
ORGANIC CHEMISTRY (LL)-W/MOD.MASTERING.
8th Edition
ISBN: 9780134466712
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10.3, Problem 12P
Show how 1-propanol can be converted into the following compounds by means of a sulfonate ester:
- a. CH3CH2CH2SCH2CH3
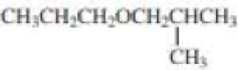
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the best set of reagents to achieve deoxygenation of 2-pentanone to pentane?
A. NaClO2/NaH2PO4
B. LiAlH4, Et2O
C. DIBAL-H, THF
D. NH2NH2/t-BuOK, DMSO
Give the major product when 2-bromobutanal reacts with acidic aqueous KCN.
What is the best choice of reagents to achieve the following reaction?
A. SOCl2 (1/2 equivalent)
B. KOH (pellets)
C. K2SO4
D. PhCO2Na
Chapter 10 Solutions
ORGANIC CHEMISTRY (LL)-W/MOD.MASTERING.
Ch. 10.1 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 10.1 - Explain the difference in reactivity between...Ch. 10.1 - Prob. 5PCh. 10.1 - Prob. 6PCh. 10.1 - Prob. 8PCh. 10.2 - Prob. 9PCh. 10.3 - Prob. 11PCh. 10.3 - Show how 1-propanol can be converted into the...Ch. 10.4 - Which of the following alcohols dehydrates the...Ch. 10.4 - Prob. 14P
Ch. 10.4 - Prob. 15PCh. 10.4 - Propose a mechanism for each of the following...Ch. 10.4 - Draw the product of each of the following...Ch. 10.4 - Explain why the following alcohols, when heated...Ch. 10.4 - What stereoisomers are formed in the following...Ch. 10.4 - Prob. 20PCh. 10.4 - What alcohol would you treat with phosphorus...Ch. 10.5 - Prob. 22PCh. 10.6 - What are the major products obtained when each of...Ch. 10.6 - Prob. 26PCh. 10.7 - Prob. 27PCh. 10.7 - Would you expect the reactivity of a five-membered...Ch. 10.7 - Prob. 29PCh. 10.7 - What products are obtained from the reaction of...Ch. 10.7 - Prob. 31PCh. 10.7 - Prob. 32PCh. 10.7 - Prob. 33PCh. 10.8 - Draw the mechanism for formation of the two...Ch. 10.8 - Prob. 35PCh. 10.8 - Prob. 36PCh. 10.8 - How do the major products obtained from...Ch. 10.8 - Explain why the two arene oxides in Problem 38...Ch. 10.8 - Which compound is more likely to be carcinogenic?Ch. 10.8 - Three arene oxides can be obtained from...Ch. 10.9 - Explain why the half-life (the time it takes for...Ch. 10.10 - Prob. 43PCh. 10.10 - Prob. 44PCh. 10.10 - Prob. 45PCh. 10.10 - Prob. 46PCh. 10.10 - Prob. 47PCh. 10.10 - Describe a synthesis for each of the following...Ch. 10.11 - Using an alkyl halide and a thiol as starting...Ch. 10.11 - The following three nitrogen mustards were studied...Ch. 10.11 - Why is melphalan a good cancer drug?Ch. 10.11 - Prob. 53PCh. 10.12 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 55PCh. 10 - Which compound is more likely to be carcinogenic?Ch. 10 - Prob. 57PCh. 10 - Prob. 58PCh. 10 - When heated with H2SO4, both...Ch. 10 - What is the major product obtained from the...Ch. 10 - Write the appropriate reagent over each arrow.Ch. 10 - What alkenes would you expect to be obtained from...Ch. 10 - Prob. 63PCh. 10 - Prob. 64PCh. 10 - When deuterated phenanthrene oxide undergoes a...Ch. 10 - An unknown alcohol with a molecular formula of...Ch. 10 - Explain why the acid-catalyzed dehydration of an...Ch. 10 - Prob. 68PCh. 10 - Prob. 69PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - What product would be formed if the four-membered...Ch. 10 - Which of the following ethers would be obtained in...Ch. 10 - Using the given starting material any necessary...Ch. 10 - Prob. 74PCh. 10 - When 3-methyl-2-butanol is heated with...Ch. 10 - Draw structures for compounds AF.Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - How could you synthesize isopropyl propyl ether,...Ch. 10 - When ethyl ether is heated with excess HI for...Ch. 10 - When the following seven-membered ring alcohol is...Ch. 10 - Ethylene oxide reacts readily with HO because of...Ch. 10 - Describe how each of the following compounds could...Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - Triethylene glycol is one of the products obtained...Ch. 10 - Prob. 85PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 87PCh. 10 - An ion with a positively charged nitrogen atom in...Ch. 10 - The following reaction takes place several times...Ch. 10 - Prob. 90PCh. 10 - Propose a mechanism for each of the following...Ch. 10 - A vicinal diol has OH groups on adjacent carbons....Ch. 10 - Prob. 93PCh. 10 - Prob. 94PCh. 10 - Two stereoisomers are obtained from the reaction...Ch. 10 - Propose a mechanism for each or the following...Ch. 10 - Triethylenemelamine (TEM) is an antitumor agent....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What products would you expect from the oxidation of thefollowing compounds with (i) CrO3 in aqueous acid? (ii) withPCC?(a) tert-butanol(b) cyclohexanol(c) cyclohexanonearrow_forwardShow how the following compounds can be prepared from benzene: a. benzaldehyde c. 1-bromo-2-phenylethane e. aniline b. styrene d. 2-phenylethanol f. benzoic acidarrow_forwardWhat products would you expect from oxidation of the following compounds with CrO3 in aqueous acid? With the Dess-Martin periodinane?(a) 1-Hexanol(b) 2-hexanol(c) hexanalarrow_forward
- Draw a structural formula for the product formed by treating butanal with each reagent. (a) LiA1H4LiA1H4 followed by H2OH2O (b) NaBH4NaBH4 in CH3OH/H2O (c) H2/Pt (d) Ag(NH3)2+in NH3/H2O (e) H2CrO4, heat (f) HOCH2CH2OH,HClarrow_forwardUndecanal is commercially available, but 6-methyl-1-bromoheptane is not. Propose two methods for the synthesis of 6-methyl-1-bromoheptane using precursors containing fewer than five carbon atoms (Cn n < 5). A protecting group will probably be required.arrow_forward1. (a) Oxidation of 1-butanol with PCC will produce which of the following? 2-chlorobutanal butanone butanoic acid butanal (b) Which synthetic route is the best way to prepare ethyl isopropyl ether? CH3CH2OH + (CH3)2CHOH + H2SO4, 140 oC CH3CH2ONa + (CH3)2CHOH CH3CH2ONa + (CH3)2CHBr (CH3)2CHONa + CH3CH2Brarrow_forward
- 1. (a) Oxidation of 1-butanol with PCC will produce which of the following? 2-chlorobutanal butanone butanoic acid butanal (b) Which synthetic route is the best way to prepare ethyl isopropyl ether? CH3CH2OH + (CH3)2CHOH + H2SO4, 140 oC CH3CH2ONa + (CH3)2CHOH CH3CH2ONa + (CH3)2CHBr (CH3)2CHONa + CH3CH2Br (c) The Williamson ether synthesis involves nucleophilic substitution involving an alkyl halide and an alcohol. (True or False)arrow_forwardGive the products formed when benzaldehyde and benzoic acid are treated with the given reagents. a. Tollen’s reagentb. phenylhydrazine, H+c. HCNd. NH2OHe. 1 mole H2, Nif. 1 mole CH3OH, H+g. LiAlH4 then H2O, H+h. 2 moles CH3OH, H+i. CH3MgCl, then H2O, H+j. H2Oarrow_forwardHow would you convert the following :(i) Phenol to benzoquinone(ii) Propanone to 2-methylpropan-2-ol(iii) Propene to propan-2-olarrow_forward
- Given the following reagents: a. Br₂ in CH₂Cl₂ b.CrO₃ , H₂SO₄, acetone c. concentrated HCl with ZnCl₂ d. aqueous FeCl₃ e. aqueous NaHCO₃ f. ammoniacal AgNO₃ which will match and quickly differentiate the compounds in the ff: 1. 4-bromophenol from benzene 2. hexane from 1-hexene 3. 1-hexanol from hexanal 4. hexane from hexanoic acid 5. 2-hexanol from 2-methyl-2-hexanol 6. 1-hexene from 1-hexynearrow_forwardWhat would be the major product if diethyl malonate (EtO2CCH2CO2Et) was decarboxylated using H3O+ and heat? A. CH3CH2CO2H B. CH3CO2H C. CH2(CO2H)2 D. CH3CO2Etarrow_forward1. Which compound is expected to have a higher melting point, 1-pentanol or 1-pentanal? Explain 2. Which compound is expected to be more soluble in water, propanal or butanal? Explain. 3. Why is acetyl chloride more reactive towards hydrolysis than ethyl acetate?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License