
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Question
Chapter 10.6, Problem 6LTS
Interpretation Introduction
Interpretation:
The stereochemical outcome of radical bromination of the following
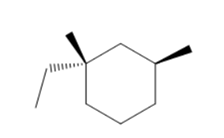
Concept Introduction :
As a branch of chemistry, stereochemistry focuses on modifying and studying the relative spatial arrangement of the atoms that constitute molecules.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The bicyclic alkene P can be prepared by thermal electrocyclic ringclosure from cyclodecadiene Q or by photochemical electrocyclic ringclosure from cyclodecadiene R. Draw the structures of Q and R, andindicate the stereochemistry of the process by which each reactionoccurs.
Predict the product(s) of the following Diels-Alder reaction. Include stereochemistry if appropriate.
Explain the stereochemistry of E2 eliminations to form alkenes, and predict the products of E1 reactions on stereoisomers and on cyclohexane systems.
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Similar questions
- Optically active (R)-2-bromobutane can be converted to 2-butanol under either conditions A or conditions B. Describe the stereochemistry of the product solutions for the two different conditions.arrow_forwardPredict the products for the hydrohalogenation reaction of 1-methyl-1,3-cyclohexadiene with 1 equivalent of HBr in thermodynamic conditions.arrow_forwardPredict the products and include stereochemistry.arrow_forward
- Explain the stereochemistry of the product of the Diels-Alder reaction shown below:arrow_forwardPredict the products and mechanisms of the following reactions. When more than oneproduct or mechanism is possible, explain which are most likely. ) 1-bromo-1-methylcyclopentane heated in methanolarrow_forwardUse the extended version of Markovnikov’s rule to predict the regiochemistry (orienta- tion) of electrophilic additions to alkenes.arrow_forward
- For each one of the following reactions, show step by step mechanism of the reactions and indicate the stereochemistry of the products and their relationshiparrow_forwardWrite the mechanisms of the following reactions and examine the products as stereoisomeric.arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs due to a nucleophilic substitution pathway. The reaction is 100% stereospecific. Please explain what this observation tells about the mechanism of the reaction.arrow_forward
- The following triene undergoes an intramolecular Diels-Alder reaction to give a bicyclic product. Propose a structural formula for the product. Account for the observation that the Diels-Alder reaction given in this problem takes place under milder conditions (at lower temperature) than the analogous Diels-Alder reaction shown in Problem 20.34.arrow_forwardWhen 2-2-dimethylbutane undergoes free radical chlorination, four monohalogenated products are formed. Calculate the expected % yield of (R)-3-chloro-2,2-dimethylbutane.arrow_forwardConsider the substitution reaction that takes place when (R)-3-bromo-3-methylhexane is treated with sodium methoxide. Deduce the mechanism of this reaction working step by step. Write all possible products showing the stereochemistry.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning