
Concept explainers
(a)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(a)
Explanation of Solution
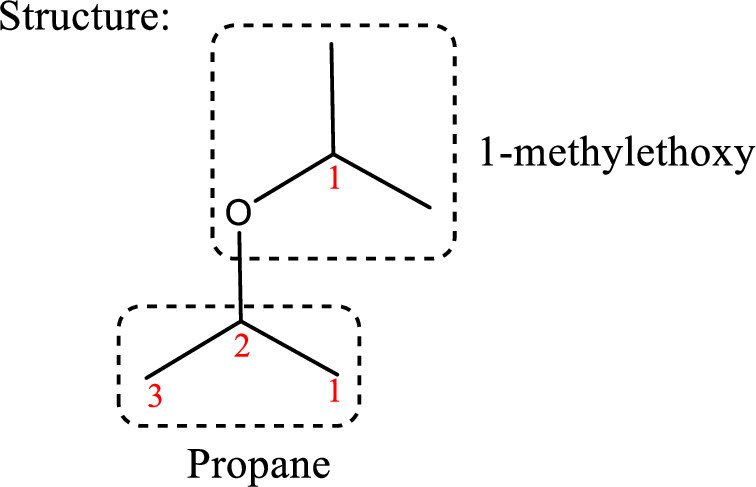
IUPAC name: 2-(1-Methylethoxy)propane
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Using the above principle, the longest carbon chain is propane (contains three carbons) as the parent alkane; naming the –OR group as alkoxy that is ‘2-(1-methylethoxy).
Therefore, the structure obtained is,
(b)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(b)
Explanation of Solution
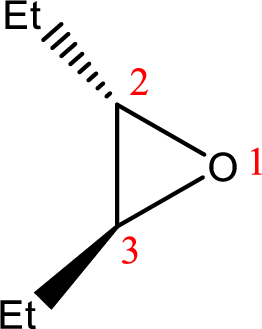
IUPAC name: trans-2,3-Diethyloxirane
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
Using the above principle, the name‘oxirane’ contains two carbons along with one Oxygen atom in a cyclic ringalong with two ‘ethyl’ substituentsin the ring.
(c)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(c)
Explanation of Solution
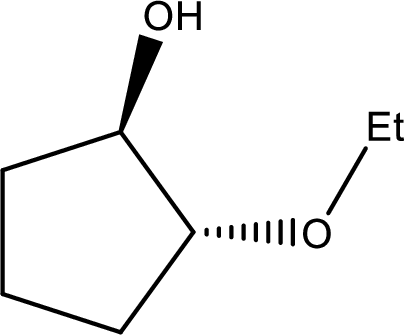
IUPAC name:trans-2-Ethoxycyclopentanol
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Here, the –OH group is preferred as the first priority and the compound is ‘alcohol’.
In the given compound, the longest carbon chain contains five carbons in a cyclic ring; the parent name is CYCLOPETANE and numbering the parent chain begins with carbon attached to the –OH group located at C-1 and the substituent ‘ethoxy’ is located at C-2.
Therefore, the structure is,
(d)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(d)
Explanation of Solution
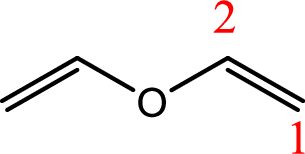
IUPAC name:Ethenyloxyethene.
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Using the above principle, the longest carbon chain isethene (contains two carbons with a double bond) as the parent
(e)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(e)
Explanation of Solution
IUPAC name:Cyclohexene oxide:
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
Using the above principle, the longest carbon cyclic ring contains sixcarbon atoms andone Oxygen atom is located at C-1 and 2 as three membered ring.
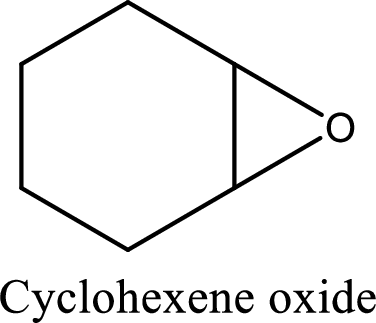
(f)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(f)
Explanation of Solution
IUPAC name:3-Cyclopropyloxy-1-propene
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Using the above principle, the longest carbon chain ispropene (contains three carbons with a double bond) as the parent alkene; naming the–OR groups as alkoxy that is 3-Cyclopropyloxy.
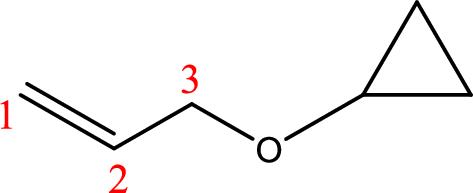
(g)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(g)
Explanation of Solution
IUPAC name:(R)-2-Methyloxirane
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
Using the above principle, the longest three membered cyclic ringscontaintwo carbon atoms and one Oxygen atom and the substituent methyl group is located at C-2.
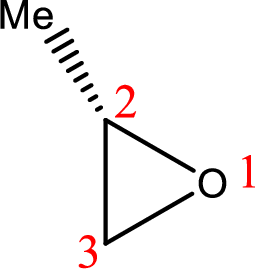
(h)
Interpretation:
Structural formula for the given name has to be drawn.
Concept Introduction:
Nomenclature of Ether compounds:
The naming of theorganic compound is given by IUPAC (International Union for pure and applied chemistry). In the IUPAC names consist of certain rules for giving chemical names they are,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Common name: List the alkyl groups bonded to Oxygen in alphabetical order and adding the word ‘ether’.
Cyclic ether: The presence of Oxygen atom in a saturated ring is indicated by the prefix ox-, and ring sizes from three to six are indicated by the endings –irane, etane, olane, and –ane, respectively.
Numbering of the atoms of the ring begins with the oxygen atom. These compounds and others in which there is a heteroatom (non-carbon atom) in the ring are called heterocycles.
(h)
Explanation of Solution
IUPAC name:1,1-Dimethyloxycyclohexane,
Ethers are named by selecting the longest carbon chain as the parent alkane and naming the –OR group bonded to it an alkoxy group.
Using the above principle, the longest carbon ringiscyclohexane (contains six carbons) as the parent alkane; naming the two–OR groups as alkoxy that bothmethoxy groups located at C-1.
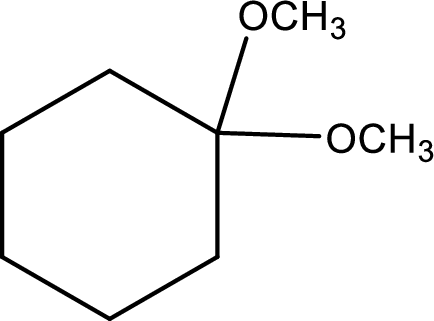
Want to see more full solutions like this?
Chapter 11 Solutions
Organic Chemistry
- Give a simple chemical test that can differentiate the two compounds from each other. Specify the reagent(s) needed and state what you expect to see. benzene and ethylbenzene 1 – butyne and 2 – butyne 2 – methylpentane and 2 - methylcyclohexenearrow_forwardAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst. Hydrocarbon A also reacts with OsO4 to give diol B. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2CO2H, and the other fragment is ketone C. What are the structures of A, B, and C? Write all reactions, and show your reasoning.arrow_forwardYou are required to synthesize 2-bromopentane from the reaction between an alkene with HBr. Which alkene, 1-pentene or 2-pentene, should you react with HBr in order to get 2-bromopentane? Give an explanation.arrow_forward
- Starting with acetylene and ethylene oxide as the only sources of carbon atoms, show how to prepare the compound Q.)1,6-Hexanediolarrow_forwardHydrocarbon A, C8H12 absorbs 3 equiv. of hydrogen to give B, C8H18 when hydrogenated over a Pd/C catalyst. Treatment of A with aqueous H2SO4 and Hg(II) gives a single ketone, C. Oxidation of A with KMnO4 gives CO2 and the two carboxylic acids D & E shown below. What would be a structure for A?arrow_forwardAn unknown hydrocarbon A with the formula C6H12 reacts with 1 molar equivalent of H2 over a palladium catalyst to give hydrocarbon B. Hydrocarbon A also reacts with OsO4 to give the glycol C. When oxidized with KMnO4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH2COOH, and the other fragment is ketone D (R2C=O). What are the structures of A, B, C and D? Write all reactions.arrow_forward
- Propose the structure of the following: a. An alkane, C6H14 b. A crylic saturated hydrocarbon, C6H12 c. A diene (dialkene), C5H8 d. A keto alkene, C5H8Oarrow_forwardThree constitutional isomers of molecular formula C 5H 8O can be converted to 1-pentanol (CH 3CH 2CH 2CH 2CH 2OH) on treatment with two equivalents of H 2 in the presence of a Pd catalyst. Draw the structures of the three possible compounds, all of which contain a carbonyl grouparrow_forwardAn unknown hydrocarbon Q has a formula C6H12. Q reacts with osmium tetroxide to give a diol R when oxidized with KMnQ4 in an acidic medium Q gives two products. One product is propanoic acid and the other is ketone S. Provide reaction equations to identify the possible structures of Q, R and S.arrow_forward
- Compound AA has a molecular formula of C3H6O and gives a positiveresult using Tollen’s reagent. The reaction of compound AA with hotacidified potassium permanganate, KMnO4 gives compound BB. Thecatalytic hydrogenation of compound AA with nickel, Ni producedcompound CC. The reaction of compound BB with ethanamine,CH3CH2NH2 produces compound DD I) Draw the structural formula of compounds AA, BB, CC and DD. 2)Name the type of chemical reaction for the formation of compound CC.arrow_forwardAcid-catalyzed hydration of 2-Methyl-1-butene yields two alcohols. The major product does not undergo oxidation, while the minor product will undergo oxidation. Explain why, by showing the structures of theproducts.arrow_forwarda) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomeric products are formed. Give their structures, and label each asymmetric carbon atom as (R) or (S). What is the relationship between these isomers?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





