
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 10.2, Problem 3LTS
Interpretation Introduction
Interpretation:
The appropriate fishhook arrows for the following radical process need to be drawn:
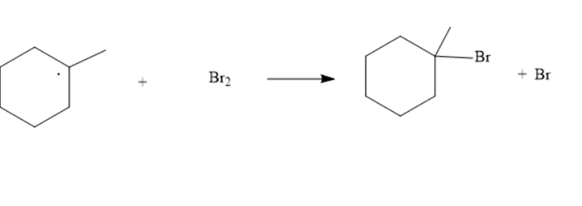
Concept Introduction :
When the bonding cannot be represented by a single Lewis formula, resonance is a means to describe delocalized electrons inside specific molecules or polyatomic ions. With such delocalized electrons, a molecule or ion can be represented by a number of resonance structures.
A radical substitution reaction is one in which one or more of the atoms or groups present in the substrate are replaced by new atoms or groups using a free radical mechanism. A radical chain reaction's initiation step is the point at which a free radical is created for the first time.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the following radicals in order from least to most selective.
R
please draw out both mechanisms for part a&b
What is the mechanism and reagents for this reaction? with the circled molecule being attacked first since it more electronegative.
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the electron-pushing mechanism for each radical reaction below. Identify each step as initiation, propagation, or termination.arrow_forwardPlease draw the COMPLETE electron pushing arrow mechanism! ALL STEPS!arrow_forwardDraw curved arrows for each step of the following mechanism: (SEE IMAGE)arrow_forward
- Draw the mechanism for the following reaction with arrows and include all intermediates. Include formal charges and lone pairs if needed.arrow_forwardDraw the mechanism arrows for both propagation steps for the radical addition of HBr to the alkene.arrow_forwardQ;Provide missing curved arrows and draw the product of the radical addition mechanistic step. Add lone pairs and radicals to all appropriate species. Provide missing curved arrows. Make sure to use the correct curved arrows.arrow_forward
- Radicals in some instances can lead to undesired reactions foods are known to cause radical Inhibitors known as preservatives or antioxidants these Species Act by preventing radical chain reactions that can lead to degradation of packaged food. One synthetic preservative used commonly in both the food industry and the chemical industry is BHT. Draw the propagation of steps for the reaction of BIT with an all my radical.arrow_forwardWhat is the exact mechanism with the arrows for the dianion becoming the ketone? Does a loss of water occur/an additional protonation?arrow_forwardHomolysis of the indicated C–H bond in propene forms a resonancestabilizedradical. Question: Draw the two possible resonance structures for this radical.arrow_forward
- Which of the following mechanisms is consistent with the reaction profile shown above? Circle the correct mechanism and justify your choice.arrow_forwardSolve correctly please. answers circled are 100% correct. pls draw mechanism as to whyarrow_forwardWhat is the the resonance contributor(s) for each of the following radicales?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
