
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 70P
Phenolphthalein is an acid–base indicator. In solutions of pH < 8.5, it is colorless; in solutions of pH > 8.5, it is deep red-purple. Account for the change in color.
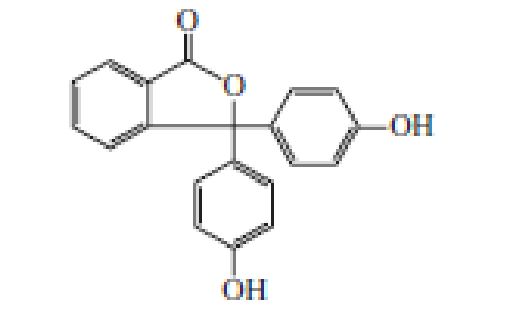
phenolphthalein
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Phenolphthalein is an acid–base indicator. In solutions of pH 6 8.5, it is colorless; in solutions of pH 7 8.5, it is deep red-purple. Account for thechange in color.
Phenolphthalein indicator is a weak acid with Ka = 10-9 mol/dm-3. It is colourless while its conjugate base is pink in solution.
Calculate the ratio of the number of pink particles to the number of colourless particles of this indicator in a solution of pH = 9
For strong acid and weak base titration,methyl orange is a suitable indicator whereas phenolphthalein is not.Justify the statement.Can equivalence point be determined experimentally?
Chapter 10 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 10.1 - Prob. 1PCh. 10.2 - What would distinguish the mass spectrum of...Ch. 10.2 - Prob. 3PCh. 10.3 - Prob. 5PCh. 10.3 - Suggest possible molecular formulas for a compound...Ch. 10.3 - Prob. 7PCh. 10.4 - Prob. 8PCh. 10.4 - Prob. 9PCh. 10.5 - Prob. 10PCh. 10.5 - Prob. 11P
Ch. 10.6 - Identify the ketone responsible for the mass...Ch. 10.6 - Prob. 13PCh. 10.8 - Prob. 14PCh. 10.8 - Prob. 15PCh. 10.12 - Which will occur at a larger wavenumber: a. a C :...Ch. 10.13 - Which will occur at a larger wavenumber: a. the C...Ch. 10.13 - Prob. 18PCh. 10.13 - Prob. 19PCh. 10.13 - Which will show an O 8 H stretch at a larger...Ch. 10.14 - Prob. 21PCh. 10.14 - Prob. 22PCh. 10.15 - Prob. 23PCh. 10.15 - Prob. 24PCh. 10.17 - Prob. 25PCh. 10.18 - Prob. 26PCh. 10.18 - Prob. 27PCh. 10.19 - Prob. 28PCh. 10.19 - Prob. 29PCh. 10.22 - How many signals would you expect to see in the 1H...Ch. 10.22 - Prob. 31PCh. 10.22 - Prob. 32PCh. 10.23 - Where would you expect to find the 1H NMR signal...Ch. 10.24 - Prob. 34PCh. 10.25 - Prob. 35PCh. 10.25 - Prob. 36PCh. 10.25 - Prob. 37PCh. 10.26 - Prob. 38PCh. 10.26 - Which of the following compounds is responsible...Ch. 10.27 - Prob. 40PCh. 10.27 - Prob. 41PCh. 10.27 - The 1H NMR spectra of two carboxylic acids with...Ch. 10.28 - Prob. 43PCh. 10.28 - Prob. 44PCh. 10.28 - Prob. 45PCh. 10.28 - Describe the 1H NMR spectrum you would expect for...Ch. 10.28 - Identify the compound with molecular formula...Ch. 10.29 - Prob. 48PCh. 10.29 - Prob. 49PCh. 10.29 - Identify the compound with a molecular formula of...Ch. 10 - In the mass spectrum of the following compounds,...Ch. 10 - For each of the following pairs of compounds,...Ch. 10 - Draw the structure of a saturated hydrocarbon that...Ch. 10 - Prob. 54PCh. 10 - Prob. 55PCh. 10 - How could you use UV spectroscopy to distinguish...Ch. 10 - Prob. 57PCh. 10 - Predict the relative intensities of the molecular...Ch. 10 - Prob. 59PCh. 10 - List the following compounds in order from highest...Ch. 10 - How can 1H NMR be used to prove that the addition...Ch. 10 - There are four esters with molecular formula...Ch. 10 - Prob. 63PCh. 10 - Prob. 64PCh. 10 - Each of the IR spectra presented here is...Ch. 10 - Prob. 66PCh. 10 - Five compounds are shown for each of the following...Ch. 10 - Prob. 68PCh. 10 - Prob. 69PCh. 10 - Phenolphthalein is an acidbase indicator. In...Ch. 10 - Which one of the following five compounds produced...Ch. 10 - Prob. 72PCh. 10 - Prob. 73PCh. 10 - Prob. 74PCh. 10 - How could 1H NMR distinguish between the compounds...Ch. 10 - Prob. 76PCh. 10 - Prob. 77PCh. 10 - The 1H NMR spectra of three isomers with molecular...Ch. 10 - The 1H NMR spectra of three isomers with molecular...Ch. 10 - Identify the following compounds. (Relative...Ch. 10 - An alkyl halide reacts with an alkoxide ion to...Ch. 10 - Determine the structure of a compound with...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A 250 cm3 volumetric flask contains exactly 200,0 cm3 of a 0,025 mol.dm3sulphuric acid solution. Thereafter ten (10) sodium hydroxide pellets, eachof mass 0,1 g are dropped into the flask. After the pellets have dissolvedcompletely, the flask is topped to the 250 cm3 mark with water and thecontents are thoroughly homogenised. Determine the pH of the resultingsolution.arrow_forward) A truck driver carrying a load of lead nitrate (Pb(NO3)2) lost control of his semi- truck after hitting a patch of ice and crashed the truck into Blue Lake, which was right next to the highway. Despite the best efforts of the emergency workers, several of the crates containing lead nitrate were damaged, and the highly soluble compound dissolved immediately. Assuming the lake is initially at circumneutral pH (7), and that the spill resulted the in a total lead concentration of 10-3 M within the lake. Based on the following information, will PbO(s) precipitate out of Blue Lake. Assume all lead nitrate dissociates into Pb+2 and NO3-, no other sources of lead exist in the lake and that no other reactions besides the equations shown below occur. PbO(s) + 2H+ ⇌ PbO(s) + H+ ⇌ PbO(s) + H2O ⇌ PbO(s) + 2H2O ⇌ Pb2+ + H2O PbOH+ + H2O Pb(OH)2o Pb(OH)3- + H+ logKs0/ksp =14 logK1=3.4 logK2=-0.5 logK3=-12arrow_forwardPhenolphthalein is an acid–base indicator. In solutions of pH < 8.5, it is colorless; in solutions of pH > 8.5, it is deep red-purple. Account for the change in color.arrow_forward
- The indicator phenolphthalein may be regarded as a weak monobasic acid. Explain briefly why the indicator is coloured pink when the solution in which it is present has excess of alkali.arrow_forwardCalculate the volume in milliliters of 0.368 M KOH necessary to titrate 0.0209 moles of acetic acid (HOAc, HC2H3O2) to a phenolphthalein end-point. Report the answer with a precision of two decimal places.arrow_forwardA professor prepares a buffer solution that they need for the purification of protein from human cell lysates. They did mix weak acid and conjugate base and obtained the initial solution, which is characterised by these parameters. Final volume: 200mltotal buffer compound concentration: 150mMIntitial concentration weak acid: 0.03MInitial concentration conjugate base: 0.12MInitial pH 6.9buffer compound pka 6.3arrow_forward
- A microbiologist is preparing a medium on which to cultureE. colibacteria. She buffers the medium at pH 7.00 to minimize the effect of acid-producing fermentation. What volumes ofequimolar aqueous solutions of K₂HPO₄and KH₂PO₄must shecombine to make 100. mL of the pH 7.00 buffer?arrow_forwardAn acid-base indicator is usually a weak acid with a characteristic color in the protonated and deprotonated forms. Because bromocresol green is an acid, it is convenient to represent its rather complex formula as HBCG. HBCG ionizes in water according to the following equation: HBCG + H2O ⇌ BCG- + H3O+ (yellow) (blue) a. Write the Ka expression for bromocresol green based on the equation above. b. When [BCG-] = [HBCG], then show that the expression simplifies to Ka = [H3O+]. If you know the pH of the solution, then the [H3O+] and Ka can be determined. c. What would be the color of the solution if there were equal concentrations of HBCG and BCG-?arrow_forwardHow to write the methodology? I have a practical of the MEASUREMENT USING PH METER, I don't know how to write the methodology. Here is the introduction: A pH meter is an electronic device mainly used for qualitative measurement, for thedetermination of acid and the basic value of a solution. Generally, it measures thehydrogen ion concentration/activity [H+] in a solution. It is denoted as:pH = - log10 [H+]In a pure water solution, the concentration of [H+] and [OH-] ions, respectively, rangefrom 1.0 x 10-1 M to 1.0 x 10-14 M. When [H+] is equal to [OH-] as when pure waterdissociates, the hydrogen ion concentration of pure water is equal to 1.0 x 10-7 M orpH = 7.00, defined as a neutral solution at 25°C, to be of temperaturedependent/endothermic dissociation.[H+] = [OH-] = 1.0 x 10-7 MWhen an ionic or polar substance is dissolved in water, it may change the relativenumbers of H+ and OH-. The higher the pH number, the lower the hydrogen ion concentration, and vice versa. Solution with an…arrow_forward
- Why is ethylene-di-amine tetra-acetic acid (EDTA) work as a base in complexometric titration? answer at your own words and answer should be to the point, not any irrelevant wordsarrow_forwardPrepared buffer solution GivenAmmonia Volume = 68 mL | Ammonia Concentration = 0.17 MAmmonium Chloride Volume = 42 mL | Ammonium Chloride Concentration = 0.13 MDissociation Constant of Ammonia: 1.8x10-5What is the total vol. of bufferarrow_forwardGiven the following acid-base indicators with pH ranges as shown below; PH ranges Methylorange 3.0 - 4.4 Methylred 4.4 - 6.3 Phenolphthalein 8.2 - 10 Select a suitable indicator for titrating: Sodium hydroxide and benzoic acid. Explain Aqueous ammonia and benzoic acid. Explainarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY