
(a)
Interpretation:
The element having the given configuration has to be identified. Also whether the configuration represent ground state or not has to be identified.
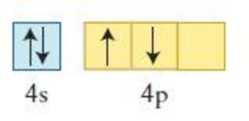
Figure 1
Concept Introduction:
Electronic configuration: The electron configuration is the distribution of electrons of an atom or molecule in atomic or molecular orbitals.
Electrons occupy the lowest energy orbitals. The increasing order of orbital energy is
The energy order of the orbital for the first three periods is as follows,
The orbital which is closer to the nucleus has lower energy; therefore the
In general, the orbitals can hold maximum of two electrons, the two electrons must have opposite spin.
The subshell ordering by Aufbau principle is given below,
Electrons are filled in each orbital one after another in the increasing order of energy. While filling the orbitals, more than two electrons can be placed in an orbital. And also, the spin of the two electron in the same orbital must be paired.
If there are more than one orbitals in a subshell are available for filling the electron, then electrons with parallel spin goes to different subshell rather than pairing two electrons in one orbital.
If an atom having electrons in energy states higher than predicted by the above rules then it is said to be in an excited state.
(b)
Interpretation:
The element having the given configuration has to be identified. Also whether the configuration represent ground state or not has to be identified.
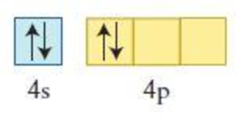
Figure 2
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The element having the given configuration has to be identified. Also whether the configuration represent ground state or not has to be identified.
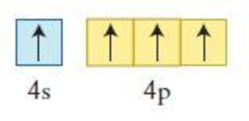
Figure 3
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The element having the given configuration has to be identified. Also whether the configuration represent ground state or not has to be identified.
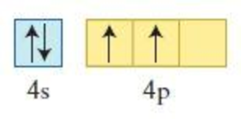
Figure 4
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Chemical Principles: The Quest for Insight
- Although we may draw the 4s orbital with the shape of a ball, there is some probability of finding the electron outside the ball we draw. Is this statement true or false? Comment on this statement.arrow_forwardGiven the valence electron orbital level diagram and the description, identify the element or ion. a. A ground state atom b. An atom in an excited state (assume two electrons occupy the 1s orbital) c. A ground state ion with a charge of 1arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





