
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 52P
- a. Which of the indicated bonds in each compound is shorter?
- b. Indicate the hybridization of the C, O, N, and halogen atoms in each of the compounds.
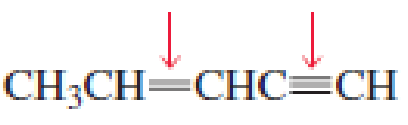
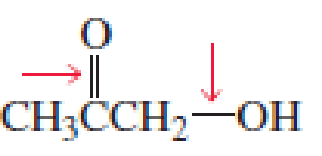
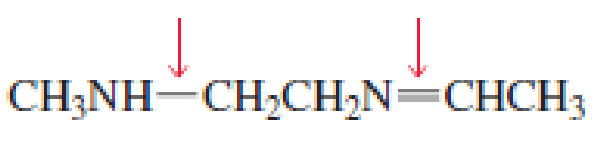
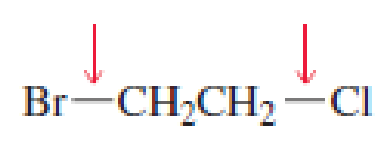
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What orbitals are used to form the bonds indicated in each molecule? Of the indicated C – C bonds, which is the shortest?
In the hydrocarbon:
(a) What is the hybridization at each carbon atom in themolecule? (b) How many σ bonds are there in the molecule?(c) How many π bonds? (d) Identify all the 120° bond anglesin the molecule.
CH3+ and CH3− are two highly reactive carbon species.
a. What is the predicted hybridization and geometry around each carbon atom?
b.Two electrostatic potential plots are drawn for these species. Which ion corresponds to which diagram and why?
Chapter 1 Solutions
Essential Organic Chemistry, Global Edition
Ch. 1.1 - Oxygen has three isotopes, 16O, 17O, and 18O. The...Ch. 1.2 - Prob. 2PCh. 1.2 - How many valence electrons do chlorine, bromine,...Ch. 1.2 - Look at the relative positions of each pair of...Ch. 1.3 - a. Find potassium (K) in the periodic table and...Ch. 1.3 - Which bond is more polar?Ch. 1.3 - Which of the following has a. the most polar bond?...Ch. 1.3 - Use the symbols + and to show the direction of...Ch. 1.3 - After examining the potential maps for LiH, HF,...Ch. 1.4 - An atom with a formal charge does not necessarily...
Ch. 1.4 - Prob. 12PCh. 1.4 - a. Draw two Lewis structures for C2H6O. b. Draw...Ch. 1.4 - Draw the lone-pair electrons that are not shown in...Ch. 1.4 - Prob. 16PCh. 1.4 - Which of the atoms in the molecular models in...Ch. 1.4 - Prob. 18PCh. 1.7 - What orbitals are used to form the 10 sigma bonds...Ch. 1.9 - Put a number in each of the blanks: a. ___ s...Ch. 1.11 - Predict the approximate bond angles in a. the...Ch. 1.11 - According to the potential map for the ammonium...Ch. 1.12 - Prob. 25PCh. 1.13 - a. Predict the relative lengths and strengths of...Ch. 1.13 - Prob. 28PCh. 1.14 - Which of the bonds in a carbonoxygen double bond...Ch. 1.14 - Caffeine is a natural insecticide, found in the...Ch. 1.14 - a. What is the hybridization of each of the carbon...Ch. 1.14 - Prob. 33PCh. 1.14 - Describe the orbitals used in bonding and the bond...Ch. 1.15 - Account for the difference in the shape and color...Ch. 1.15 - Which of the following molecules would you expect...Ch. 1 - Draw a Lewis structure for each of the following...Ch. 1 - Prob. 38PCh. 1 - What is the hybridization of all the atoms (other...Ch. 1 - Prob. 40PCh. 1 - Draw the condensed structure of a compound that...Ch. 1 - Prob. 42PCh. 1 - Prob. 43PCh. 1 - Draw a Lewis structure for each of the following...Ch. 1 - Prob. 45PCh. 1 - List the bonds in order from most polar to least...Ch. 1 - What is the hybridization of the indicated atom in...Ch. 1 - Write the Kekul structure for each of the...Ch. 1 - Assign the missing formal charges.Ch. 1 - Predict the approximate bond angles for the...Ch. 1 - Prob. 51PCh. 1 - a. Which of the indicated bonds in each compound...Ch. 1 - In which orbitals are the lone pairs in nicotine?...Ch. 1 - Draw the missing lone-pair electrons and assign...Ch. 1 - Rank the following compounds from highest dipole...Ch. 1 - Prob. 56PCh. 1 - a. Which of the species have bond angles of 109.5?...Ch. 1 - Prob. 58PCh. 1 - Sodium methoxide (CH3ONa) has both ionic and...Ch. 1 - a. Why is a H 8 H bond (0.74 ) shorter than a C 8...Ch. 1 - Which compound has a larger dipole moment, CHCl3...Ch. 1 - Which compound has a longer C 8 Cl bond?Ch. 1 - Prob. 63PCh. 1 - The following compound has two isomers. One isomer...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach (4th Edition)
