
Concept explainers
Answer the questions in Problem 10.35 for the
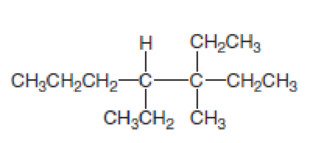
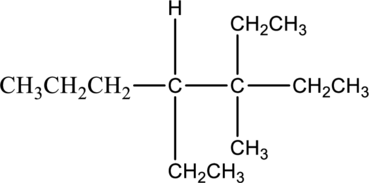
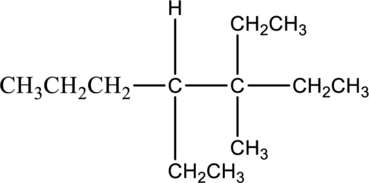
Answer the following questions about the alkane drawn below.
- a. Give the IUPAC name.
- b. Draw one constitutional isomer.
- c. Predict the solubility in water.
- d. Predict the solubility in an organic solvent.
- e. Write a balanced equation for complete combustion.
a.
Interpretation:
The IUPAC name of given compound has to be identified.
Concept Introduction:
IUPAC Nomenclature:
The IUPAC Nomenclature is a systematic writing name for organic compounds. The full form of IUPAC is known as International Union of Pure and Applied Chemistry.
There are some rules followed for writing IUPAC Nomenclature for alkane compound is given below,
- The longest chain in the compound is known as parent name which represent the number of carbon atom present in the continuous carbon chain. The root name for alkane compound is –ane. The single bonds are formed in alkane compounds.
- The suffix group denotes the functional group present in a molecule. The prefix group indicates the identity, location and number of substituents attached to the carbon compound.
- Then name and number the substituents present in compounds. Then use prefix di-to represent two groups, tri- refers to three groups and so on.
- Then numbers has to be separated by using commas and the letters from numbers has to be separated by using dashes.
Explanation of Solution
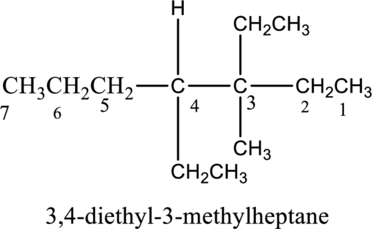
In this compound, the longest continuous chain has seven carbon atoms; so the parent name can be given as hept- and add suffix name –ane for alkane molecule. The parent name of given compound is heptane. Then number the substituents in alphabetical order. The ethyl group
The IUPAC name of compound is given below,
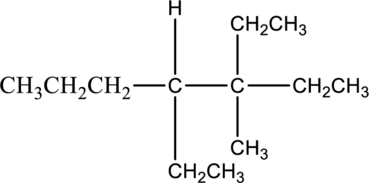
b.
Interpretation
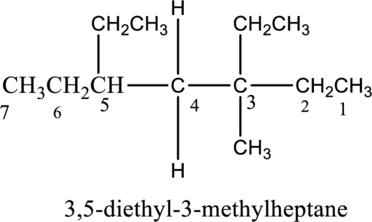
The constitutional isomer of 3, 4-diethyl-3-methylheptane has to be explained.
Concept Introduction:
Isomers:
Isomers contain two or more compounds in which the molecular formula of compounds is same but the atoms are arranged in different order. The arrangement of atoms will be in different manner.
Constitutional isomer:
The compounds have the same molecular formula but atoms are arranged in different connectivity is known as constitutional isomer.
Explanation of Solution
The isomer 3,4-diethyl-3-methylheptane has seven carbon atoms present in the longest chain. The two substituents ethyl groups
The constitutional isomers of 3,4-diethyl-3-methylheptane is given below,
c.
Interpretation:
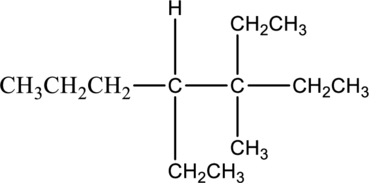
The solubility of given compound in water has to be predicted.
Concept Introduction:
Solubility:
The solubility is defined as the solid or liquid or gaseous substance which dissolves in suitable solvent. The nonpolar molecule has no separate positive and negative charge. Polar molecules have separate positive and negative charges.
