
Concept explainers
(a)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(b)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
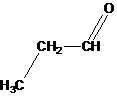
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(c)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(d)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(e)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
(f)
Interpretation:
The bond angle of each atom of carbon, nitrogen and oxygen in given structural formula should be predicted.
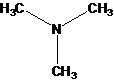
Concept Introduction:
VSEPR model is useful to predict bond angles occurred between atoms of a molecule. The bond angle of atom is a point where two atoms are joined together. The valence-shell electron-pair repulsion model is full form of VSEPR model.
Trending nowThis is a popular solution!

Chapter 10 Solutions
Introduction to General, Organic and Biochemistry
- What is the difference between the hybridization of carbon atoms' valence orbitals in saturated and unsaturated hydrocarbons?arrow_forwardBased from the figures below, which of the following statements is correct?I. A is an skeletal isomer of D while B and C are positional isomers.II. B and C are alkynes with six Carbons but their structures are different.III. All of the above have the possibility of a cis-trans isomerism.arrow_forwardDraw a complete structure for a molecule with the molecular formula C3H8Oarrow_forward
- Draw all the possible structural isomers with the molecular formula C6H14, Name themarrow_forward1. The correct structure that below represents CH3C(CH3)2CH3 is ___________ . 2. Write the name of each structure given above. (same picture used for number 1 and 2)arrow_forwardProvide a complete, labeled orbital diagram for 1-fluoro-butadiene (FHC=CH-CH=CH2). Indicate all bond types (including which orbitals have overlapped to form each bond) and bond angles.arrow_forward
- Draw all possible structure(s) and give the IUPAC systematic name(s) of an alkane or cycloalkane with the formula C8H18 that has only primary hydrogen atoms...arrow_forwardDraw 10 isomers (skeletal structure) with the formula C8H18arrow_forwardDescribe the types of bonds (σ or π) in acetone, (CH3)2CO.arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning



