
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Question
Chapter 10.7, Problem 7LTS
Interpretation Introduction
Interpretation:
The products that are obtained when methylene cyclohexane is treated with NBS and irradiated with UV light:
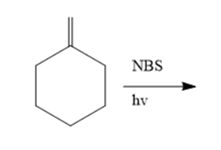
Concept Introduction :
N-Bromo succinimide (NBS) with cyclohexene will combine to generate 3-bromocyclohexene. The radical initiator will be benzoyl peroxide (BPO), and the solvent will be cyclohexane.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the major products of the following reactions. Include appropriate stereochemistry in the product structures.
S-1,3-dimethylcyclohexene + aqueous Hg(OAc)2Hg(OAc)2 followed by treatment with NaBH4
Predict the products of the following reactions.(a) sec-butyl isopropyl ether + concd. HBr, heat
Predict the major products of the following reactions. o@xylene + H2 (1000 psi, 100 °C, Rh catalyst)
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Similar questions
- Q2. What are the products of the following reactions? Q3. 2-Bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant? arrow_forwardAccount for the regioselectivity and stereoselectivity observed when 1-methylcyclopentene is treated with reagent Q.Hg(OAc)2 in H2Oarrow_forwardPredict the major products of the following reactions.(a) 1@methylcyclohexene + aqueous Hg(OAc)2 (b) the product from part (a), treated with NaBH4arrow_forward
- Predict the major products (including stereochemistry) when cis-3-methylcyclohexanol reacts with the following reagents. ) TsCl/pyridine, then NaBrarrow_forwardWhat order of reagents should be used to synthesize the following product from methylcyclopentane?(A) Br2(B) Br2, hv(C) KOH, heatarrow_forwardTranexamic acid, a drug useful against blood clotting, is prepared commercially from p-methylbenzonitrile. Formulate the steps likely to be used in the synthesis. (Don’t worry about cis-trans isomers; heating to 300 C interconverts the isomers.)arrow_forward
- Treatment of p-bromotoluene with NaOH at 300°C yields a mixture of two products, but treatment of m-bromotoluene with NaOH yields a mixture of three products. Explain.arrow_forwardA step in a synthesis of PGE1 (prostaglandin E1, alprostadil) is the reaction of a trisubstituted cyclohexene with bromine to form a bromolactone. Propose a mechanism for formation of this bromolactone and account for the observed stereochemistry of each substituent on the cyclohexane ring. Alprostadil is used as a temporary therapy for infants born with congenital heart defects that restrict pulmonary blood flow. It brings about dilation of the ductus arteriosus, which in turn increases blood flow in the lungs and blood oxygenation.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning