
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10.3C, Problem 10.4P
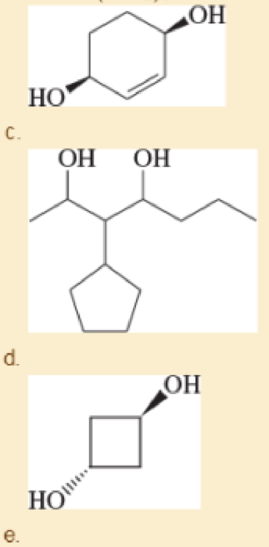
Give a systematic (IUPAC) name for each
- a. CH3CH(OH)(CH2)4CH(OH)C(CH3)3
- b. HO—(CH2)8—OH
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Give three reasons why ethers make good laboratory reagents
Which alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3l
Which of the following alcohols can be prepared from a Grignard reagent and ethylene oxide?
A. only 1
B. only 1 and 2
C. only 1, 2 and 3
D. 1, 2, 3 and 4
Chapter 10 Solutions
Organic Chemistry (9th Edition)
Ch. 10.3A - Prob. 10.1PCh. 10.3B - Give both the IUPAC name and the common name for...Ch. 10.3B - Prob. 10.3PCh. 10.3C - Give a systematic (IUPAC) name for each diol. a....Ch. 10.4B - Predict which member of each pair will be more...Ch. 10.4B - Dimethylamine (CH3)2NH, has a molecular weight of...Ch. 10.6A - Prob. 10.7PCh. 10.6A - Prob. 10.8PCh. 10.6C - A nitro group (NO2) effectively stabilizes a...Ch. 10.6C - Prob. 10.10P
Ch. 10.8B - Prob. 10.11PCh. 10.8B - Prob. 10.12PCh. 10.9A - Prob. 10.13PCh. 10.9B - Prob. 10.14PCh. 10.9C - Show how you would synthesize each tertiary...Ch. 10.9D - Prob. 10.16PCh. 10.9D - Show how you would add Grignard reagents to acid...Ch. 10.9D - A formate ester, such as ethyl formate, reacts...Ch. 10.9E - Prob. 10.19PCh. 10.9E - In Section9-7B, we saw how acetylide ions add to...Ch. 10.9F - Prob. 10.21PCh. 10.10A - Prob. 10.22PCh. 10.10B - Prob. 10.23PCh. 10.11B - Predict the products you would expect from the...Ch. 10.11B - Prob. 10.25PCh. 10.11B - Prob. 10.26PCh. 10.12 - Prob. 10.27PCh. 10.12 - Prob. 10.28PCh. 10.12 - Authentic skunk spray has become valuable for use...Ch. 10 - Give a systematic (IUPAC) name for each alcohol....Ch. 10 - Give systematic (IUPAC) names for the following...Ch. 10 - Draw the structures of the following compounds...Ch. 10 - Predict which member of each pair has the higher...Ch. 10 - Predict which member of each pair is more acidic,...Ch. 10 - Predict which member of each group is most soluble...Ch. 10 - Draw the organic products you would expect to...Ch. 10 - Prob. 10.37SPCh. 10 - Show how you would synthesize the following...Ch. 10 - Show how you would use Grignard syntheses to...Ch. 10 - Show how you would accomplish the following...Ch. 10 - Show how you would synthesize the following: a....Ch. 10 - Complete the following acid-base reactions. In...Ch. 10 - Prob. 10.43SPCh. 10 - Prob. 10.44SPCh. 10 - Geminal diols, or 1,1-diols, are usually unstable,...Ch. 10 - Vinyl alcohols are generally unstable, quickly...Ch. 10 - Compound A (C7H11Br) is treated with magnesium in...Ch. 10 - Prob. 10.48SPCh. 10 - Prob. 10.49SPCh. 10 - Prob. 10.50SPCh. 10 - Prob. 10.51SPCh. 10 - Prob. 10.52SPCh. 10 - Prob. 10.53SPCh. 10 - Prob. 10.54SPCh. 10 - Prob. 10.55SPCh. 10 - Prob. 10.56SPCh. 10 - Show how this 1 alcohol can be made from the...Ch. 10 - Prob. 10.58SPCh. 10 - Prob. 10.59SPCh. 10 - Prob. 10.60SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
4. 38 Strontium has four naturally occurring isotopes, with mass numbers 84, 86, 87, arid 88.
a. Write the atom...
Basic Chemistry (5th Edition)
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
The chapter sections to review are shown in parentheses at the end of each problem. A "chemical-free” shampoo i...
Basic Chemistry
45. Calculate the mass of nitrogen dissolved at room temperature in an 80.0-L home aquarium. Assume a total pre...
Chemistry: Structure and Properties
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Arrange these compounds in order of increasing boiling point. (a) 1-butanol, butane, diethylether (b) hexane, 1-hexanol, dipropyletherarrow_forwardShow how each alcohol or diol can be prepared from an alkene. (a) 2-Pentanol (b) 1-Pentanol (c) 2-Methyl-2-pentanol (d) 2-Methyl-2-butanol (e) 3-Pentanol (f) 3-Ethyl-3-pentanol (g) 1,2-Hexanediolarrow_forward1. O-hydroxybenzoic acid is a major product formed with phenol and which other reactant/s I-primary alcohol II-sodium hydroxide III-water IV-carbon dioxide A.I and III B. I and IV C. II and III D. II and IVarrow_forward
- 1. Which alcohol has a higher boiling point?a. (i) 2-methylpropan-2-ol or (ii) butan-2-olb. (i) hexan-1-ol or (ii) 3,3-dimethylbutan-1-olarrow_forwardGive the structure corresponding to each IUPAC name. 2,4 dimethyl- 2 hexanolarrow_forwardWhy is H3C-O-SiH3 ether a weaker base than (CH3)3O?arrow_forward
- 1. Which compound is more soulable in water : CH3 CH2 CH2 CH2 CH2 CH3 OR CH3 CH2 CH2 CH2 COOH 2. Which compound is more soulable in organic solvent : CH3 CH2 CH2 CH2 CH2 CH3 OR CH3 CH2 CH2 CH2 COOHarrow_forwardDraw the product resulting from mild oxidation of (a) 2-butanol; (b) 2-methylpropanal; (c) cyclopentanol.arrow_forwardWhat is the correct reagent?arrow_forward
- n-Butyl methyl ether is an isomer of MTBE and has a boiling point of 70 oC. Explain why the boiling point is significantly different compared to MTBE.arrow_forwardList the reagents necessary to produce the given product.arrow_forwardShow how each alcohol or diol can be prepared from an alkene.(a) 2-Pentanol (b) 1-Pentanol (c) 2-Methyl-2-pentanol(d) 2-Methyl-2-butanol (e) 3-Pentanol (f) 3-Ethyl-3-pentanol(g) 1,2-Hexanedioarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY