
Concept explainers
a.
Interpretation: The hybridization state and geometry for each carbon atom in the following compound should be identified:
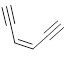
Concept Introduction: The concept of mixing two atomic orbitals that possess almost same energy resulting in formation of new hybridized orbitals that are suitable for pairing the electrons to form
b.
Interpretation: The hybridization state and geometry for each carbon atom in the following compound should be identified:
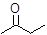
Concept Introduction: The concept of mixing two atomic orbitals that possess almost same energy resulting in formation of new hybridized orbitals that are suitable for pairing the electrons to form chemical bond is said to be hybridization.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
ORGANIC CHEMISTRY-PRINT COMPANION (LL)
- Write a condensed structural formula for the following compounds: a. b.arrow_forwardLipoic acid is required by many microorganisms for proper growth. As a disulfide, it functions in the living system by catalyzing certain oxidation reactions and is reduced in the process. Write the structure of the reduction product.arrow_forwardThe most common factor at the structural level that lowers the boiling point among compounds that are isomers is: a.)valence electrons b.)hybridization c.)hydrogen bridges d.)ramifications And why ?arrow_forward
- Compound X with the formula CH undergoes Hydration by specific product, then oxidation by H₂O₂, water and strong base to produce compound Y with the formula C₂H₁0. What might be true of X? 6 12 Select one: a. X might have one double bond and one triple bond O b. X might have one double bond and one ring c. X might have two double bonds and one ring O d. X might have three double bondsarrow_forward1. Give the IUPAC name for the following structures: a. b. C d. Karrow_forwardDraw the structure of all compounds that t the following descriptions.a. ve constitutional isomers having the molecular formula C4H8b. nine constitutional isomers having the molecular formula C7H16c. twelve constitutional isomers having the molecular formula C6H12 and containing one ringarrow_forward
- Jj.81.arrow_forwardCarbon compounds undergo A. Coordinate Covalent bonding B. Covalent bonding C. Intermediate bonding D. lonic bondingarrow_forwardWhich of the following best describe the unique characteristic of aromatic compounds? a. Their high reactivity b. Their high stability c. Their toxicity d. Their unique structure.arrow_forward
- Which of the following compounds is an ether? a. CH2CHO b. CH3OCH3 c. CH3COOH d. CH3CH2OHarrow_forwardWrite structural formulas for the following: a.o-ethylphenol b.m-chlorobenzoicacid c.3-methyl-3-phenylpentanearrow_forwardThe three-carbon diol used in antifreeze is It is nontoxic and is used as a moisturizing agent in foods. Oxidation of this substance within the liver produces pyruvic acid, which can be used by the body to supply energy. Give the structure of pyruvic acid.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