Explanation of Solution
The compound 3,4-diethyl-3-methylheptane is insoluble in water. The water molecule is said to be polar molecules. It has both positive and negative charge. The given compound is a nonpolar alkane. The nonpolar alkanes cannot soluble in water molecule. Hence the given compound 3,4-diethyl-3-methylheptane is insoluble in water.
d.
Interpretation:
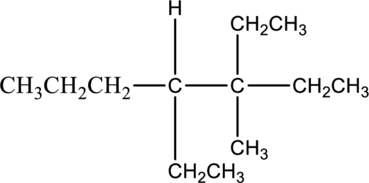
The solubility of given compound in organic compound has to be predicted.
Concept Introduction:
Solubility:
The solubility is defined as the solid or liquid or gaseous substance which dissolves in suitable solvent. The nonpolar molecule has no separate positive and negative charge. The Polar molecules have separate positive and negative charges.
Explanation of Solution
The given compound 3,4-diethyl-3-methylheptane is soluble in organic solvent. The reason for the solubility of the given compound, it belongs to nonpolar molecule. Nonpolar molecules cannot soluble in polar molecule. The nonpolar molecule has no separate charges. Hence given molecule 3,4-diethyl-3-methylheptane is soluble in organic solvent.
e.
Interpretation:
The balanced equation for complete combustion of given compound has to be written.
Concept Introduction:
Combustion reaction:
The hydrocarbon compound reacts with oxygen to form carbon dioxide and water as product is known as hydrocarbon combustion reaction. It is an exothermic reaction. The hydrocarbon compounds are generally organic compounds contain carbon and hydrogen element. The compounds such as alkanes, alkenes, alkynes and aromatic compounds.
The general equation is given as,
Where,
- x represents the number of carbon atoms present in the hydrocarbon
- y represents the number of carbon atoms present in the hydrocarbon
- N represents the number of oxygen atoms requires in the hydrocarbon combustion reaction
Chemical equation:
In chemical equation, the substance reacts (reactant) to give products. The number of atoms present in reactant side should be equal to the number of atom present in the product side.
Explanation of Solution
The compound 3,4-diethyl-3-methylheptane
The chemical reaction for complete combustion of ethane is given below,
In reactant side, it has twenty four carbon atoms and fifty two hydrogen and seventy four atoms oxygen atoms is present. In product side there are twenty four carbon atoms and forty eight oxygen atoms present in carbon dioxide. The fifty six hydrogen atoms and twenty six oxygen atoms are present in water molecule. In this reaction the molecule
The balanced equation for complete combustion reaction of 3-ethyl-4,4-dimethylheptane is given below,
Want to see more full solutions like this?
Chapter 10 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Use the following information to answer the next question. Nonane is a saturated hydrocarbon with the empirical formula C9H20- This hydrocarbon can exhibit 35 different isomers. One of these isomers is given below. H. H H. H H. CH 3 H. H. C. C C. C. C. H - H. H. H. H. H. H. The IUPAC name of this isomer isarrow_forward1. How many total covalent bonds are present in the compound? (Please refer to the first image attached.) A. 8 B. 10 C. 12 D. 16 2. What is the correct IUPAC name for this compound? (Please refer to the second image attached.) A. 2,5,5-Trimethylhexane B. 2,2,5-Trimethylhexane C. 2,2,5-methylhexane D. 2,5,5-methylhexane 3. Which of the following is a tertiary alcohol? A. 2-Pentanol B. 2-Methyl-2-butanol C. 1-Butanol D. 2-Methyl-1-pentanolarrow_forward36. Assign the IUPAC name of the structure below. A. 1-Bromopentan-3-one B. 3-Bromocyclopentanone C. 3-bromocylopentanal D. Both B and C 37. Assign the IUPAC name of the structure below. (Hint: Check the order of precedence) H A. Pentan-1,4-dione B. 1,4-Pentanaldehyde C. 4-Oxopentanal D. None of the above 38. Assign the IUPAC name of the structure below. (Hint: Check the order of precedence) A. 4-hydroxy-3-methylcyclohexanone B. 2-Methyl-4-oxophenol C. 2-Methyl-3-hydroxycylohexanone D. 4-phenyl-2-methylhexanonearrow_forward
- 1. The following are isomers of each other I. pentane II. 2,2,-dimethylpropane III. 2-methylbutaneIV. 2,3-dimethylhexane A. Alkanes are nonpolar. B. Alkanes are non-flammable. C. Every carbon in an alkane has four bonds. D. Alkanes contain only C and H atoms. E. Alkanes do not contain carbon-carbon double or triple bonds. 1b. Which of these is a weak base? A. NaOH B. H3PO4 C. CH3COOH D. NaHCO3 E. NH4NO3 1c. Which of these pairs are formed when NaHCO3 (baking soda) is dissolved in water? NaHCO3 → ______ + ______. A. NaOH + CO2 B. Na+ +HCO3- C. NaH+ +CO3- D. Na + HCO3 E. NaH- + CO3+ 1D. Which of these statements about water is(are) incorrect? A. It has a pH of 7.0 at ambient temperatures. B. It contains no ions. C. It is neutral. D. It can act as a proton donor. E. It can act as a proton acceptor.arrow_forwardn an alkene with four carbon atoms and only one carbon-carbon double bond, how many hydrogen atoms would it contain? a. 4 b. 6 c. 8 d. 10 e. 12arrow_forward2. Identify the configuration of the alkene present in the compound. You will need to redraw the structure and insert your answer next to the carbon double bond. H. Brarrow_forward
- 6. Any organic compound that contains a benzene ring or similar feature. O a. addition reaction Ob. aliphatic compound Oc. alkene d. alkyne Oe. aromatic hydrocarbon Of. hydration g. hydrogenation h. monomer Oi. phenyl group j. polycyclic aromatic hydrocarbon k. polymer I. unsaturated hydrocarbonarrow_forward2. Draw the correct structure for each given name below. a. 1-butyl-3-ethyl-2-propylcycloheptane b. cis-1,3-diisopropylcyclohexane c. cyclobutylcyclohexanearrow_forwardAlkenes are not soluble in water because they are a. polar b. hydrophilic c. hydrophobic d. less dense than waterarrow_forward
- 1. Identify the SYSTEMATIC name of the aliphatic hydrocarbon a. 1-pentenyicyclopentane b. 1-cyclopentylpent-2-yne c. 1-pentenecyclopent-2-yne d. 1-pentenyicyclopentane 2. Identify the SYSTEMATIC name of the aliphatic hydrocarbon a. Trans-1,2-propylcyclopropane b. Trans-1,2-diisopropylcyclopropane c. Cis-1,2-propylcyclopropane d. Cis-1,2-diisopropylcyclopropane 3. Identify the SYSTEMATIC name of the aliphatic hydrocarbon 3,10-dimethyl-2-decacen-6-yne 3,10-dimethyl-2-dodocen-6-yne 3,10-dimethyl-10-decacen-6-yne d. 3,10-dimethyl-10-dodocen-6-yne a. b. C. 4. Identify which type of isomer the following structures represent HO a. Skeletal Isomer b. E/Z isomer c. Cis/Trans isomer OH OH d. Positional isomer 5. Identify which type of isomer the following structures represent a. Skeletal Isomer Br b. E/Z isomer c Cis/Trans isomer Br d. Positional isomerarrow_forward1. What is the IUPAC name of the compound below? (Please refer to the image attached.) A. 2,2,5-pentylcyclohexane B. 1-Cyclohexyl-2,2,5-trimethylhexane C. 1-Cyclohexyl-1,1,4-trimethylpentane D. 2-Cyclohexyl-2,5-dimethylhexane E. None of the choices 2. The –ene ending suffix of alkene indicates that there is a presence of carbon-carbon _______ bonds in the organic compound. A. Single B. Double C. Triple D. Quadruple 3. It is an acyclic unsaturated hydrocarbon that contains one or more carbon–carbon triple bonds: A. Alkene B. Cycloalkene C. Alkyne D. Cycloalkynearrow_forwardConsider the structure in the attached picture: 1. the carbon where OH is attached is classified as a: a. primary b. secondary c. tertiary 2. When the compound is reacted with a dehydrating reagent like H2SO4, the compound formed will be a/an . a. alkane b. alkene c. aldehyde d. ketone 3. The product formed from its dehydration using sulfuric acid is _______. a. butane b. 2-butene c. butanal d. 2-butanonearrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
