
Concept explainers
a.
Interpretation: The hybridization state and geometry of the following structure need to be predicted:
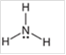
Concept Introduction: The arrangement of electron pairs is identified according to VSEPR (Valence Shell Electron Pair Repulsion) theory wherein all the electron pairs are aligned in such a way that they are at maximum distance from each other. The structure of every molecule in VSEPR is explained by considering the steric number (combination of lone pairs and
b.
Interpretation: The hybridization state and geometry of the following structure need to be predicted:
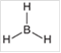
Concept Introduction: The arrangement of electron pairs is identified according to VSEPR (Valence Shell Electron Pair Repulsion) theory wherein all the electron pairs are aligned in such a way that they are at maximum distance from each other. The structure of every molecule in VSEPR is explained by considering the steric number (combination of lone pairs and
c.
Interpretation: The hybridization state and geometry of the following structure need to be predicted:
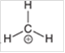
Concept Introduction: The arrangement of electron pairs is identified according to VSEPR (Valence Shell Electron Pair Repulsion) theory wherein all the electron pairs are aligned in such a way that they are at maximum distance from each other. The structure of every molecule in VSEPR is explained by considering the steric number (combination of lone pairs and
d.
Interpretation: The hybridization state and geometry of the following structure need to be predicted:
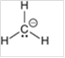
Concept Introduction: The arrangement of electron pairs is identified according to VSEPR (Valence Shell Electron Pair Repulsion) theory wherein all the electron pairs are aligned in such a way that they are at maximum distance from each other. The structure of every molecule in VSEPR is explained by considering the steric number (combination of lone pairs and
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
ORGANIC CHEMISTRY
- Two important industrial chemicals, ethene, C2H4, and propene, C3H6, are produced by the steam (or thermal) cracking process: 2C3H8(g)C2H4(g)+C3H6(g)+H2(g) For each of the four carbon compounds, do the following: (a) Draw a Lewis structure. (b) Predict the geometry about the carbon atom. (c) Determine the hybridization of each type of carbon atom.arrow_forward• predict the geometry of a molecule from its, Lewis structure.arrow_forward1. A. What is an energy (electron or valence) shell and B. What determines whether it makes an atom react? A. В. 2. Name and describe the 4 major types of molecular bonds: А. В. C. D.arrow_forward
- Which of the following bonds would be the least polar? A. B-F B. O-F C. F-F D.C-Farrow_forwardRefer to the structure below to answer the following question H Which of the labeled bonds are trans to bond b? 2 3 a. b. C. d. a b C d bond a bond c bond d d bond earrow_forwardWhich of the following can be best explained by temporary dipole moments? Choose one or more: A. This explains why ammonia and nitrogen gas exhibit an attractive force between them. B. This explains how the molecules hydrogen fluoride and methanol can exhibit uncharacteristically strong intermolecular forces. C. This explains why long hydrocarbon chains have relatively high boiling points. D. This explains how two noble gases' molecules can have an attractive force between them. E. This explains why the dipole-dipole attractive force between dimethyl ether and acetone does not entirely account for the attractive force between these molecules.arrow_forward
- In structure B the carbon-oxygen bond will be a A. double bond B. single bond C. triple bond D. None of the abovearrow_forwardSee the attached lewis structure and answer the following: 1.Predict the molecular shape of methane. a. tetrahedral b. trigonal planar (120°) c. bent d. trigonal pyramidal e. linear 2. Predict the molecular shape of the carbonate ion. a. tetrahedral b. trigonal planar (120°) c. bent d. linear e. trigonal pyramidal 3. Predict the molecular shape of carbon dioxide. a. linear b. bent c. trigonal pyramidal d. trigonal planar (120°) e. tetrahedral 4. Predict the molecular shape of the sulfite ion. a. bent b. trigonal planar (120°) c. tetrahedral e. linear f. trigonal pyramidalarrow_forwardThe molecular shapes depend on: A. The total number of valence shell electron pairs present around the terminal atoms in the molecule and the number of bonded pairs. B. The total number of lone pairs present around the central atom in the molecule and the number of bonded pairs. C. The total number of valence shell electron pairs present around the central atom in the molecule and the number of lone pairs. D. The total number of valence shell electron pairs present around the central atom in the molecule and the number of bonded pairs.arrow_forward
- 1. State Whether following statements are true (T) or False (F) in the space provided. a) Electron geometry and molecular geometry is not the same for all molecules. b) The enthalpy of neutralization of strong acid -strong base reaction is a constant. c) In the ideal gas law equation, the unit of temperature is in Kelvin (K). d) In an endothermic reaction heat is released from the system to the surroundings. e) Both AgCl and PbCl2 precipitates have the same color. contained in a calorimeter. When thermal equilibrium is reached, the temperature of the water is measured to be 30.6 °C. What is the specific heat of the metal? (10 pts) (heat capacity of water is 4.18 J/g°C) GHIZA 1 WLO TREICONAY aducation.ti.comarrow_forwardQuestion five in the review section. They are not giving us the value of a or B and I believe the answer is going to be D. M I don't really think of corinne as being polar nor do I think it has a lot of Vanderwall forces. How to think about this. ? I looked at the a value in the table for chlorine and it is pretty high. As you go down a group I kniw london forces increase to to more electrons.arrow_forwardBased on periodic trends in electonegativity, which of the following bonds is expected to be MOST polar? A. H-F B. C-C C. N-O D. C-H E. C-Oarrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





