
Concept explainers
(a)
Interpretation:
The respective reactant (methyl substituted piperidine) and respective product (diene) should be given.
Concept introduction:
Generally
These quaternary ammonium salt under goes an elimination reaction easily.
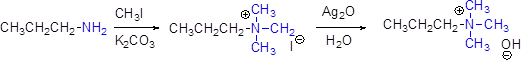
Hofmann elimination:
Quaternary ammonium ion undergoes elimination when using strong base like hydroxide ion this reaction is called as Hofmann elimination.
In the Hofmann elimination, abstraction of proton form β- carbon atom which is having more number of hydrogen to result the elimination product.

(b)
Interpretation:
The respective reactant (methyl substituted piperidine) and respective product (diene) should be given.
Concept introduction:
Hofmann elimination:
Quaternary ammonium ion undergoes elimination when using strong base like hydroxide ion this reaction is called as Hofmann elimination.
In the Hofmann elimination, abstraction of proton form β- carbon atom which is having more number of hydrogen to result the elimination product.

(c)
Interpretation:
The respective reactant (methyl substituted piperidine) and respective product (diene) should be given.
Concept introduction:
Hofmann elimination:
Quaternary ammonium ion undergoes elimination when using strong base like hydroxide ion this reaction is called as Hofmann elimination. Proton abstraction is takes place in β- carbon atom which is having more number of hydrogen.
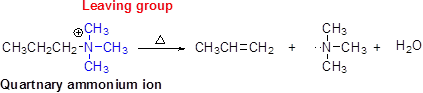
(d)
Interpretation:
The respective reactant (methyl substituted piperidine) and respective product (diene) should be given.
Concept introduction:
Generally amines can’t undergo elimination reaction, therefore amines can be converted in to quaternary ammonium halide by treating with ethyl iodide in basic solution of potassium carbonate, and this ammonium halide is treating with silver oxide to give ammonium hydroxide salt.
These quaternary ammonium salt under goes an elimination reaction easily.
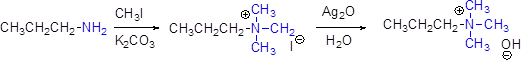
Hofmann elimination:
Quaternary ammonium ion undergoes elimination when using strong base like hydroxide ion this reaction is called as Hofmann elimination.
In the Hofmann elimination, abstraction of proton form β- carbon atom which is having more number of hydrogen to result the elimination product.

Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Pearson eText Organic Chemistry -- Instant Access (Pearson+)
- Please write down the structures for the main products of each of the following reactionsarrow_forwardDraw the structures of all monobromo derivatives of 2-methylpentane, C6H13Br, which have the bromine on a primary carbon.arrow_forwardWrite the monobromination products of 1,4-dimethylcyclohexane and calculate the percentages of each product. Which is the major product?arrow_forward
- You are required to synthesize 2-bromopentane from the reaction between an alkene with HBr. Which alkene, 1-pentene or 2-pentene, should you react with HBr in order to get 2-bromopentane? Give an explanation.arrow_forwardAcrolein and 1,3-cyclohexadiene react in a one-step concerted manner to yield a single product. Give the structure of the product. What kind of reaction is this an example of? In terms of this reaction, how would you classify acrolein? How would you classify 1,3-cyclohexadiene? Hint: acrolein is not a systematic name so you may need to look up its structure if you are not already familiar with it.arrow_forward(1) Predict the outcome of the addition of HBr to (a) trans-2-pentene, (b) 2-methyl-2-butene, and (c) 4-methylcyclohexene. How many isomers can be formed in each case? (2) Addition of HBr to 3,3-dimethyl-1-butene gives a mixture of two isomeric alkyl bromide products. Draw structures for the two products, and give a mechanistic explanation for their formation.arrow_forward
- Draw the structures for penta-1,4-diene and cyclopenta-1,3-diene.arrow_forward74. When piperidine undergoes the series of reactions shown here, 1,4-pentadiene is obtained as the product. When the four different methyl-substituted piperidines undergo the same series of reactions, each forms a different diene: 1,5-hexadiene; 1,4-pentadiene; 2-methyl-1,4-pentadiene; and 3-methyl-1,4-pentadiene. Which methyl-substituted piperidine forms which diene? 1. еxcess CHз!/К2CO3 2. Ag20, H20 3. Д 1. еxcess CHз!/K2CO3 2. Ag20, H20 CH3NCH,CH,CH;CH=CH2 3. A CH2=CHCH2CH=CH2 || `N' H CH3 1,4-pentadiene piperidinearrow_forward2-chloropropane is a major product of the reaction of chlorine with propane under ultraviolet light. Write the mechanism for this reaction including the initiation step and the two propagation steps.arrow_forward
- Draw the structure for these molecules. (a) 3-phenylmethylhex-4-en-1-yne; (b) 7-phenylcyclohepta-1,3,5-triene;(c) 3,5,6-trinitro-4-phenylmethylhepta-1,3,5-triene; (d) 5,5-dichloro-6-ethenyl-7-phenylcyclooct-3-en-1-ynearrow_forwardEthane reacts with chlorine (Cl2) in the presence of ultra-violet (UV) light, to produce 1-chloropropane (CH3CH2Cl) and hydrogen bromide (HCl). As shown in the equation below. CH3CH3 + Cl2 → CH3CH2Cl + HCl Describe the reaction mechanism of the reaction between ethane and chlorine to produce 1-chloroethane. The description should be detailed and must include the type of bond fission that takes placearrow_forwardEthane reacts with chlorine (Cl2) in the presence of ultra-violet (UV) light, to produce 1-chloropropane (CH3CH2Cl) and hydrogen bromide (HCl). As shown in the equation below. CH3CH3 + Cl2 → CH3CH2Cl + HCl Describe the reaction mechanism of the reaction between ethane and chlorine to produce 1-chloroethane. The description should be detailed and must include the type of bond fission that takes place. You may sketch and insert suitable diagrams to aid your description if you wish.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
