
GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
11th Edition
ISBN: 9780134193601
Author: Petrucci
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 42E
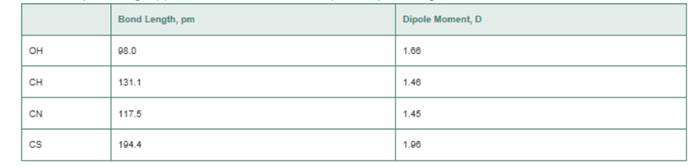
Use a cross-base arrow
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculate the partial charges on the hydrogen and halogen atoms in HF, HBr, and HI
If energy is required to form monatomic ions from metals and nonmetals, why do ionic compounds exist?
Assume imaginary compound XY has a polar covalent bond. How does its charge distribution differ from that of XX molecules?
Chapter 10 Solutions
GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
Ch. 10 - Write Lewis symbols for the following atoms. (a)...Ch. 10 - Write Lewis symbols for the following ions. (a)...Ch. 10 - Write plausible Lewis structures for the following...Ch. 10 - Each of the following molecules contains at least...Ch. 10 - By means of Lewis structures, represent bonding...Ch. 10 - Which of the following have Lewis structures that...Ch. 10 - Prob. 7ECh. 10 - Suggest reasons why the following do not exist as...Ch. 10 - Describe what is wrong with each of the following...Ch. 10 - Describe what is wrong with each of the following...
Ch. 10 - Prob. 11ECh. 10 - Indicate what is wrong with each of the following...Ch. 10 - Write Lewis structures for the following ionic...Ch. 10 - Under appropriate conditions, both hydrogen and...Ch. 10 - Derive the correct formulas for the following...Ch. 10 - Each of the following ionic compounds consists of...Ch. 10 - Assign formal charges to each of the atoms in the...Ch. 10 - Assign formal charges to each of the atoms in the...Ch. 10 - Both oxidation state and formal charge involve...Ch. 10 - Prob. 20ECh. 10 - Prob. 21ECh. 10 - Assign formal charges to the atoms in the...Ch. 10 - Prob. 23ECh. 10 - Show that the idea of minimizing the formal...Ch. 10 - Write acceptable Lewis structures for the...Ch. 10 - Two molecules that have the same formulas but...Ch. 10 - The following polyatomic anions involve covalent...Ch. 10 - Represent the following ionic compounds by Lewis...Ch. 10 - Write a plausible Lewis structure for...Ch. 10 - Prob. 30ECh. 10 - Write Lewis structures for the molecules...Ch. 10 - Write Lewis structures for the molecules...Ch. 10 - Write Lewis structures for the molecules...Ch. 10 - Write Lewis structures for the molecules...Ch. 10 - Identify the main group that the element X belongs...Ch. 10 - Prob. 36ECh. 10 - Use your knowledge of electronegativities, but do...Ch. 10 - Which of the blowing molecules would you expect to...Ch. 10 - What is the percent ionic character of each of the...Ch. 10 - Prob. 40ECh. 10 - Prob. 41ECh. 10 - Use a cross-base arrow () to represent the...Ch. 10 - Which electrostatic potential map corresponds to...Ch. 10 - Prob. 44ECh. 10 - Two electrostatic potential maps are shown, one...Ch. 10 - Prob. 46ECh. 10 - Prob. 47ECh. 10 - Which of the following species requires a...Ch. 10 - Dinitrogen oxide (nitrous oxide, or "laughing...Ch. 10 - The Lewis structure of nitric acid, HONO2, is a...Ch. 10 - Draw Lewis structures for the following species,...Ch. 10 - Draw Lewis structures for the following species,...Ch. 10 - Write plausible Lewis structures for the following...Ch. 10 - Write plausible Lewis structures for the following...Ch. 10 - Which of the following species would you expect to...Ch. 10 - Write a plausible Lewis structure for NO2 , and...Ch. 10 - In which of the following species is it necessary...Ch. 10 - Prob. 58ECh. 10 - Use VSEPR theory to predict the geometric shapes...Ch. 