
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.9, Problem BQ
Interpretation Introduction
Interpretation:
The product distribution that tells a chemist about the mechanism of the acid-catalyzed epoxide ring opening has to be discussed.
Concept Introduction:
Stereochemistry: The spatial arrangement of atoms or groups present in compound.
Ring opening: Ring opening of epoxide takes place by acid-catalyzed hydrolysis of an epoxide.
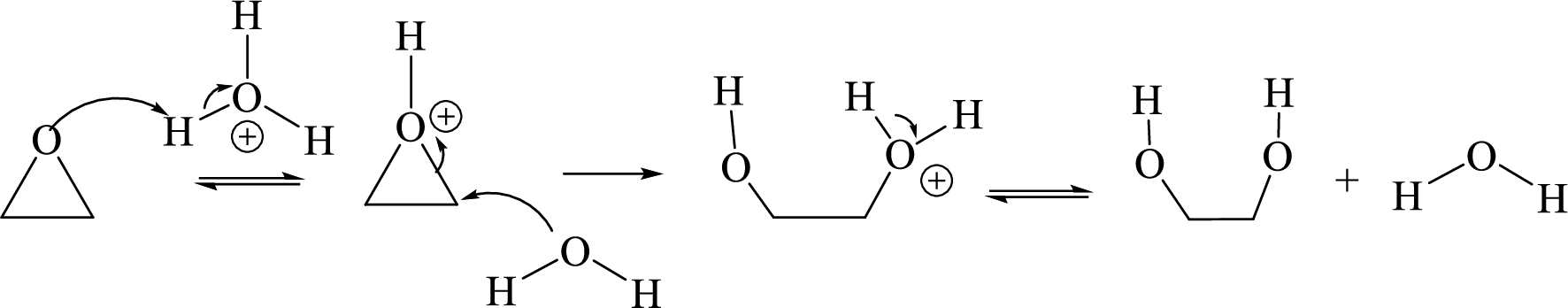
Acid-catalyzed hydrolysis of an epoxide:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Enolates are formed by deprotonation of an α-carbon hydrogen. Answer the following questions about enolate formation.
In the molecule shown, select the α-carbon hydrogen that would be removed to form an enolate when NaOEt is used as a base.
Draw the thermodynamic enolate that results for the molecule in Part 1. Draw only the enolate resonance form that includes a formal charge on the α carbon. Be sure to indicate that formal charge as well as any lone pair of electrons in your answer.
Rank the primary, secondary, tertiary, and aryl halides in terms of reactivity with the nucleophile. Which type of halide react faster by an SN2 reaction? Explain
Arrange these compounds in increasing order of reactivity in SN2 nucleophilic substitution?
Chapter 11 Solutions
Organic Chemistry
Ch. 11.2 - Write IUPAC and common names for these ethers. (a)...Ch. 11.3 - Arrange these compounds in order of increasing...Ch. 11.4 - Show how you might use the Williamson ether...Ch. 11.4 - Show how ethyl hexyl ether might be prepared by a...Ch. 11.5 - Account for the fact that treatment of tert-butyl...Ch. 11.5 - Draw structural formulas for the major products of...Ch. 11.6 - Prob. 11.7PCh. 11.8 - Draw the expected products of Sharpless...Ch. 11.9 - Prob. AQCh. 11.9 - Prob. BQ
Ch. 11.9 - Prob. CQCh. 11.9 - Prob. DQCh. 11 - Write names for these compounds. Where possible,...Ch. 11 - Prob. 11.11PCh. 11 - Each compound given in this problem is a common...Ch. 11 - Account for the fact that tetrahydrofuran (THF) is...Ch. 11 - Prob. 11.14PCh. 11 - Write equations to show a combination of reactants...Ch. 11 - Propose a mechanism for this reaction.Ch. 11 - Prob. 11.17PCh. 11 - Prob. 11.18PCh. 11 - Prob. 11.19PCh. 11 - Prob. 11.20PCh. 11 - Ethylene oxide is the starting material for the...Ch. 11 - Prob. 11.22PCh. 11 - Predict the structural formula of the major...Ch. 11 - The following equation shows the reaction of...Ch. 11 - Propose a mechanism to account for this...Ch. 11 - Acid-catalyzed hydrolysis of the following epoxide...Ch. 11 - Prob. 11.27PCh. 11 - Prob. 11.28PCh. 11 - Prob. 11.29PCh. 11 - Propose a mechanism for the following...Ch. 11 - Show reagents and experimental conditions to...Ch. 11 - Starting with cis-3-hexene, show how to prepare...Ch. 11 - Show reagents to convert cycloheptene to each of...Ch. 11 - Show reagents to convert bromocyclopentane to each...Ch. 11 - Prob. 11.35PCh. 11 - Starting with acetylene and ethylene oxide as the...Ch. 11 - Following are the steps in the industrial...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - Aldehydes and ketones react with one molecule of...Ch. 11 - Prob. 11.42PCh. 11 - Write the products of the following sequences of...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - Using your reaction roadmap as a guide, show how...Ch. 11 - During the synthesis of the antiasthmatic drug...Ch. 11 - Prob. 11.48P
Knowledge Booster
Similar questions
- Why does increasing alkyl substitution increase the rate of an E2 reaction?arrow_forwardIn the chemical reaction between crotyl chloride and ethanolic silver nitrate solution, a precipitate is definitely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially within this SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.arrow_forwardEnolates are formed by deprotonation of an α-carbon hydrogen. Answer the following questions about enolate formation. In the molecule shown, select the α-carbon hydrogen that would be removed to form an enolate when sodium hydroxide is used as a base. (use photo to answer this question). Then, Draw the thermodynamic enolate that results for the molecule in Part 1. Draw only the enolate resonance form that includes a formal charge on the α carbon. Be sure to indicate that formal charge as well as any lone pair of electrons in your answer.arrow_forward
- Which of the following statements is true about SN1 reactions of alkyl halides? Select one: a. Complete inversion of configuration occurs b. The reaction rate depends only on the concentration of the nucleophile c. These reactions are favored by nonpolar solvents d. These reactions are favored by polar solventsarrow_forwardIn the chemical reaction between benzyl chloride and ethanolic silver nitrate solution, a precipitate is barely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially within this SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.arrow_forwardIn the chemical reaction between bromocyclopentane and ethanolic silver nitrate solution, a precipitate is definitely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially within this SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.arrow_forward
- Why is the alkyl bromide substrate below not capable of undergoing an E2 elimination reaction upon treatment with potassium hydroxide (KOH) in ethanol (EtOH)? -Br– is too poor a leaving group. -Too much angle strain would be present in the alkene product. -Potassium hydroxide is a poor base to use in E2 reactions. -An anti-periplanar E2 elimination cannot occur due to the lack of a beta-hydrogen in the substratearrow_forwardin a reaction where trans-stilbene reacts with a molecule of bromine while using ethanol as a solvent, what would happen if the ethanol was a more reactive nucleophile than the bromide ion(s)? would the product be affected?arrow_forwardIn the chemical reaction between bromocyclohexane and ethanolic silver nitrate solution, a precipitate is definitely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially within this SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.arrow_forward
- 1. Provide the mechanism of the reaction shown in the picture. 2. Explain why it cannot be done in basic conditions. 3. Why is the enol tautomer favored over the keto tautomer?arrow_forward12c. If you could, please provide an explaination for why the major product would be the SN2 product rather than E2 product?arrow_forwardIn the chemical reaction between 1-Bromobutane and ethanolic silver nitrate solution, a precipitate is barely formed. Using the skeletal structures of the molecules, write out the skeletal structure of each reaction (don't include the mechanism). Identify what the nucleophile is in each type of reaction, especially the SN1 reaction. Explain the reactivity of each electrophile/substrate in terms of whether they are tertiary, secondary, primary, etc. for each reaction. Include the overall chemical reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning