
Concept explainers
(a)
Interpretation:
The
Concept Introduction:
Functional group:
Functional group contains particular atom or group of atoms bonded in a molecule which has specific chemical and physical properties. The functional group contains heteroatoms and a multiple bond in a compound.
(a)
Explanation of Solution
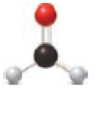
The ball and stick model is given below,
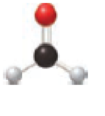
The black ball represents carbon atom, the white ball represents hydrogen atom and the red ball represents to oxygen atom. The functional group present in the given model is
The functional group present in model is given below,
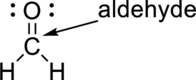
(b)
Interpretation
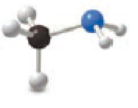
The functional group present in given ball and stick model has to be determined.
Concept Introduction:
Refer part (a)
(b)
Explanation of Solution
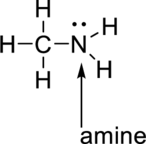
The black ball represents carbon atom, the white ball represents hydrogen atom and the
blue ball represents nitrogen group. The nitrogen atom is attached to the carbon atom. The functional group present in the given model is
The functional group present in given model is given below,
Want to see more full solutions like this?
Chapter 10 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Determining Intermolecular Forces in Organic Compounds Rank the following compounds in order of increasing strength of intermolecular forces: CH3CH2CH2CH2CH3 (pentane), CH3CH2CH2CH2OH (butan-1-ol), and CH3CH2CH2CHO (butanal).arrow_forward5. a) Draw two different structures with the molecular formula C2H6O. b) Name the functional group in each structure c)Which one will have the higher boiling point, and whyarrow_forward5. Build another hexane molecule and place it alongside the first one you built. (f) What types of intermolecular forces help hold hexane molecules to each other? (g) What aspect of the molecules' geometry makes these bonds effective?arrow_forward
- Out of CH3—NH2 and (CH3)3N, which one has higher boiling point?arrow_forwardClassify each of the following molecules as alcohol,aldehyde, ketone, carboxylic acid, amine, or ether.arrow_forwardThe chemical formula C4H10O results in four alcohols and three ethers for a total of seven structuralisomers. Draw pairs of structural formulas for these molecules that illustrate positional and functional isomerism on a sheet of paper. You will be drawing a total of four formulas. Label each pair as positional or functional.arrow_forward
- identify the functional group of each compound.arrow_forwardCircle and name ten different functional groups without repeating a functional group. ОН Но CI NH2arrow_forwardCH2-CH3 у. Type of functional group CH2-CH3 Common Name 1 CH2-CH3 Common Name 2 (Derived Name) z. Type of functional group IUPAC Derived Namearrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





