
Concept explainers
Interpretation:
The distinguishing mass spectra for the given molecules have to be interpreted. .
Concept Introduction:
Mass spectroscopy: It is a form of spectroscopic technique which is used for the elucidation of the molecular formula and molecular weight of the compound, depending upon the mass of the molecule.
Molecular formula: It represents the types of atoms with their total number present in a given molecule.
Molecular ion peak
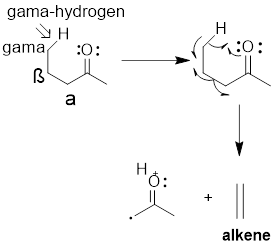
Mc Lafferty Rearrangement: This rearrangement can occur in
In this rearrangement a radical center in molecular ion derived from a lone pair or pi-bond removes hydrogen from the Gamma position (
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Essential Organic Chemistry (3rd Edition)
- Which of the following structures corresponds to the mass spectrum shown below?arrow_forwardDraw the structure of a compound with a molecular formula of C11H16 that generates the following spectra.arrow_forwardGiven the following mass spectra of compounds, give at least 2 fragments for each with the corresponding m/e values. With the molecular formula is given, predict the possible structure of the compound.(the answer is already given please exapand)arrow_forward
- Both of the mass spectra are of an aromatic compund. Identify the fragments.arrow_forwardgive the mass spectroscopy interpretation of the peaks Propan-1-amine / Propylaminearrow_forwardA carbonyl compound has a molecular ion with an m/z of 86. The mass spectra of this compound also has a base peak with an m/z of 57. Draw the correct structure of this molecule.arrow_forward
- Identify the base peak, the molecular ion, and C13 isotope in the mass spectrum shown belowarrow_forwardAssign the signals in the following spectra to the hydrogens that generate them.arrow_forwardThe mass spectrum of Compound D, with a molecular formula C8H7BrO2 is shown below. Identify and draw the structure of Compound D.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

