
Concept explainers
Answer the following questions about the
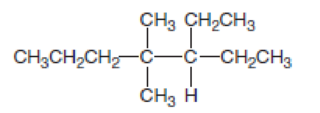
- a. Give the IUPAC name.
- b. Draw one constitutional isomer.
- c. Predict the solubility in water.
- d. Predict the solubility in an organic solvent.
- e. Write a balanced equation for complete combustion.
a.
Interpretation:
The IUPAC name of given compound has to be identified.
Concept Introduction:
IUPAC Nomenclature of alkane compounds.
IUPAC nomenclature is a system of writing a name for organic compounds. The full form of IUPAC name is International Union of Pure and Applied chemistry.
There are certain rules followed for writing IUPAC name:
- The longest continuous carbon chain present in compound can be given as parent name. Then add the prefix and suffix name to the parent name. The suffix group represents the functional group present in a molecule. The prefix name explains about the identity, location, and number of substituents present in compound.
- The functional groups or alkyl groups have to be determined. The numbering of carbon starts from left side and ends at right side.
- The suffix group –ane represents alkane molecule. The prefix group represents the number of carbon atom present in the longest carbon chain. Then the substituent group attached to the longest chain can be numbered in alphabetical order. The prefix name can be given, depends on the number of substituent present in molecule.
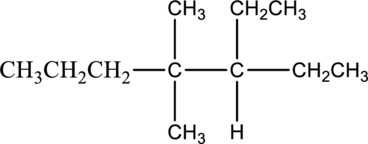
Explanation of Solution
In this compound the longest chain has seven carbon atoms. The parent name of given compound is heptane. Then number the substituents in alphabetical order. The ethyl group
The IUPAC name of compound is given below,
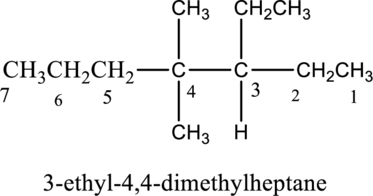
b.
Interpretation
The one constitutional isomer for the given compound has to be drawn.
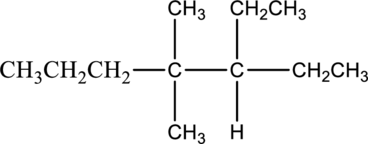
Concept Introduction:
Isomers:
Isomers contain two or more compounds in which the molecular formula of compounds is same but the atoms are arranged in different order. The arrangement of atoms will be in different manner.
Constitutional isomer:
The compounds have the same molecular formula but atoms are arranged in different connectivity is known as constitutional isomer.
Explanation of Solution
The isomer 3-ethyl-4,5-dimethylheptane has seven carbon atoms present in the longest chain. The two substituents methyl groups
The constitutional isomers of
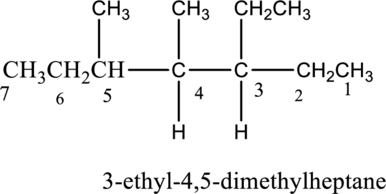
c.
Interpretation:
The solubility of given compound in water has to be predicted.
Concept Introduction:
Solubility:
The solubility is defined as the solid or liquid or gaseous substance which dissolves in suitable solvent. The solubility also depends upon polar or nonpolar molecule. The nonpolar molecule has no separate positive and negative charge. Polar molecules have separate positive and negative charges.
Explanation of Solution
The 3-ethyl-4,4-dimethylheptane is insoluble in water. The water molecule is said to be polar molecules. It has both positive and negative charge. The given compound is a nonpolar alkane. The nonpolar alkanes cannot soluble in water molecule. Hence the given compound 3-ethyl-4,4-dimethylheptane is insoluble in water.
d.
Interpretation:
The solubility of given compound in organic compound has to be predicted.
Concept Introduction:
Refer part: c.
Explanation of Solution
The given compound is soluble in organic solvent. The reason for the solubility of the given compound, it belongs to nonpolar molecule. Nonpolar molecules cannot soluble in polar molecule. The nonpolar molecule has no separate charges. Hence given molecule
3-ethyl-4,4-dimethylheptane is soluble in organic solvent.
e.
Interpretation:
The balanced equation for complete combustion of given compound 3-ethyl-4, 4-dimethylheptane has to be written.
Concept Introduction:
Combustion reaction:
The hydrocarbon compound reacts with oxygen to form carbon dioxide and water as product is known as hydrocarbon combustion reaction. It is an exothermic reaction. The hydrocarbon compounds are generally organic compounds contain carbon and hydrogen element. The compounds such as alkanes, alkenes, alkynes and aromatic compounds.
The general equation is given as,
Where,
- x represents the number of carbon atoms present in the hydrocarbon
- y represents the number of carbon atoms present in the hydrocarbon
- N represents the number of oxygen atoms requires in the hydrocarbon combustion reaction
Chemical equation:
In chemical equation, the substance reacts (reactant) to give products. The number of atoms present in reactant side should be equal to the number of atom present in the product side.
Explanation of Solution
The compound 3-ethyl-4, 4-dimethylheptane
The chemical reaction for complete combustion of ethane is given below,
In reactant side the molecules of
The balanced equation for complete combustion reaction of 3-ethyl-4,4-dimethylheptane is given below,
Want to see more full solutions like this?
Chapter 10 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- 1. In dash-wedge-line structure, the dashes represent the: * A. bonds in the plane of the page. B. bonds away from observer. C. bonds towards observer. D. bonds in the plane of the paper. 2. Which of the following is an ACYCLIC SATURATED hydrocarbon? * A. 1-Ethyl-3-methylcyclopentane B. 3-Ethyl-3-methylpentane C. 1,3,5,7-Octatetraene D. 5-Methyl-1,3-cyclopentadiene 3. Which of the following is a CYCLIC SATURATED hydrocarbon? * A. 3-Ethyl-4-methylcyclohexene B. 3,5-Diethyloctane C. 2,2-Dimethyl-3-heptyne D. 3-Butyl-1,1-dimethyl-5-propylcyclohexanearrow_forwardChemistry 3. Discuss the physical properties of aliphatic hydrocarbons based on: a. Boiling and melting point of alkanes (as affected by increasing number of carbon atoms) b. Alkane solubility (as affected by increasing number of carbon atoms) c. Combustion Reactions of Alkanes (Example of Reaction and Use)arrow_forward6. Any organic compound that contains a benzene ring or similar feature. O a. addition reaction Ob. aliphatic compound Oc. alkene d. alkyne Oe. aromatic hydrocarbon Of. hydration g. hydrogenation h. monomer Oi. phenyl group j. polycyclic aromatic hydrocarbon k. polymer I. unsaturated hydrocarbonarrow_forward
- 3. The reaction of water with alkene to produce an alcohol is a/an C. Combustion D. Addition reaction. A. Saponification B. Condensationarrow_forwardPlease do them all not just 1 plsarrow_forward1. The following are isomers of each other I. pentane II. 2,2,-dimethylpropane III. 2-methylbutaneIV. 2,3-dimethylhexane A. Alkanes are nonpolar. B. Alkanes are non-flammable. C. Every carbon in an alkane has four bonds. D. Alkanes contain only C and H atoms. E. Alkanes do not contain carbon-carbon double or triple bonds. 1b. Which of these is a weak base? A. NaOH B. H3PO4 C. CH3COOH D. NaHCO3 E. NH4NO3 1c. Which of these pairs are formed when NaHCO3 (baking soda) is dissolved in water? NaHCO3 → ______ + ______. A. NaOH + CO2 B. Na+ +HCO3- C. NaH+ +CO3- D. Na + HCO3 E. NaH- + CO3+ 1D. Which of these statements about water is(are) incorrect? A. It has a pH of 7.0 at ambient temperatures. B. It contains no ions. C. It is neutral. D. It can act as a proton donor. E. It can act as a proton acceptor.arrow_forward
- 5. Draw line structures to correspond with the following names a. 3-isopropylpentane b. cis-1,2-dichlorocyclopentane C. trans-1-hydroxy-2-methylcyclopropane (aka trans-2-methylcyclopropanol)arrow_forwardPlease explain the chosen letter. Acetone and acetaldehyde are a. Position isomers b. Functional isomers c. Not isomers d. Chain isomersarrow_forward2. Determine whether cis-trans isomerism is possible for each of the following cycloalkanes. If so, then draw structural formulas for the cis and trans isomers.. a. Methylcyclohexane b. 1,1-Dimethylcyclohexane c. 1,3-Dimethylcyclobutane d. 1-Ethyl-2-methylcyclobutanearrow_forward
- Please don't provide handwritten solutionarrow_forwardMultiple Choice questions Organic Chemistryarrow_forward1. Which of the following situations meet the "bonding requirement" for carbon atoms. * A. Two single bonds and a double bond. B. A single bond and two double bonds. C. Three single bonds and a triple bond. D. A double bond and a triple bond. 2. What is the line-angle formula for the expanded structural formula below? (Please refer to the image attached.) 3. Which of the following is CORRECT about organic compounds? A. They generally make good electrolytes. B. They generally exhibit ionic bonding. C. They have lower melting points compared to inorganic compounds. D. They are more likely to be soluble in water compared to inorganic compounds.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