10 - Use VSEPR theory to predict the geometric shapes...Ch. 10 - Each of the following is either linear, angular...Ch. 10 - Predict the geometric shapes of (a) CO ; (b)...Ch. 10 - One of the following ions has a trigonal-planer...Ch. 10 - Two of the following have the same shape. Which...Ch. 10 - Prob. 65ECh. 10 - Sketch the probable geometric shape of molecule of...Ch. 10 - Use the VSEPR theory to predict the shapes of the...Ch. 10 - Use the VSEPR theory to predict the shape of (a)...Ch. 10 - The molecular shape of BF2 is planar (see Table...Ch. 10 - Explain why it is not necessary to find the Lewis...Ch. 10 - Comment on the similarities and differences in the...Ch. 10 - Comment on the similarities and differences in the...Ch. 10 - Draw a plausible Lewis structure for the following...Ch. 10 - Draw a plausible Lewis structure for the following...Ch. 10 - Sketch the propyne molecule, CH2CCH. Indicate the...Ch. 10 - Sketch the propene molecule, CH2CHCH2. Indicate...Ch. 10 - Lactic acid has the formula CH2CH(OH)COOH. Sketch...Ch. 10 - Levulinic acid has the formula CH2(CO)CH2CH2COOH....Ch. 10 - Prob. 79ECh. 10 - Prob. 80ECh. 10 - Predict the shapes of the following molecules, and...Ch. 10 - Which of the blowing molecules would you expect to...Ch. 10 - The molecule H2O2 has a resultant dipole moment of...Ch. 10 - Prob. 84ECh. 10 - Without referring to tables in the text, indicate...Ch. 10 - Estimate the lengths of the blowing bonds and...Ch. 10 - A relationship between bond lengths and...Ch. 10 - In which of the following molecules would you...Ch. 10 - Prob. 89ECh. 10 - Prob. 90ECh. 10 - A reaction involved in the formation of ozone the...Ch. 10 - Use data from Table 10.3, but without performing...Ch. 10 - Use data from Table 10.3 to estimate the enthalpy...Ch. 10 - One of the chemical reactions that occurs in the...Ch. 10 - Estimate the standard enthalpies of formation at...Ch. 10 - Prob. 96ECh. 10 - Use bond energies from Table 10.3 to estimate rH...Ch. 10 - Equations (1) end (2) can be combined to yield the...Ch. 10 - One reaction involved in the sequence of reactions...Ch. 10 - Prob. 100ECh. 10 - Given the bond-dissociation energies:...Ch. 10 - Prob. 102IAECh. 10 - Prob. 103IAECh. 10 - Prob. 104IAECh. 10 - Prob. 105IAECh. 10 - Draw Lewis structures for two different molecules...Ch. 10 - Sodium azide, NaN2 is the nitrogen gas-forming...Ch. 10 - Prob. 108IAECh. 10 - Prob. 109IAECh. 10 - A few years ago the synthesis of a salt containing...Ch. 10 - Prob. 111IAECh. 10 - In certain polar solvents, PCI, undergoes an...Ch. 10 - Prob. 113IAECh. 10 - Prob. 114IAECh. 10 - Use the VSEPR theory to predict a probable shape...Ch. 10 - The standard enthalpy of formation of...Ch. 10 - Prob. 117IAECh. 10 - Prob. 118IAECh. 10 - Prob. 119IAECh. 10 - R. S. Mulliken proposed that the electronegativity...Ch. 10 - When molten sulfur reacts with chlorine gas, a...Ch. 10 - Hydrogen azide, HN2 , can exist in two forms. One...Ch. 10 - Prob. 123IAECh. 10 - Prob. 124IAECh. 10 - Prob. 125IAECh. 10 - One of the allotropes of sulfur is a ring of eight...Ch. 10 - One of the allotropes of phosphorus consists of...Ch. 10 - In this problem, we examine the basis of three...Ch. 10 - Prob. 129FPCh. 10 - Prob. 130FPCh. 10 - Prob. 131SAECh. 10 - Briefly describe each of the following ideas: (a)...Ch. 10 - Explain the important distinctions between (a)...Ch. 10 - Prob. 134SAECh. 10 - The formal charges on the O atoms in the ion...Ch. 10 - Which molecule is nonlinear?...Ch. 10 - Which molecule is nonpolar?...Ch. 10 - The highest bond-dissociation energy is found in...Ch. 10 - The greatest bond length is found in...Ch. 10 - Draw plausible Lewis structures for the blowing...Ch. 10 - Predict the shapes of the following...Ch. 10 - Which of the following ionic compounds is composed...Ch. 10 - Which of the following molecules does not obey the...Ch. 10 - Which of the following molecules has no polar...Ch. 10 - The electron-group geometry of H2O is (a)...Ch. 10 - For each of the following compounds, give the...Ch. 10 - Use bond enthalpies from Table 10.3 to determine...Ch. 10 - Prob. 148SAECh. 10 - Prob. 149SAECh. 10 - What is the VSEPR theory? On what physical basis...Ch. 10 - Prob. 151SAECh. 10 - Prob. 152SAECh. 10 - Prob. 153SAECh. 10 - Prob. 154SAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Think of forming an ionic compound as three steps (this is a simplification, as with all models): (I) removing an electron from the metal; (2) adding an electron to the nonmetal; and (3) allowing the metal cation and nonmetal anion to come together. a. What is the sign of the energy change for each of these three processes? b. In general, what is the sign of the sum of the first two processes? Use examples to support your answer. c. What must be the sign of the sum of the three process d. Given your answer to part c, why do ionic bonds occur? e. Given your above explanations, why is NaCl stable but not Na2Cl? NaCl2? What about MgO compared to MgO2? Mg2O?arrow_forwardThe equation for the combustion of gaseous methanol is 2 CH3OH(g) + 3 O2(g) 2 CO2(g) + 4 H2O(g) (a) Using the bond dissociation enthalpies in Table 8.8, estimate the enthalpy change for this reaction. What is the enthalpy of combustion of one mole of gaseous methanol? (b) Compare your answer in part (a) with the value of tHcalculated using enthalpies of formation data.arrow_forwardWrite all resonance structures of chlorobenzene, C6H5Cl, a molecule with the same cyclic structure as benzene. In all structures, keep the CCl bond as a single bond. Which resonance structures are the most important?arrow_forward
- Define the term lattice energy. Why, energetically, do ionic compounds form? Fig. 3-8 illustrates the energy changes involved in the formation of MgO(s) and NaF(s). Why is the lattice energy of MgO(s) so different from that of NaF(s)? The magnesium oxide is composed of Mg2+ and O2 ions. Energetically, why does Mg2+O2 form and not Mg+O? Why doesnt Mg3+O3 form?arrow_forwardIn which of the following compounds does hydrogen bear a partial negative charge: (a) CH4, (b) NH3, (c) H2O, (d) SiH4 or (e) H2S?arrow_forwardWhich of the compounds has higher lattice energy? Explain your answer. i) Na2O or Na2Farrow_forward
- Place the following in order of decreasing magnitude of lattice energy. Explain your answer. K2O Rb2S Li2Oarrow_forwardThe below atoms will form a compound. Provide the Lewis dot structures of the formed compounds and the name of each formed compound. The compounds may be neutral or ionic. The compounds may be made up of more than two atoms. a) K, Cl b) Br, O c) N, P d) B, F e) F, Sarrow_forwardUse condensed electron configuration and Lewis symbols to depict the formation of Mg2+ and Cl- ions from the atoms, and give the formula of the compound formed.arrow_forward
- Which polyatomic ion has shorter NO bond lengths NO21– OR NO21+? How would i show this using lewis structures?arrow_forwardCompare the bond enthalpy of F2 with the energy change for the following process: F2(g) → F+(g) + F-(g) Which is the preferred dissociation for F2, energetically speaking?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY