
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Question
Chapter 10.3, Problem 4LTS
Interpretation Introduction
Interpretation:
A mechanism for the radical chlorination of methyl chloride to produce methylene chloride needs to be drawn:
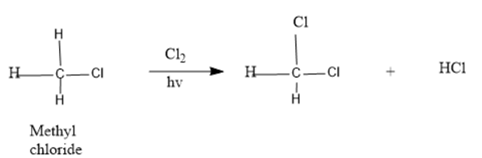
Concept Introduction :
Free-radical halogenation is a form of halogenation in organic chemistry.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are the mechanism of the following reaction?
What is the mechanism of the reaction below.
Draw the mechanism for the reaction of 1-Methylcyclohexene with hydrogen bromide.
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Similar questions
- In the presence of a radical initiator (Z•), tributyltin hydride (R3SnH, R = CH3CH2CH2CH2) reduces alkyl halides to alkanes: R′X + R3SnH → R′H + R3SnX. The mechanism consists of a radical chain process with an intermediate tin radical: This reaction has been employed in many radical cyclization reactions. Draw a stepwise mechanism for the following reaction.arrow_forwardDraw the electron-pushing mechanism for each radical reaction below. Identify each step as initiation, propagation, or termination.arrow_forwardWhat is the mechanism and product for the following reaction?arrow_forward
- The following radical-initiated reaction with HBr produces a formal Anti-Markovnikov's rule product. Provide a curved arrow mechanism, making sure to use single headed arrows and drawing all intermediates. Only initiation and propagation steps are needed. Termination does not need to be included. Part of the initiation step is provide for use to generate an OH radical.arrow_forwardDecide on the mechanism for the following substitution, and then pick the solvent that affords the faster reaction. (CH3CH2)2CHCl + CH3O− in CH3OH or HMPAarrow_forwardChemistry Glutathione (or GSH) can form a disulfide bridge with another glutathione molecule via radical reaction. Show a mechanism of a hydroxide radical reacting with two glutathione molecules to a produce a dimer (GSSG).arrow_forward
- 3-methyl-2-butanol forms 2-bromo-2-methylbutane as a single product with HBr. This reactionWrite down the mechanism.arrow_forwardPropose a mechanism for the free-radical chlorination of ethane,CH3¬CH3 + Cl2 ¡hv CH3¬CH2Cl + HClarrow_forwardModify the given copy of the starting material to draw the major radical bromination product(s), with close attention paid to the stereochemical outcome. If more than one stereoisomer is expected, then draw it.arrow_forward
- Radical bromination of cyclohexene using NBS gives 3-bromo-1-cyclohexene. Draw the allylic radical intermediate formed during this reaction, showing both resonance structuresarrow_forwardDraw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br and Br2.arrow_forwardWhat is the reaction mechanism for the following reaction?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
