
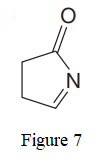
In the given line structure, a five membered ring containing a nitrogen atom is present. While writing a condensed formula, a ring is shown in its partial condensed formula. One of the carbon atoms in the ring forms a double bond with the oxygen atom which is shown as is.
Thus, the condensed structure for the given line structure is:
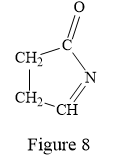
The condensed structure for the given line structure is shown in Figure 8 above.
(e)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. Each non-hydrogen atom is written explicitly, followed immediately by the number of hydrogen atoms that are bonded to it. Multiple bonds are not shown explicitly in the condensed structure. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. Rings are generally shown in their condensed formulas, but they are commonly shown in their partially condensed form. Parentheses are also used to clarify when two or three groups are attached to the same carbon atom.
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
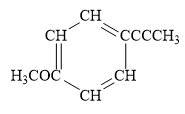
Explanation of Solution
The given line structure is:
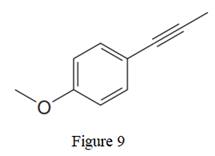
In the above line structure, the disubstituted benzene ring is present. One substituent of the benzene ring is a three carbon chain with an internal triple bond. The other substituent is a methoxy group,
Thus, the condensed structure for the given line structure is:
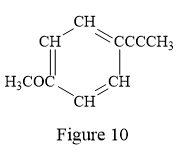
The condensed structure for the given line structure is shown in Figure 10 above.
(f)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. Each non-hydrogen atom is written explicitly, followed immediately by the number of hydrogen atoms that are bonded to it. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. Rings are generally now shown in their condensed formulas, but they are commonly shown in their partially condensed form. Parentheses are also used to clarify when two or three groups are attached to the same carbon atom. A formal charge is shown explicitly on the atom.
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
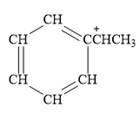
Explanation of Solution
The given line structure is:
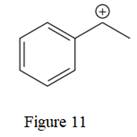
The above line structure is a structure for a cation. A carbocation is a carbon bearing a positive formal charge which is explicitly shown in the condensed formula. A six membered carbon ring with alternate double and single bonds is present and is shown as a partial condensed structure.
Thus, the condensed structure for the given line structure is:
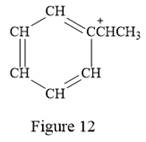
The condensed structure for the given line structure is shown in Figure 12 above.
(g)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. Each non-hydrogen atom is written explicitly, followed immediately by the number of hydrogen atoms that are bonded to it. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. Rings are generally now shown in their condensed formulas, but they are commonly shown in their partially condensed form. Parentheses are also used to clarify when two or three groups are attached to the same carbon atom. A formal charge is shown explicitly on the atom.
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
Explanation of Solution
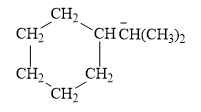
The given line structure is:
The above line structure is a structure for an anion. A six membered carbon ring with single bonds is present. Each carbon atom is written explicitly followed immediately by the number of hydrogen atoms that are bonded to it. The negative charge on the carbon atom is shown explicitly. The two methyl groups attached to the carbon bearing a negative charge are shown in the parenthesis.
Thus, the condensed structure for the given line structure is:
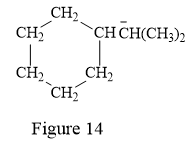
The condensed structure for the given line structure is shown in Figure 14 above.
(h)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. Each non-hydrogen atom is written explicitly, followed immediately by the number of hydrogen atoms that are bonded to it. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. Rings are generally now shown in their condensed formulas, but they are commonly shown in their partially condensed form. Parentheses are also used to clarify when two or three groups are attached to the same carbon atom. A formal charge is shown explicitly on the atom.
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
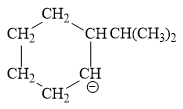
Explanation of Solution
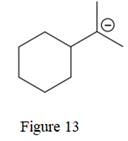
The given line structure is:
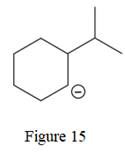
The above line structure is a six membered ring with a negative charge on one of the carbon atoms of the ring. The carbon bearing a negative charge is shown as is. The ring is monosubstituted with an isopropyl group. The two methyl groups in the isopropyl fragments is shown with the help of parenthesis.
Thus, the condensed structure for the given line structure is:
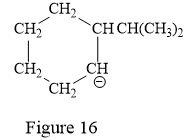
The condensed structure for the given line structure is shown in Figure 16 above.
(i)
Interpretation:
Given line structure is to be drawn using condensed formula.
Concept introduction:
The condensed formula indicates how the atoms should be connected in a given molecule. To arrive at a total charge of zero, each carbon should have a maximum of four bonds while each oxygen should have a maximum of two bonds and two lone pairs. A parenthesis in the condensed formula represents that the repetitive unit is attached to the previous carbon atom. The
Answer to Problem 1.64P
For the given line structure, the condensed structure is:
![]()
Explanation of Solution
The given line structure is:
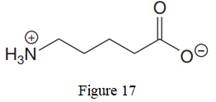
The above line structure is a chain of five carbon atoms. One end of the chain has carboxylate ion, in which the oxygen carries a negative charge. The other end of the chain has a nitrogen atom with three hydrogen atoms directly attached to it and carrying a positive charge. Formal charges on atoms are shown explicitly.
Thus, the condensed structure for the given line structure is:
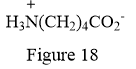
The condensed structure for the given line structure is shown in Figure 18 above.
Want to see more full solutions like this?
Chapter 1 Solutions
ORGANIC CHEMISTRY:PRIN...(PB)-W/ACCESS
- Write the dash formula of this compound. Expand the following condensed formula so as to show all the bonds and unshares electrons. (CH3)2CCH(CH3)2arrow_forwardFor the compound below draw all resonance structures that are possible. Make sure to include formal charges, curved arrows (to show electron flow), and all electron pairs.arrow_forwardUsing the line structure of C13H18ClNO, draw the expanded structural formula of the molecule (showing all atoms and all covalent bonds). Add lone pairs of electrons to each atom othe than C or H.arrow_forward
- Write the dash formula of the following compounds. Expand the following condensed formulas so as to show all the bonds and unshared electron pairs.arrow_forwarda) Draw the formal charges to the molecule below. Label atleast two functional groups found in this molecule. Identify and circle the π bonds. b) Draw a curly arrow notation to show how the electrons are redistributed to show a new resonance structure in which the formal charge has moved to a different heteroatom.arrow_forwardConsider the Lewis structure shown for thionitromethane. Draw the major resonance structure for the compound shown; include lone pairs of electrons, formal charges, and condensed hydrogen atoms (located in the More menu). Then draw curved arrows to show how this can be converted to the Lewis structure given.arrow_forward
- What is the dash formula of CH3CH2CH(NH2)CH3. Expand the following condensed formula so as to show all the bonds and unshares electrons.arrow_forwardFor each of the organic molecules listed below write the name and condensed formular in the respective space below.arrow_forwardWhat is the relation between the following pair of compounds? (The compounds are in the image)arrow_forward
- C4H10O is the formula of diethyl ether. The same group of atoms is attached on either side of the oxygen atom. Draw the Lewis diagram. Diethyl ether was among the first anesthetics used in surgery, but it has been largely replaced by safer anesthetics today.arrow_forwardConsider the following three molecules, A-C:arrow_forwardDraw a Lewis dot structure for each of the following molecules and assign charges where appropriate.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