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Phosphoserine is a less-common amino acid. (a) Identify the hybridizations of atoms I through 5. (b) What are the approximate values of the bond angles A, B. C, and D? (c) What are the most polar bonds in the molecule?arrow_forwardGive the shape that describes each hybrid orbital set: (a) sp2 (b) sp3d (c) sp (d) sp3d2arrow_forwardNumerous molecules are detected in deep space. Three of them are illustrated here. (a) Are these compounds isomers? (b) Indicate the hybridization of each C atom in each molecule. (c) What is the value of the HCH angle in each of the three molecules? (d) Which of these molecules is/are polar? (e) Which molecule should have the strongest carbon-carbon bond? The strongest carbon-oxygen bond?arrow_forward
- (a) Determine the hybridization of each N atom in ibrutinib, a drug used to treat mantle cell lymphoma and chronic lymphocytic leukemia. (b) In what type of orbital does the lone pair on each N atom reside? (c) How many sp2 hybridized atoms does ibrutinib contain?arrow_forwardIn the hydrocarbon(a) What is the hybridization at each carbon atom in themolecule? (b) How many s bonds are there in the molecule?(c) How many p bonds? (d) Identify all the 120° bond anglesin the molecule. [Section 9.6]arrow_forward1. Which of the bonds would have the highest bond dissociation energy? Why 2. How many sigma and pi bonds are present? 3. Identify the hybridization of all carbons in the compound.arrow_forward
- Harry creates a compound that contains only 1 pi (π) bond in total. The central atom has 4s2, 3d10, and 4p5. There are only 3 surrounding atoms with the electron configuration: 1s2 2s2 2p5. What is the formula and VSEPR shape for this compound? Explain how this is possible with respect to hybridized orbitals and how electrons are moved around to create this compound.arrow_forwardWhat statement about sigma(lowercase) bonds is true? a)They are created by the overlap of p orbitals b)They only occur in single bonds c)It's the first covalent bond between two atoms in a molecular compound d)They only occur between hybridized orbitals e)none on the abovearrow_forward1.Complete the structure for anthracene, C14H10, by adding bonds and hydrogen atoms as necessary. 2.What type of hybrid orbitals are utilized by carbon in anthracene? 3. How many σ bonds and π bonds are there in an anthracene molecule? 4.How many valence electrons occupy σ‑bond orbitals, and how many occupy π‑bond orbitals?arrow_forward
- What is the total number of carbon atoms in this molecule? What is the total number of Hydrogen atoms in this molecule? i. How many sp3, sp2,and sp hybridized carbon atoms are in this compound?arrow_forwardWhat are the hybridizations of each carbon atom in the following molecules?arrow_forwardDetermine the hybridization on the underlined element in each compound. a. CH3CH2CH3 b. CHCH c. NH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY